02.13.14
Miami, Fla.-base Opko Health Inc. has completed the calibration confirmation phase of its 4Kscore test clinical validation study currently underway at 21 urology clinics in the United States.
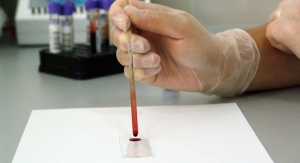
The 4Kscore is a blood test intended to measure the serum levels of four different prostate-derived kallikrein proteins: total PSA, free PSA, intact PSA and hK2. Levels of these biomarkers are combined with a patient's age and digital rectal exam status using a proprietary algorithm to calculate the probability of a finding of aggressive prostate cancer. The 4Kscore test is based on over a decade of research of the four kallikrein panel of biomarkers conducted by scientists at Memorial Sloan Kettering Cancer Center and leading research centers in Europe on over 10,000 patients. The information provided by the 4Kscore test can help facilitate the shared decision-making process between urologist and patient in determining the advisability of a prostate biopsy.
In 2013, more 238,000 new cases of prostate cancer will be identified and 29,720 men will die from the disease, according to estimates released by the National Cancer Institute, making it the second most deadly cancer in U.S. men. Prostate cancer is usually first detected by elevations in serum PSA. However, PSA level is often high for reasons unrelated to prostate cancer and, since an elevated PSA level often leads to biopsy, 75 percent of all prostate biopsies performed are either negative or indicate a low likelihood of aggressive cancer.
The Opko 4Kscore test clinical validation protocol involves two parts: a calibration confirmation phase and a validation phase. The calibration confirmation phase evaluated the applicability of the 4Kscore algorithm, developed using banked samples from European studies, to the Opko study samples collected in the United States and shipped directly to Opko Lab in Nashville, Tenn. for testing. The results, according to the company, confirmed the validity of the algorithm. The study is now more than 50 percent complete toward the final goal of 1,200 subjects. These results support the U.S. launch of the 4Kscore test scheduled for March.
“We are extremely pleased that results of the calibration phase confirmed the 4Kscore algorithm is an accurate predictor of prostate cancer, and especially aggressive disease,” said David Okrongly, president of Opko Diagnostics. “The reproducibility of the algorithm speaks to the robustness of the biomarkers used in the 4Kscore algorithm, as well as the quality of the multiple clinical studies published in peer-reviewed journals.”
“These results reinforce our belief that the 4Kscore test will be an important addition to the urologists’ means of assessing a patient’s risk for aggressive prostate cancer as well as an important new diagnostic product for Opko,” said Phillip Frost, M.D., chairman and CEO of Opko.
Just in time for the new release, Opko appointed Greg Stanley as the new vice president of sales and marketing for its global diagnostics business unit. According to Okongly, he has experience “launching and gaining reimbursement for novel cancer diagnostic test … and a broad background in IVD sales and marketing.”
The 4Kscore is a blood test intended to measure the serum levels of four different prostate-derived kallikrein proteins: total PSA, free PSA, intact PSA and hK2. Levels of these biomarkers are combined with a patient's age and digital rectal exam status using a proprietary algorithm to calculate the probability of a finding of aggressive prostate cancer. The 4Kscore test is based on over a decade of research of the four kallikrein panel of biomarkers conducted by scientists at Memorial Sloan Kettering Cancer Center and leading research centers in Europe on over 10,000 patients. The information provided by the 4Kscore test can help facilitate the shared decision-making process between urologist and patient in determining the advisability of a prostate biopsy.
In 2013, more 238,000 new cases of prostate cancer will be identified and 29,720 men will die from the disease, according to estimates released by the National Cancer Institute, making it the second most deadly cancer in U.S. men. Prostate cancer is usually first detected by elevations in serum PSA. However, PSA level is often high for reasons unrelated to prostate cancer and, since an elevated PSA level often leads to biopsy, 75 percent of all prostate biopsies performed are either negative or indicate a low likelihood of aggressive cancer.
The Opko 4Kscore test clinical validation protocol involves two parts: a calibration confirmation phase and a validation phase. The calibration confirmation phase evaluated the applicability of the 4Kscore algorithm, developed using banked samples from European studies, to the Opko study samples collected in the United States and shipped directly to Opko Lab in Nashville, Tenn. for testing. The results, according to the company, confirmed the validity of the algorithm. The study is now more than 50 percent complete toward the final goal of 1,200 subjects. These results support the U.S. launch of the 4Kscore test scheduled for March.
“We are extremely pleased that results of the calibration phase confirmed the 4Kscore algorithm is an accurate predictor of prostate cancer, and especially aggressive disease,” said David Okrongly, president of Opko Diagnostics. “The reproducibility of the algorithm speaks to the robustness of the biomarkers used in the 4Kscore algorithm, as well as the quality of the multiple clinical studies published in peer-reviewed journals.”
“These results reinforce our belief that the 4Kscore test will be an important addition to the urologists’ means of assessing a patient’s risk for aggressive prostate cancer as well as an important new diagnostic product for Opko,” said Phillip Frost, M.D., chairman and CEO of Opko.
Just in time for the new release, Opko appointed Greg Stanley as the new vice president of sales and marketing for its global diagnostics business unit. According to Okongly, he has experience “launching and gaining reimbursement for novel cancer diagnostic test … and a broad background in IVD sales and marketing.”