10.02.14
Direct Flow Medical Inc. has received CE mark for an enhanced transfemoral delivery system for the company’s transcatheter aortic valve system.
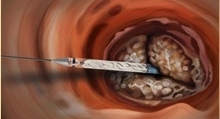
The enhanced delivery system offers an ergonomic handle designed for easy, precise and controlled delivery of the valve, according to the company. The system features a low-profile, ultra-flexible sheath that, in addition to the non-metallic valve. All of the company’s valves can be delivered through the same delivery system.
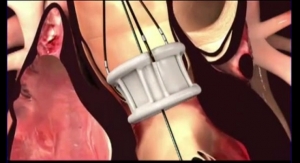
The company’s valve features a unique, double-ring design that conforms to the anatomy and creates a tight and durable seal around the annulus. The valves, according to the company, allow for complete assessment of hemodynamic performance and unlimited repositioning until optimal results are obtained. The company now has a valve portfolio that includes 23-, 25-, 27- and 29-millimeter (mm) valves, which can treat patients with annulus sizes from 19 mm to 28 mm.
“Direct Flow Medical continues to innovate with the focus on improving patient outcomes. The approval of the enhanced Direct Flow Medical delivery system is further evidence of our commitment to the medical community that treats TAVI (transcatheter aortic valve implant) patients,” said Charles Davidson, M.D., Direct Flow’s chief medical officer. “We continue to deliver on our promise to develop innovative products delivering the best patient outcomes, and continue to investigate new indications for our unique therapeutic platform.”
According to the company, its valve is designed to improve TAVI outcomes by eliminating aortic regurgitation (blood flow back through the valve), offering unlimited repositionability during the procedure and reducing overall complications.
“We have been able to access vessels as small as 5.2 mm with the Direct Flow Medical delivery system. The system is very flexible and tracks very well in highly calcified anatomies,” said Christoph Naber, M.D. from the Contilia Heart and Vascular Center in Essen, Germany. “The newly approved system offers enhanced precision and improved control for valve delivery while being extremely simple to use.”
The company’s TAVI system is available in Europe. In the United States, the company is moving toward regulatory approval and is enrolling patients in a pivotal trial.
Founded in 2004, Direct Flow Medical is based in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif. The company’s technology is not limited to aortic valve disease, and also is applicable to mitral and other heart valve anatomical sites.
The company is backed by venture capital firms.
The enhanced delivery system offers an ergonomic handle designed for easy, precise and controlled delivery of the valve, according to the company. The system features a low-profile, ultra-flexible sheath that, in addition to the non-metallic valve. All of the company’s valves can be delivered through the same delivery system.
The company’s valve features a unique, double-ring design that conforms to the anatomy and creates a tight and durable seal around the annulus. The valves, according to the company, allow for complete assessment of hemodynamic performance and unlimited repositioning until optimal results are obtained. The company now has a valve portfolio that includes 23-, 25-, 27- and 29-millimeter (mm) valves, which can treat patients with annulus sizes from 19 mm to 28 mm.
“Direct Flow Medical continues to innovate with the focus on improving patient outcomes. The approval of the enhanced Direct Flow Medical delivery system is further evidence of our commitment to the medical community that treats TAVI (transcatheter aortic valve implant) patients,” said Charles Davidson, M.D., Direct Flow’s chief medical officer. “We continue to deliver on our promise to develop innovative products delivering the best patient outcomes, and continue to investigate new indications for our unique therapeutic platform.”
According to the company, its valve is designed to improve TAVI outcomes by eliminating aortic regurgitation (blood flow back through the valve), offering unlimited repositionability during the procedure and reducing overall complications.
“We have been able to access vessels as small as 5.2 mm with the Direct Flow Medical delivery system. The system is very flexible and tracks very well in highly calcified anatomies,” said Christoph Naber, M.D. from the Contilia Heart and Vascular Center in Essen, Germany. “The newly approved system offers enhanced precision and improved control for valve delivery while being extremely simple to use.”
The company’s TAVI system is available in Europe. In the United States, the company is moving toward regulatory approval and is enrolling patients in a pivotal trial.
Founded in 2004, Direct Flow Medical is based in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif. The company’s technology is not limited to aortic valve disease, and also is applicable to mitral and other heart valve anatomical sites.
The company is backed by venture capital firms.