Globe Newswire02.01.22
Seno Medical Instruments Inc.'s CONFIDENCE registry study has begun.
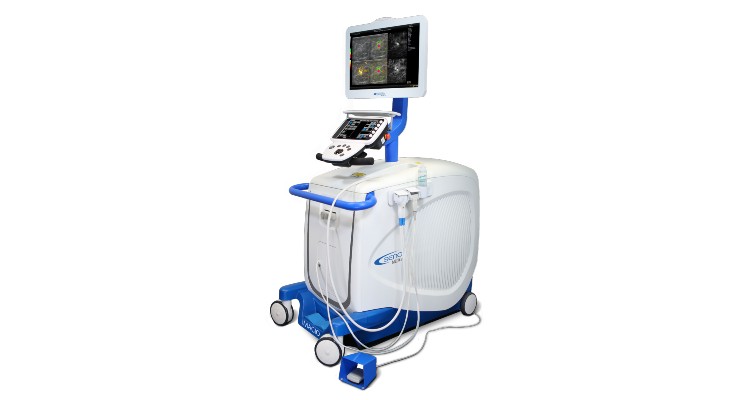
Seno has launched the trial to evaluate the efficacy of imaging with opto-acoustic/ultrasound (OA/US) to diagnose breast abnormalities including cancer using its groundbreaking Imagio Breast Imaging System. Abouty 12 sites will participate in the international registry, with a target of over 600 total patients participating through the study’s timeframe, ending in 2024. “This registry study will help us substantiate the efficacy of the Imagio System in a real-world setting and the ability for OA/US to benefit physicians and patients around the world,” said Seno CEO Tom Umbel.
Several international sites in the Netherlands are recruiting for the study. The trial will obtain real-life, post-market data on the use of the Imagio System and to provide evidence on the general use and performance of the system in a real-world setting.
CONFIDENCE registry endpoints to be analyzed include:
The company’s OA/US technology combines laser optics and grayscale ultrasound to provide fused functional and anatomical breast imaging. The opto-acoustic images provide a unique blood map in and around breast masses, while the ultrasound provides a traditional anatomical image. Through the appearance or absence of two hallmark indicators of cancer—angiogenesis and deoxygenation—Seno Medical has shown that the Imagio OA/US Breast Imaging System will be a more effective tool to help radiologists confirm or rule out malignancy compared to traditional diagnostic imaging modalities—without exposing patients to potentially harmful ionizing radiation (X-rays) or contrast agents. In addition to the imaging provided by the Imagio System, Seno includes an artificial intelligence (AI) decision-support tool (the SenoGram) to aid physicians in interpreting the new images that, along with training and certification, helps radiologists make the transition from ultrasound alone to OA/US imaging.
“We believe, much like other hybrid imaging modalities such as PET/CT, the sum of opto-acoustics and ultrasound together provide greater sensitivity and specificity than either of the imaging modalities individually,” said Shaan Schaeffer, vice president of Clinical Operations at Seno.
The system is indicated for use by trained and qualified healthcare providers to evaluate palpable and non-palpable breast abnormalities in adult patients who are referred for diagnostic imaging breast work-up following clinical presentation or other imaging examinations such as screening mammography.
Seno Medical Instruments Inc. is a San Antonio, Texas-based medical imaging company developing and commercializing a new modality in cancer diagnosis: opto-acoustic imaging. Approved by the U.S. Food and Drug Administration in January 2021, Seno Medical’s Imagio Breast Imaging System fuses opto-acoustic technology with ultrasound (OA/US) to generate real-time functional and anatomical images of the breast.
Reference
i Vlahiotis A, Griffin B, Stavros AT, Margolis J. Analysis of utilization patterns and associated costs of the breast imaging and diagnostic procedures after screening mammography. Clinicoeconomics Outcomes Res 2018;10:157-167.
Seno has launched the trial to evaluate the efficacy of imaging with opto-acoustic/ultrasound (OA/US) to diagnose breast abnormalities including cancer using its groundbreaking Imagio Breast Imaging System. Abouty 12 sites will participate in the international registry, with a target of over 600 total patients participating through the study’s timeframe, ending in 2024. “This registry study will help us substantiate the efficacy of the Imagio System in a real-world setting and the ability for OA/US to benefit physicians and patients around the world,” said Seno CEO Tom Umbel.
Several international sites in the Netherlands are recruiting for the study. The trial will obtain real-life, post-market data on the use of the Imagio System and to provide evidence on the general use and performance of the system in a real-world setting.
CONFIDENCE registry endpoints to be analyzed include:
- Specificity, negative-likelihood ratio, sensitivity, and positive-likelihood ratio for the Imagio System versus other standard-of-care imaging modalities,
- Use of the Imagio System in comparison to, and in combination with, other imaging modalities in impacting the decision to biopsy,
- Quality of Life Assessment - Testing Morbidity Index,
- In malignant masses, assessment of the roles of OA/US as a prognostic biomarker,
- Characteristics of lymph node lesions/masses the device is used for.
The company’s OA/US technology combines laser optics and grayscale ultrasound to provide fused functional and anatomical breast imaging. The opto-acoustic images provide a unique blood map in and around breast masses, while the ultrasound provides a traditional anatomical image. Through the appearance or absence of two hallmark indicators of cancer—angiogenesis and deoxygenation—Seno Medical has shown that the Imagio OA/US Breast Imaging System will be a more effective tool to help radiologists confirm or rule out malignancy compared to traditional diagnostic imaging modalities—without exposing patients to potentially harmful ionizing radiation (X-rays) or contrast agents. In addition to the imaging provided by the Imagio System, Seno includes an artificial intelligence (AI) decision-support tool (the SenoGram) to aid physicians in interpreting the new images that, along with training and certification, helps radiologists make the transition from ultrasound alone to OA/US imaging.
“We believe, much like other hybrid imaging modalities such as PET/CT, the sum of opto-acoustics and ultrasound together provide greater sensitivity and specificity than either of the imaging modalities individually,” said Shaan Schaeffer, vice president of Clinical Operations at Seno.
The system is indicated for use by trained and qualified healthcare providers to evaluate palpable and non-palpable breast abnormalities in adult patients who are referred for diagnostic imaging breast work-up following clinical presentation or other imaging examinations such as screening mammography.
Seno Medical Instruments Inc. is a San Antonio, Texas-based medical imaging company developing and commercializing a new modality in cancer diagnosis: opto-acoustic imaging. Approved by the U.S. Food and Drug Administration in January 2021, Seno Medical’s Imagio Breast Imaging System fuses opto-acoustic technology with ultrasound (OA/US) to generate real-time functional and anatomical images of the breast.
Reference
i Vlahiotis A, Griffin B, Stavros AT, Margolis J. Analysis of utilization patterns and associated costs of the breast imaging and diagnostic procedures after screening mammography. Clinicoeconomics Outcomes Res 2018;10:157-167.