02.26.14
Cyberonics Inc. received CE mark in Europe for the AspireSR generator, the company's sixth-generation vagus nerve stimulation (VNS) Therapy generator.
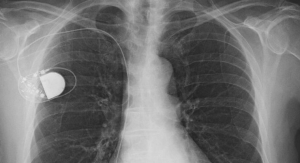
VNS Therapy uses an implanted medical device that delivers pulsed electrical signals to the vagus nerve to help treat refractory epilepsy and treatment-resistant depression.
The AspireSR generator combines existing VNS Therapy with a new feature: automatic stimulation in response to detection of a seizure. Proprietary technology enables the AspireSR generator to analyze relative heart rate changes to detect and respond to seizures. The technology is based on a growing body of evidence that seizures often are accompanied by an increase in heart rate (ictal tachycardia). With the automatic stimulation feature, the AspireSR generator better aligns stimulation with the clinical onset of a seizure, according to the company.
Currently, patients using VNS Therapy can use a hand-held magnet to activate stimulation manually when they anticipate the onset of a seizure. The company claims that on-demand stimulation has been shown to stop or shorten a seizure, reduce seizure severity, and improve or shorten the postictal (post-seizure) recovery period. The automatic stimulation feature of the AspireSR generator may benefit patients who experience seizures accompanied by ictal tachycardia and are unable to perform on-demand stimulation with the hand-held magnet, do not have a magnet available when needed, or experience a seizure while sleeping.
Results of the E-36 clinical study, which evaluated the performance and safety of the AspireSR generator, were presented at the American Epilepsy Society meeting in December. The study met its primary performance endpoint, and the safety profile for the AspireSR generator is consistent with currently available VNS Therapy systems.
"A significant number of people living with refractory epilepsy experience heart rate changes during their seizures," said Prof. Christian E. Elger, M.D., head of the Department of Epileptology at the University Hospital in Bonn, Germany. "AspireSR builds upon the VNS Therapy platform by providing a unique, innovative possible therapeutic option for these patients by automatically detecting and responding to seizures upon heart rate increase."
"European approval of the AspireSR generator represents an important milestone in Cyberonics' ongoing commitment to provide technologically advanced, device-based solutions for people with epilepsy," said Dan Moore, president and CEO of Houston, Texas-based Cyberonics. "This new generator will be particularly helpful to VNS Therapy patients who are unable to use magnet-activated stimulation consistently. We believe the Automatic Stimulation feature will continue to advance VNS Therapy as a foundational therapy for people with refractory epilepsy."
VNS Therapy is U.S. Food and drug Administration approved, though the AspireSR generator has not yet been approved by the agency.
VNS Therapy uses an implanted medical device that delivers pulsed electrical signals to the vagus nerve to help treat refractory epilepsy and treatment-resistant depression.
The AspireSR generator combines existing VNS Therapy with a new feature: automatic stimulation in response to detection of a seizure. Proprietary technology enables the AspireSR generator to analyze relative heart rate changes to detect and respond to seizures. The technology is based on a growing body of evidence that seizures often are accompanied by an increase in heart rate (ictal tachycardia). With the automatic stimulation feature, the AspireSR generator better aligns stimulation with the clinical onset of a seizure, according to the company.
Currently, patients using VNS Therapy can use a hand-held magnet to activate stimulation manually when they anticipate the onset of a seizure. The company claims that on-demand stimulation has been shown to stop or shorten a seizure, reduce seizure severity, and improve or shorten the postictal (post-seizure) recovery period. The automatic stimulation feature of the AspireSR generator may benefit patients who experience seizures accompanied by ictal tachycardia and are unable to perform on-demand stimulation with the hand-held magnet, do not have a magnet available when needed, or experience a seizure while sleeping.
Results of the E-36 clinical study, which evaluated the performance and safety of the AspireSR generator, were presented at the American Epilepsy Society meeting in December. The study met its primary performance endpoint, and the safety profile for the AspireSR generator is consistent with currently available VNS Therapy systems.
"A significant number of people living with refractory epilepsy experience heart rate changes during their seizures," said Prof. Christian E. Elger, M.D., head of the Department of Epileptology at the University Hospital in Bonn, Germany. "AspireSR builds upon the VNS Therapy platform by providing a unique, innovative possible therapeutic option for these patients by automatically detecting and responding to seizures upon heart rate increase."
"European approval of the AspireSR generator represents an important milestone in Cyberonics' ongoing commitment to provide technologically advanced, device-based solutions for people with epilepsy," said Dan Moore, president and CEO of Houston, Texas-based Cyberonics. "This new generator will be particularly helpful to VNS Therapy patients who are unable to use magnet-activated stimulation consistently. We believe the Automatic Stimulation feature will continue to advance VNS Therapy as a foundational therapy for people with refractory epilepsy."
VNS Therapy is U.S. Food and drug Administration approved, though the AspireSR generator has not yet been approved by the agency.