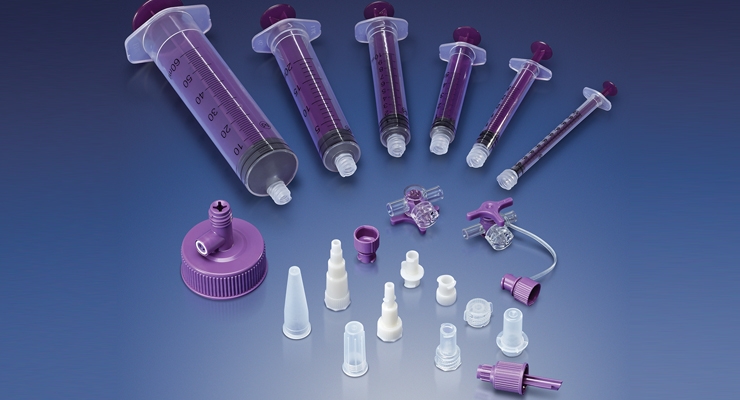
Ronkonkoma, NY – May 25, 2016 – Qosina is pleased to announce the addition of 40 enteral feeding connectors that meet the ISO 80369-3 standard and will not mate with the ISO 594-1 and ISO 594-2 luers. Qosina is offering ENFit™ male and female connectors, caps, stepped adapters, Y connectors, stopcocks and syringes, as well as the ENFit to ENLock adapters, ENPlus cross spikes and universal bottle adapters for the ENPlus cross spike. All of these parts have been added in various sizes and materials. Qosina will continue to add more ENFit connectors to meet the needs of your enteral project. Please visit our ENFit section at www.qosina/ENFit. Qosina can modify any existing component to meet your needs or build new tooling to meet your exact requirements.
Enteral is the first clinical application to make the transition to new, safer connectors. The New Enteral Feeding Connectors have been designed to improve patient safety and reduce the risk of enteral feeding misconnections. The new standard has changed the configuration of the male and female connectors. These enteral connectors are larger and will not allow connectivity with the male luer or female luer connectors that meet the ISO 594 standards or any other connectors for any other clinical use. Administration sets and syringes have a female connector end that fit into a male patient-access feeding tube port.
The Global Enteral Device Suppliers Association (GEDSA.org) was formed to facilitate the transition to the new type of connectors and has created the brand ENFit to represent the new ISO 80369-3 standard. Qosina is actively researching the upcoming changes and is a member of GEDSA. Qosina will be adding compliant components to its extensive line as the standards become approved. To learn more about the ISO 80369 standard, please visit www.qosina.com/iso-80369.
Qosina is a worldwide supplier of thousands of stock OEM disposable components to the medical and pharmaceutical industries including bag ports, connectors, luers, clamps, adapters, clips, valves, needle hubs, sanitary flanges, stopcocks, hemostasis valves, tuohy borst adapters, swabs, spikes and syringes. Qosina operates an ISO 9001 and ISO 14001 registered facility.
Qosina offers low minimums and short lead times. Call our Customer Service Specialists to request a sample, obtain our latest catalog or place an order via phone +1(631) 242-3000, fax +1(631) 242-3230 or email info@qosina.com. Visit us at www.qosina.com to see our latest products. Custom sourcing services are available through our knowledge specialists using Qosina’s extensive resources.
Contact:
Qosina
Maria Stazzone
2002-Q Orville Drive North
Ronkonkoma, NY 11779
Phone +1 (631) 242-3000
Fax +1 (631) 242-3230
Email: mstazzone@qosina.com
Enteral is the first clinical application to make the transition to new, safer connectors. The New Enteral Feeding Connectors have been designed to improve patient safety and reduce the risk of enteral feeding misconnections. The new standard has changed the configuration of the male and female connectors. These enteral connectors are larger and will not allow connectivity with the male luer or female luer connectors that meet the ISO 594 standards or any other connectors for any other clinical use. Administration sets and syringes have a female connector end that fit into a male patient-access feeding tube port.
The Global Enteral Device Suppliers Association (GEDSA.org) was formed to facilitate the transition to the new type of connectors and has created the brand ENFit to represent the new ISO 80369-3 standard. Qosina is actively researching the upcoming changes and is a member of GEDSA. Qosina will be adding compliant components to its extensive line as the standards become approved. To learn more about the ISO 80369 standard, please visit www.qosina.com/iso-80369.
Qosina is a worldwide supplier of thousands of stock OEM disposable components to the medical and pharmaceutical industries including bag ports, connectors, luers, clamps, adapters, clips, valves, needle hubs, sanitary flanges, stopcocks, hemostasis valves, tuohy borst adapters, swabs, spikes and syringes. Qosina operates an ISO 9001 and ISO 14001 registered facility.
Qosina offers low minimums and short lead times. Call our Customer Service Specialists to request a sample, obtain our latest catalog or place an order via phone +1(631) 242-3000, fax +1(631) 242-3230 or email info@qosina.com. Visit us at www.qosina.com to see our latest products. Custom sourcing services are available through our knowledge specialists using Qosina’s extensive resources.
Contact:
Qosina
Maria Stazzone
2002-Q Orville Drive North
Ronkonkoma, NY 11779
Phone +1 (631) 242-3000
Fax +1 (631) 242-3230
Email: mstazzone@qosina.com