Businesswire08.01.16
Second Sight Medical Products Inc., a developer, manufacturer and marketer of implantable visual prosthetics to restore some useful vision to blind patients, is expanding into Taiwan with Orient Europharma Co Ltd. and Iran with Arshia Gostar Darman Co Ltd. through exclusive agreements to distribute the Argus II Retinal Prosthesis System (Argus II).
In Taiwan, Orient Europharma is a well-established distributor deeply connected in the ophthalmology market. Taiwan is a densely populated state of 25 million people. Their healthcare system is one of the most advanced worldwide with a structured social security system managed by the Bureau of National Health Insurance.
In Iran, Arshia Gostar Darman is a company with a track record of introducing similar medical devices and building multi-million dollar businesses. The U.S. Department of Treasury has granted Second Sight approval to sell the Argus II retinal prosthesis system in Iran, which is the largest market in the Middle East with 80 million people.
"These agreements further expand our global reach and partnering in these markets bring the Argus II closer to those suffering from outer retinal degeneration,” said Will McGuire, president and CEO of Second Sight.
Each of the distributors will be responsible for executing in-country management of all aspects of Second Sight's technology, including sales, promotion, marketing, supporting national reimbursement, surgical, technical support and delivering rehabilitation.
There are an estimated 37,000 individuals in Taiwan and Iran with retinis pigmentosa.
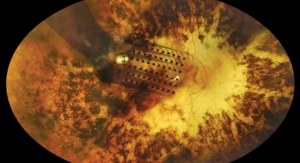
Second Sight's Argus II System provides electrical stimulation that bypasses the defunct retinal cells and stimulates remaining viable cells inducing visual perception in individuals with severe to profound outer retinal degeneration such as retinitis pigmentosa (RP). The Argus II works by converting images captured by a miniature video camera mounted on the patient's glasses into a series of small electrical pulses, which are transmitted wirelessly to an array of electrodes implanted on the surface of the retina. These pulses are intended to stimulate the retina's remaining cells, resulting in the perception of patterns of light in the brain. The patient then learns to interpret these visual patterns, thereby regaining some useful vision. The system is controlled by software and is upgradeable, which may provide improved performance as new algorithms are developed and tested. Therefore current and future Argus II users may benefit from the continuously improving technology. The Argus II is the first artificial retina to receive widespread approval, and is offered at approved centers in Austria, Canada, France, Germany, Italy, Netherlands, Saudi Arabia, Spain, Switzerland, Turkey, United Kingdom and the United States.
Second Sight develops, manufactures and markets implantable visual prosthetics to enable blind individuals to achieve greater independence. Second Sight has developed and now manufactures and markets the Argus II Retinal Prosthesis System. Enrollment has been completed in a feasibility trial to test the safety and utility of the Argus II in individuals with Dry Age-Related Macular Degeneration. Second Sight is also developing the Orion I Visual Cortical Prosthesis to restore some vision to individuals who are blind due to causes other than preventable or treatable conditions. The company's U.S. headquarters is in Sylmar, Calif., and its European headquarters is in Lausanne, Switzerland.
In Taiwan, Orient Europharma is a well-established distributor deeply connected in the ophthalmology market. Taiwan is a densely populated state of 25 million people. Their healthcare system is one of the most advanced worldwide with a structured social security system managed by the Bureau of National Health Insurance.
In Iran, Arshia Gostar Darman is a company with a track record of introducing similar medical devices and building multi-million dollar businesses. The U.S. Department of Treasury has granted Second Sight approval to sell the Argus II retinal prosthesis system in Iran, which is the largest market in the Middle East with 80 million people.
"These agreements further expand our global reach and partnering in these markets bring the Argus II closer to those suffering from outer retinal degeneration,” said Will McGuire, president and CEO of Second Sight.
Each of the distributors will be responsible for executing in-country management of all aspects of Second Sight's technology, including sales, promotion, marketing, supporting national reimbursement, surgical, technical support and delivering rehabilitation.
There are an estimated 37,000 individuals in Taiwan and Iran with retinis pigmentosa.
Second Sight's Argus II System provides electrical stimulation that bypasses the defunct retinal cells and stimulates remaining viable cells inducing visual perception in individuals with severe to profound outer retinal degeneration such as retinitis pigmentosa (RP). The Argus II works by converting images captured by a miniature video camera mounted on the patient's glasses into a series of small electrical pulses, which are transmitted wirelessly to an array of electrodes implanted on the surface of the retina. These pulses are intended to stimulate the retina's remaining cells, resulting in the perception of patterns of light in the brain. The patient then learns to interpret these visual patterns, thereby regaining some useful vision. The system is controlled by software and is upgradeable, which may provide improved performance as new algorithms are developed and tested. Therefore current and future Argus II users may benefit from the continuously improving technology. The Argus II is the first artificial retina to receive widespread approval, and is offered at approved centers in Austria, Canada, France, Germany, Italy, Netherlands, Saudi Arabia, Spain, Switzerland, Turkey, United Kingdom and the United States.
Second Sight develops, manufactures and markets implantable visual prosthetics to enable blind individuals to achieve greater independence. Second Sight has developed and now manufactures and markets the Argus II Retinal Prosthesis System. Enrollment has been completed in a feasibility trial to test the safety and utility of the Argus II in individuals with Dry Age-Related Macular Degeneration. Second Sight is also developing the Orion I Visual Cortical Prosthesis to restore some vision to individuals who are blind due to causes other than preventable or treatable conditions. The company's U.S. headquarters is in Sylmar, Calif., and its European headquarters is in Lausanne, Switzerland.