02.06.15
St. Paul, Minn.-based EnteroMedics Inc. has earned U.S. Food and Drug Administration (FDA) approval for its Vbloc vagal blocking therapy delivered via the Maestro system. The device treats obesity in adults with a body mass index of at least 40 to 45 kilograms (weight) per square meter (height); or 35 to 39.9 kg/m2 in adults with related health condition such as high blood pressure or high cholesterol levels, and who have tried to lose weight in a supervised weight management program within the past five years.
The Maestro system is the first new medical device to be approved by the FDA for obesity in more than 10 years.
Approval of the Maestro rechargeable system was based on the Recharge study, a randomized, double-blind, sham-controlled trial to evaluate the safety and effectiveness of the Maestro rechargeable system in treating obesity. In an intention-to-treat analysis (ITT) analysis of the study results, Vbloc-treated patients achieved 24.4 percent excess weight loss (EWL) at 12 months. At 18 months, Vbloc-treated patients maintained a 23.5 percent EWL. In a responder analysis of the ITT population at 12 months, more than 50 percent of Vbloc-treated patients achieved 20 percent or greater EWL.
EnteroMedics anticipates that the device will be available, on a limited basis, at select Bariatric Centers of Excellence in the United States this year. The Maestro system has received CE Mark and is listed on the Australian Register of Therapeutic Goods.
“Vbloc Therapy offers an entirely new approach to the treatment of obesity,” said Scott Shikora, M.D., EnteroMedics’ chief consulting medical officer. “By blocking signals along the nerves that connect the brain and stomach, Vbloc reduces feelings of hunger and promotes earlier feelings of fullness, which can help people with obesity reduce the number of calories consumed and promote safe, healthy and durable weight loss.”
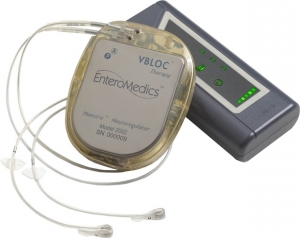
The Maestro system is a pacemaker-like device that is implanted to control both hunger and fullness by intermittently blocking the primary nerve which regulates the digestive system, the vagus nerve. Vbloc Therapy does not surgically alter or restrict the digestive system, does not create barriers to prevent absorption of nutrients and is completely reversible, allowing patients to lose weight without lifestyle compromises.
“There is no ‘one-size-fits-all’ approach to treating the disease of obesity. We believe strongly that expanded treatment options are essential to individuals affected by obesity,” said Joe Nadglowski, Obesity Action Coalition president and CEO.
“The Maestro system fills a gap in the currently available treatment options, offering clinically meaningful weight loss without the fear or many of the side effects associated with existing bariatric options,” said Mark B. Knudson, Ph.D., EnteroMedics’ president and CEO.
The Maestro system is the first new medical device to be approved by the FDA for obesity in more than 10 years.
Approval of the Maestro rechargeable system was based on the Recharge study, a randomized, double-blind, sham-controlled trial to evaluate the safety and effectiveness of the Maestro rechargeable system in treating obesity. In an intention-to-treat analysis (ITT) analysis of the study results, Vbloc-treated patients achieved 24.4 percent excess weight loss (EWL) at 12 months. At 18 months, Vbloc-treated patients maintained a 23.5 percent EWL. In a responder analysis of the ITT population at 12 months, more than 50 percent of Vbloc-treated patients achieved 20 percent or greater EWL.
EnteroMedics anticipates that the device will be available, on a limited basis, at select Bariatric Centers of Excellence in the United States this year. The Maestro system has received CE Mark and is listed on the Australian Register of Therapeutic Goods.
“Vbloc Therapy offers an entirely new approach to the treatment of obesity,” said Scott Shikora, M.D., EnteroMedics’ chief consulting medical officer. “By blocking signals along the nerves that connect the brain and stomach, Vbloc reduces feelings of hunger and promotes earlier feelings of fullness, which can help people with obesity reduce the number of calories consumed and promote safe, healthy and durable weight loss.”
The Maestro system is a pacemaker-like device that is implanted to control both hunger and fullness by intermittently blocking the primary nerve which regulates the digestive system, the vagus nerve. Vbloc Therapy does not surgically alter or restrict the digestive system, does not create barriers to prevent absorption of nutrients and is completely reversible, allowing patients to lose weight without lifestyle compromises.
“There is no ‘one-size-fits-all’ approach to treating the disease of obesity. We believe strongly that expanded treatment options are essential to individuals affected by obesity,” said Joe Nadglowski, Obesity Action Coalition president and CEO.
“The Maestro system fills a gap in the currently available treatment options, offering clinically meaningful weight loss without the fear or many of the side effects associated with existing bariatric options,” said Mark B. Knudson, Ph.D., EnteroMedics’ president and CEO.