06.01.11
A new graft product is available from W. L. Gore & Associates. The company recently rolled out its Hybrid Vascular Graft for the U.S. market.
According to the company, the device expands treatment options and improves blood flow for dialysis access patients with end-stage renal disease (ESRD). More than 130 dialysis access patients have been successfully treated with the Gore Hybrid Vascular Graft. Three-month follow-up data for newly created access implants demonstrates a trend toward a reduction in revision procedures, thrombosis events and seroma, as compared to historical graft data. In the United States, more than 300,000 people suffer from ESRD and are in need of dialysis.
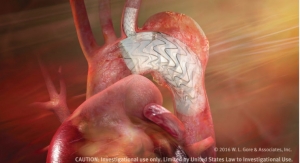
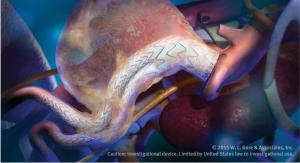
The graft is indicated for use as a vascular prosthesis for dialysis access or bypass for other peripheral vascular diseases. It is designed to address the most common causes of graft failure; intimal hyperplasia, thrombosis and seroma. The device simplifies access to vessels with a new over the wire deployment method through a smaller than usual incision in the skin that reduces vessel injury and dissection, company officials noted. The Hybrid Vascular Graft has been used to create new access sites in anatomical locations that would have been abandoned otherwise, preserving the amount of access sites available throughout the patient’s long-term therapy.
Since receiving FDA clearance in 2010, more than 130 patients were treated with the GORE Hybrid Device at multiple centers across the United States. John R. Ross, M.D., general surgeon at the Bamberg County Hospital, in Bamberg, S.C., performed the first clinical implants.
“Outflow stenosis has been a long-standing challenge for vascular grafts. The GORE Hybrid Vascular Graft is a promising new option because it targets intimal hyperplasia by creating a sutureless anastomosis that can be created percutaneously,” said Ross. “This new device directs the outflow so it is in-line with the vessel and shields the area most prone to intimal hyperplasia and failure.”
The expanded polytetrafluoroethylene (ePTFE) vascular prosthesis has a section reinforced with nitinol. The nitinol reinforced section is partially constrained to allow for easy insertion and deployment into vessels that are difficult to reach or in challenging anatomical locations. It is the only combination graft that incorporates Carmeda BioActive Surface end-point immobilization of heparin to the luminal surface, resulting in a proven thromboresistant surface, according to the company. .
According to the company, the device expands treatment options and improves blood flow for dialysis access patients with end-stage renal disease (ESRD). More than 130 dialysis access patients have been successfully treated with the Gore Hybrid Vascular Graft. Three-month follow-up data for newly created access implants demonstrates a trend toward a reduction in revision procedures, thrombosis events and seroma, as compared to historical graft data. In the United States, more than 300,000 people suffer from ESRD and are in need of dialysis.
The graft is indicated for use as a vascular prosthesis for dialysis access or bypass for other peripheral vascular diseases. It is designed to address the most common causes of graft failure; intimal hyperplasia, thrombosis and seroma. The device simplifies access to vessels with a new over the wire deployment method through a smaller than usual incision in the skin that reduces vessel injury and dissection, company officials noted. The Hybrid Vascular Graft has been used to create new access sites in anatomical locations that would have been abandoned otherwise, preserving the amount of access sites available throughout the patient’s long-term therapy.
Since receiving FDA clearance in 2010, more than 130 patients were treated with the GORE Hybrid Device at multiple centers across the United States. John R. Ross, M.D., general surgeon at the Bamberg County Hospital, in Bamberg, S.C., performed the first clinical implants.
“Outflow stenosis has been a long-standing challenge for vascular grafts. The GORE Hybrid Vascular Graft is a promising new option because it targets intimal hyperplasia by creating a sutureless anastomosis that can be created percutaneously,” said Ross. “This new device directs the outflow so it is in-line with the vessel and shields the area most prone to intimal hyperplasia and failure.”
The expanded polytetrafluoroethylene (ePTFE) vascular prosthesis has a section reinforced with nitinol. The nitinol reinforced section is partially constrained to allow for easy insertion and deployment into vessels that are difficult to reach or in challenging anatomical locations. It is the only combination graft that incorporates Carmeda BioActive Surface end-point immobilization of heparin to the luminal surface, resulting in a proven thromboresistant surface, according to the company. .