Michael Barbella, Managing Editor04.16.24
Sparrow BioAcoustics [Sparrow] has launched Stethophone, a software advancement that turns smartphones into medical-grade stethoscopes. Stethophone enables users to capture, analyze, and share critical heart health data with medical personnel from the comfort of any location.
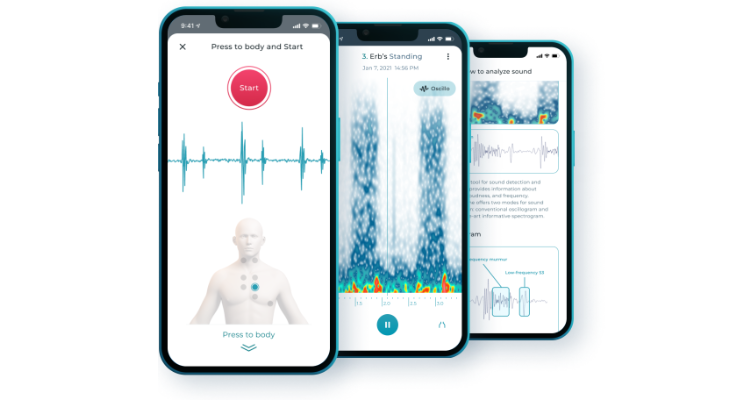
Stethophone works by harnessing a smartphone's built-in microphone to capture the heart's nuanced sounds. Through sophisticated bioacoustics engineering, these sounds are then filtered to provide clear, medical-grade diagnostic signals—some of them at the outermost edges of human auditory perception. With widespread adoption, it is expected this bioacoustics technology could enable clinicians to more easily and quickly detect and diagnose routine and difficult cardiac conditions.
Stethophone won U.S. Food and Drug Administration (FDA) clearance in 2023 for use by medical professionals in the United States, marking it as the first-ever FDA-cleared smartphone stethoscope Software-as-a-Medical-Device (SaMD). The software also gained FDA clearance last year for use by consumers—another first in the industry—empowering individuals to take an active role in their heart health.
In addition to providing the bioacoustics technology that powers Stethophone, Sparrow offers medical practitioners advanced visualizations that enable them to understand how to interpret diagnostics originating from the device. These visualizations aid in the recognition of patterns and anomalies that may not be easily discernible by conventional cardiac diagnostics, thereby potentially enhancing diagnostic accuracy and efficiency.
"We are at the forefront of a revolution in personalized heart health," Sparrow BioAcoustics CEO Mark Attila Opauzsky said. "Stethophone embodies our commitment to provide everyone the power, agency, and access to fight heart disease right in their own hands."
Sparrow is looking to the future with the development of Stethophone's next iteration, which explores integrating artificial intelligence functionalities into the bioacoustics engineering.
Stethophone is currently available on iOS.
Sparrow BioAcoustics, with offices in Newfoundland, Nova Scotia, and Toronto, Canada, is steering the Software as a Medical Device industry in new directions for cardiac and pulmonary disease detection. Its physicians, engineers and data scientists are working to unlock the richest source of diagnostic information about cardiac and pulmonary conditions.
Stethophone works by harnessing a smartphone's built-in microphone to capture the heart's nuanced sounds. Through sophisticated bioacoustics engineering, these sounds are then filtered to provide clear, medical-grade diagnostic signals—some of them at the outermost edges of human auditory perception. With widespread adoption, it is expected this bioacoustics technology could enable clinicians to more easily and quickly detect and diagnose routine and difficult cardiac conditions.
Stethophone won U.S. Food and Drug Administration (FDA) clearance in 2023 for use by medical professionals in the United States, marking it as the first-ever FDA-cleared smartphone stethoscope Software-as-a-Medical-Device (SaMD). The software also gained FDA clearance last year for use by consumers—another first in the industry—empowering individuals to take an active role in their heart health.
In addition to providing the bioacoustics technology that powers Stethophone, Sparrow offers medical practitioners advanced visualizations that enable them to understand how to interpret diagnostics originating from the device. These visualizations aid in the recognition of patterns and anomalies that may not be easily discernible by conventional cardiac diagnostics, thereby potentially enhancing diagnostic accuracy and efficiency.
"We are at the forefront of a revolution in personalized heart health," Sparrow BioAcoustics CEO Mark Attila Opauzsky said. "Stethophone embodies our commitment to provide everyone the power, agency, and access to fight heart disease right in their own hands."
Sparrow is looking to the future with the development of Stethophone's next iteration, which explores integrating artificial intelligence functionalities into the bioacoustics engineering.
Stethophone is currently available on iOS.
Sparrow BioAcoustics, with offices in Newfoundland, Nova Scotia, and Toronto, Canada, is steering the Software as a Medical Device industry in new directions for cardiac and pulmonary disease detection. Its physicians, engineers and data scientists are working to unlock the richest source of diagnostic information about cardiac and pulmonary conditions.