The organizations are conducting a workshop at the FDA headquarters in Silver Spring, Md., on October 11, which will bring together clinicians, academicians, federal employees and industry experts to discuss challenges to intraocular lens (IOL) innovation with a focus on endpoint methodologies used in evaluating IOL safety and effectiveness.
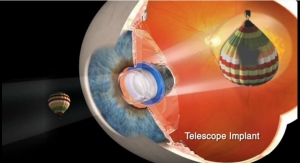
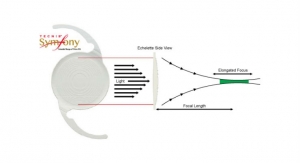
Caption: Intraocular lenses, or IOLs (pictured left), are used in cataract surgery, the most common elective surgery in the U.S., and implanted in more than three million American cataract surgery patients a year.
"If a wider variety of premium IOLs were made available in the U.S., this could help improve quality of life for cataract patients," said Paul Sternberg Jr., M.D., president of the American Academy of Ophthalmology. "The academy looks forward to working with the FDA, along with physician and medical device industry thought leaders, to ensure a more timely delivery of these devices to the American people."
The workshop will be opened by Sternberg; Jeffrey E. Shuren, M.D., J.D., director of the FDA’s Center for Devices and Radiological Health; and Malvina Eydelman, M.D., director of the FDA Division of Ophthalmic and Ear, Nose and Throat Devices. It will be moderated by Academy Trustee-at-Large Thomas Oetting, M.D., professor of clinical ophthalmology at the University of Iowa College of Medicine, and academy member Malik Y. Kahook, M.D., professor of ophthalmology and the Slater Family Endowed Chair in ophthalmology at the University of Colorado School of Medicine.
The event will take place from 8:30 a.m. to 5:30 p.m. at the FDA’s White Oak campus. There is a cost to attend. To register and learn more, visit www.aao.org/IOLworkshop.