01.27.14
W.L. Gore & Associates has received Shonin approval from the Japanese Ministry of Health, Labor and Welfare to market the Gore Excluder AAA Endoprosthesis featuring C3 Delivery System as a minimally invasive treatment for patients suffering from an abdominal aortic aneurysm (AAA).
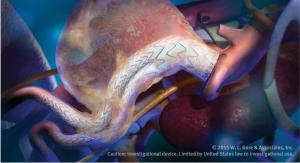
The Gore C3 Delivery System enables doctors and interventionalists to reposition the Gore Excluder Device prior to final release from the delivery catheter. The added deployment control helps physicians better treat challenging anatomies and gives them cannulation options with the ability to bring the contralateral gate to the contralateral guidewire.
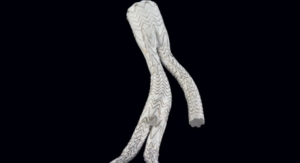
The Gore Excluder Device remains virtually unchanged—with the same low delivery profile and flexibility on catheter that facilitates access and passage through narrow and tortuous anatomies. Once delivered into the aorta, the Gore C3 Delivery System intuitively enables repositioning of the stent-graft. The ability to reposition the device may minimize complications that could occur if the stent-graft needs to be repositioned after the initial deployment. This technology, approved by the U.S. Food and Drug Administration in January 2011, is designed to give physicians two additional opportunities to accurately place the stent-graft relative to the patient’s anatomy.
“The GORE C3 Delivery System offers more opportunities to optimize infrarenal seal and provides the physician with greater control. The ability to reposition minimizes complications that can occur with graft placement, adding a level of confidence and control during endovascular procedures that physicians have not experienced before,” said Alan Lumsden, M.D., chairman, Department of Cardiovascular Surgery at the Methodist Hospital in Houston, Texas.
“Since launching the GORE C3 Delivery System for the Gore Excluder Device in 2011, it has shown a high level of performance, allowing physicians to achieve the best possible deployment of the stent-graft,” said Ryan Takeuchi, Gore Aortic Business Leader.
The Gore Medical Products Division has provided therapeutic solutions to complex medical problems for more than 35 years. During that time, more than 35 million innovative Gore medical devices have been implanted, according to the company. The Gore Medical family of products includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.
W.L. Gore & Associates is headquartered in Flagstaff, Ariz.
The Gore C3 Delivery System enables doctors and interventionalists to reposition the Gore Excluder Device prior to final release from the delivery catheter. The added deployment control helps physicians better treat challenging anatomies and gives them cannulation options with the ability to bring the contralateral gate to the contralateral guidewire.
The Gore Excluder Device remains virtually unchanged—with the same low delivery profile and flexibility on catheter that facilitates access and passage through narrow and tortuous anatomies. Once delivered into the aorta, the Gore C3 Delivery System intuitively enables repositioning of the stent-graft. The ability to reposition the device may minimize complications that could occur if the stent-graft needs to be repositioned after the initial deployment. This technology, approved by the U.S. Food and Drug Administration in January 2011, is designed to give physicians two additional opportunities to accurately place the stent-graft relative to the patient’s anatomy.
“The GORE C3 Delivery System offers more opportunities to optimize infrarenal seal and provides the physician with greater control. The ability to reposition minimizes complications that can occur with graft placement, adding a level of confidence and control during endovascular procedures that physicians have not experienced before,” said Alan Lumsden, M.D., chairman, Department of Cardiovascular Surgery at the Methodist Hospital in Houston, Texas.
“Since launching the GORE C3 Delivery System for the Gore Excluder Device in 2011, it has shown a high level of performance, allowing physicians to achieve the best possible deployment of the stent-graft,” said Ryan Takeuchi, Gore Aortic Business Leader.
The Gore Medical Products Division has provided therapeutic solutions to complex medical problems for more than 35 years. During that time, more than 35 million innovative Gore medical devices have been implanted, according to the company. The Gore Medical family of products includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.
W.L. Gore & Associates is headquartered in Flagstaff, Ariz.