08.14.13
Sunnyvale, Calif.-based medical device company Covidien plc is expanding its radiofrequency ablation (RFA) catheters. The company is launching the Barrx channel RFA endoscopic catheter for treating Barrett’s esophagus and certain gastrointestinal bleeding disorders. Left untreated, Barrett’s esophagus, a precancerous condition of the esophageal lining, can lead to life-threatening cancer of the esophagus.
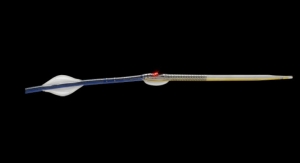
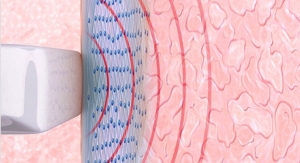
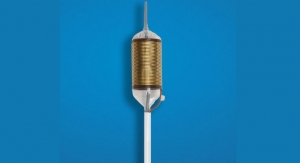
The Barrx line of RFA catheters is based on Covidien’s proprietary technology, designed to control the amount of RF energy delivered to remove diseased tissue, thereby allowing the growth of healthy tissue. Covidien claims that endoscopic surgeons and gastroenterologists can shorten procedure time with the new channel RFA device, as this patient-centric design allows for fewer endoscope introductions and removals compared to other Barrx catheters. The channel catheter design reportedly enables a 7.5 mm-wide electrode to pass through a 2.8-mm diameter endoscope working channel. It also has a transparent ablation electrode for enhanced visualization and a rotatable shaft design that provides additional control and maneuverability.
“The newest addition to the Barrx portfolio is designed to simplify the procedure by delivering our proven RF technology through the working channel of the endoscope,” said Vafa Jamali, vice president and general manager of Covidien’s gastrointestinal solutions business.
The Barrx channel RFA endoscopic catheter is available in the United States and Europe.
The Barrx line of RFA catheters is based on Covidien’s proprietary technology, designed to control the amount of RF energy delivered to remove diseased tissue, thereby allowing the growth of healthy tissue. Covidien claims that endoscopic surgeons and gastroenterologists can shorten procedure time with the new channel RFA device, as this patient-centric design allows for fewer endoscope introductions and removals compared to other Barrx catheters. The channel catheter design reportedly enables a 7.5 mm-wide electrode to pass through a 2.8-mm diameter endoscope working channel. It also has a transparent ablation electrode for enhanced visualization and a rotatable shaft design that provides additional control and maneuverability.
“The newest addition to the Barrx portfolio is designed to simplify the procedure by delivering our proven RF technology through the working channel of the endoscope,” said Vafa Jamali, vice president and general manager of Covidien’s gastrointestinal solutions business.
The Barrx channel RFA endoscopic catheter is available in the United States and Europe.