A new ASTM International standard will provide what the organication is calling "a consistent method for analyzing hip stems used in hip replacement surgery."
The new standard, ASTM F2996, "Practice for Finite Element Analysis (FEA) of Nonmodular Metallic Orthopaedic Hip Femoral Stems," was developed by a subcommittee on arthroplasty, part of ASTM International Committee F04 on medical and surgical materials and devices.
“ASTM F2996 was developed to harmonize the method by which finite element models are performed, as comments from regulatory agencies indicated many different methods were used by industry,” says Jeffrey Sprague, project manager, Smith & Nephew, and an F04 member. Sprague and Walter Schmidt, senior manager, modeling and simulation, Stryker Orthopaedics, co-chaired the task group that developed F2996. The new standard will allow for accurate comparisons to be made among different companies and researchers.
ASTM F2996 establishes requirements and considerations for the development of finite element models used in the evaluation of non-modular metallic orthopedic hip stem designs. Procedures for performing model checks and verification are provided to determine if the analysis follows recommended guidelines. Testing with such models can predict static implant stresses and strains. Results from testing to F2996 will establish worst-case size for a particular implant.
Sprague noted that the subcommittee also currently is working on a proposed new standard that will provide a finite element guide for knee femoral components. Companies and laboratories that design and test such components are encouraged to participate in this standards developing activity.
For more info, visit www.astm.org.
Known until 2001 as the American Society for Testing and Materials, ASTM is an international standards organization that develops and publishes voluntary consensus technical standards for a wide range of materials, products, systems and services. The organization's headquarters is in West Conshohocken, Pa.
Breaking News
ASTM Approves New Hip Implant Standard
ASTM also working on guidance for knee femoral components.
09.06.13
Related Knowledge Center
Related Literature / Brochures
-
Contract Manufacturing | R&D & Design

The Secret of SmartForm
Creganna Medical is the only outsourcing partner with both FDA registration and balloon capability across three continents.
-
Surgical | Testing
ASTM Guide Supports Cleaning Protocols for Reusable Medical Devices
The standard also provides procedures for selecting the appropriate test soil for different kinds of devices.ASTM International 03.21.17
-

New President of ASTM International Announced
Katharine Morgan succeeds James A. Thomas, who was in role for 25 years.ASTM International 02.01.17
-
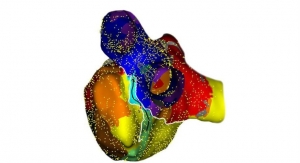
Abbott Launches Precision Cardiac Mapping System
EnSite system allows better three-dimensional images of arrhythmias in the heart.PR Newswire 01.13.17
-
Cardiovascular | R&D & Design
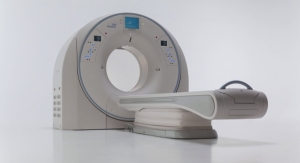
Toshiba Medical Transforms CT Imaging With the Aquilion ONE/GENESIS Edition
Flagship CT system introduces a new standard in area detector CT.Toshiba Medical 11.30.16
















