05.27.14
CardioComm Solutions Inc. has received U.S. Food and Drug Administration (FDA)
"Guava II offers a new level of regulatory credibility to ECG reading services, opening new opportunities to physicians and hospitals that wish to have ECG reviews completed that are adherent with all possible medical clearances. Physicians reading ECGs for, and hospitals receiving ECG reports generated from, ECG service providers may not be aware that an ECG system is being relied upon that may hold no current FDA clearance. Guava II will allow such service providers the option to customize their ECG management systems with an FDA compliant solution. Guava II will also offer significant data development and interoperability potential, will serve to streamline work flow, reduce staffing costs and improve the efficiency of identifying potentially life threatening arrhythmias in monitored patients," said Daniel Pawlik, vice president of sales and marketing for CardioComm.
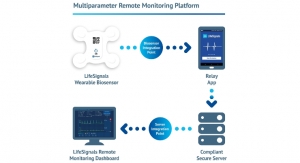
Guava II uses a modular (plug-in) based architecture allowing new features such as automated ECG analysis and interpretation algorithms, and ECG de-noising solutions to be added. GUAVA II is to be used to acquire, store, retrieve, analyze and produce measurements as well as textual interpretations, on ECG recordings and ECG data from a variety of single and multi-lead ECG devices.
Guava II is a completely rewritten, upgraded replacement for CardioComm Solutions’ prior generation software, Guava I, that offers a number of critical enhancements designed to streamline ECG interpretations. By re-writing Guava, CardioComm Solutions converted it to become a multiple bioscience/biometric monitoring device with interpretation capabilities, with the goal of building one software platform that can measure multiple biosigns from multiple device manufacturers, which CardioComm Solutions believes will be of significant interest in disease prevention management.
"Guava II is a completely rewritten and re-architected replacement for Guava I. It contains a re-implemented and expanded core, viewer, and plug-in infrastructure which enable us to add support for new devices, algorithms and additional biological signal measures. Importantly, the layered architecture enables us to develop native support and interfaces with mobile platforms such as iOS and Android, and compatibility with browsers such as IE, Chrome, Safari and Fire Fox, in the future," Chief Technology Officer Wade Barnes said.
As an example, currently, call centers may have two to three types of ECG monitoring solutions just for ECGs. The goal for CardioComm Solutions is to accommodate all monitoring, ultimately, having other software solutions that would look at correlating glucose or asthma readings, etc., where it will only be necessary to operate one program instead of multiple programs so hardware manufacturers can customize it to pair with CardioComm Solutions’ software. To this end, Guava II contains a re-implemented and expanded core, viewer, and plug-in infrastructure which enables the company to add support for new devices, algorithms, and additional biological signal measures. In line with improving its interoperability, the new architecture could interface with iOS and Android mobile platforms or various browsers like Internet Explorer, Chrome, Safari, or FireFox in the future.
"In our Gems and GC12 host applications, Guava I is an integrated ECG viewer technology. Utilizing Guava II's application programming interface, third party organizations and software developers may now license Guava II to integrate its capabilities within their own products. Adding this FDA-cleared technology for the viewing, measure and physician sign-off of acquired ECGs, a critical function of any ECG service, would provide a market differentiator to any commercial ECG service and medical call centre globally," CardioComm CEO Etienne Grima said.
Headquartered in Toronto, Ontario, Canada, with offices in Victoria, British Columbia, CardioComm
"Guava II offers a new level of regulatory credibility to ECG reading services, opening new opportunities to physicians and hospitals that wish to have ECG reviews completed that are adherent with all possible medical clearances. Physicians reading ECGs for, and hospitals receiving ECG reports generated from, ECG service providers may not be aware that an ECG system is being relied upon that may hold no current FDA clearance. Guava II will allow such service providers the option to customize their ECG management systems with an FDA compliant solution. Guava II will also offer significant data development and interoperability potential, will serve to streamline work flow, reduce staffing costs and improve the efficiency of identifying potentially life threatening arrhythmias in monitored patients," said Daniel Pawlik, vice president of sales and marketing for CardioComm.
Guava II uses a modular (plug-in) based architecture allowing new features such as automated ECG analysis and interpretation algorithms, and ECG de-noising solutions to be added. GUAVA II is to be used to acquire, store, retrieve, analyze and produce measurements as well as textual interpretations, on ECG recordings and ECG data from a variety of single and multi-lead ECG devices.
Guava II is a completely rewritten, upgraded replacement for CardioComm Solutions’ prior generation software, Guava I, that offers a number of critical enhancements designed to streamline ECG interpretations. By re-writing Guava, CardioComm Solutions converted it to become a multiple bioscience/biometric monitoring device with interpretation capabilities, with the goal of building one software platform that can measure multiple biosigns from multiple device manufacturers, which CardioComm Solutions believes will be of significant interest in disease prevention management.
"Guava II is a completely rewritten and re-architected replacement for Guava I. It contains a re-implemented and expanded core, viewer, and plug-in infrastructure which enable us to add support for new devices, algorithms and additional biological signal measures. Importantly, the layered architecture enables us to develop native support and interfaces with mobile platforms such as iOS and Android, and compatibility with browsers such as IE, Chrome, Safari and Fire Fox, in the future," Chief Technology Officer Wade Barnes said.
As an example, currently, call centers may have two to three types of ECG monitoring solutions just for ECGs. The goal for CardioComm Solutions is to accommodate all monitoring, ultimately, having other software solutions that would look at correlating glucose or asthma readings, etc., where it will only be necessary to operate one program instead of multiple programs so hardware manufacturers can customize it to pair with CardioComm Solutions’ software. To this end, Guava II contains a re-implemented and expanded core, viewer, and plug-in infrastructure which enables the company to add support for new devices, algorithms, and additional biological signal measures. In line with improving its interoperability, the new architecture could interface with iOS and Android mobile platforms or various browsers like Internet Explorer, Chrome, Safari, or FireFox in the future.
"In our Gems and GC12 host applications, Guava I is an integrated ECG viewer technology. Utilizing Guava II's application programming interface, third party organizations and software developers may now license Guava II to integrate its capabilities within their own products. Adding this FDA-cleared technology for the viewing, measure and physician sign-off of acquired ECGs, a critical function of any ECG service, would provide a market differentiator to any commercial ECG service and medical call centre globally," CardioComm CEO Etienne Grima said.
Features of Guava II include:
- The most recent ECG viewer software technology to enter the market;
- Analyzes and interprets ECG recordings and data from a variety of single and multi-lead ECG devices;
- Utilizes a modular (plug-in) based architecture allowing new features such as automated ECG analysis and interpretation algorithms, and ECG de-noising solutions to be added;
- Developed under strict ISO standards and international regulations;
- Offers a new level of regulatory credibility to ECG reading services, as many such services currently are used by IDTFs without FDA compliance since they are considered designed for internal use by the IDTF only;
- Opens new opportunities to physicians and hospitals that wish to have ECG reviews completed that are adherent with all possible medical clearances;
- Allows ECG service providers the option to customize their ECG management systems with an FDA-compliant solution;
- Offers significant data development and interoperability potential;
- Streamlines work flow;
- Reduces staffing costs;
- Improves the efficiency of identifying potentially life threatening arrhythmias in monitored patients; and
-
Utilizing Guava II's application programming interface (API), third party organizations and software developers may license GUAVA II to integrate its capabilities within their own products.
Headquartered in Toronto, Ontario, Canada, with offices in Victoria, British Columbia, CardioComm