11.20.14
Second Sight Medical Products Inc., maker of artificial retinas designed to partially restore sight to the blind, had quite the financial coming out party on Wednesday, Nov. 19.
The company’s initial public offering price on NASDAQ was $9 on 3.5 million shares, which would raise $31.5 million. The stock hit a high of $22.45 on Wednesday. The stock was up to $19.80 after about an hour of trading, with about 1.6 million shares exchanged. The stock closed the day at $19.97. The company’s trading symbol is EYES. Midday on Thursday, Nov. 20, EYES was trading around $23.57.
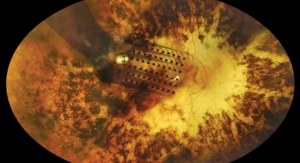
Second Sight’s technology, the Argus II system, which received U.S. Food and Drug Administration (FDA) approval in February last year, is a 10.3-millimeter (mm) by 3.2-mm device implanted in the eye. It functions via 60 electrodes (each measuring 0.2 mm) across a superthin film laid across the retina. The implant receives signals from two external components (a camera mounted on a pair of sunglasses and a communications module). The pulses produced by the implant from the camera’s images bypass dead photoreceptors and stimulate underlying neutrons. As a result, the patient is allowed to “see” a pattern of lights that give the patient the ability to make out fuzzy forms and shapes.
It is the only retinal prosthesis system currently approved by the FDA, and Argus II currently is indicated for U.S. patients with full vision loss due to Retinitis Pigmentosa.
The first commercial implantation of the device occurred in January at the University of Michigan’s Kellogg Eye Center. The first ever commercial implant of Argus II took place in Italy in 2011.
The company’s initial public offering price on NASDAQ was $9 on 3.5 million shares, which would raise $31.5 million. The stock hit a high of $22.45 on Wednesday. The stock was up to $19.80 after about an hour of trading, with about 1.6 million shares exchanged. The stock closed the day at $19.97. The company’s trading symbol is EYES. Midday on Thursday, Nov. 20, EYES was trading around $23.57.
Second Sight’s technology, the Argus II system, which received U.S. Food and Drug Administration (FDA) approval in February last year, is a 10.3-millimeter (mm) by 3.2-mm device implanted in the eye. It functions via 60 electrodes (each measuring 0.2 mm) across a superthin film laid across the retina. The implant receives signals from two external components (a camera mounted on a pair of sunglasses and a communications module). The pulses produced by the implant from the camera’s images bypass dead photoreceptors and stimulate underlying neutrons. As a result, the patient is allowed to “see” a pattern of lights that give the patient the ability to make out fuzzy forms and shapes.
It is the only retinal prosthesis system currently approved by the FDA, and Argus II currently is indicated for U.S. patients with full vision loss due to Retinitis Pigmentosa.
The first commercial implantation of the device occurred in January at the University of Michigan’s Kellogg Eye Center. The first ever commercial implant of Argus II took place in Italy in 2011.