09.04.14
Biosensors International Group Ltd. reports the enrollment of the first patient in BioFreedom USA, an investigational device exemption (IDE) feasibility trial designed to collect additional U.S.-based safety and effectiveness data for BioFreedom, the company's polymer and carrier-free drug-coated stent (DCS).
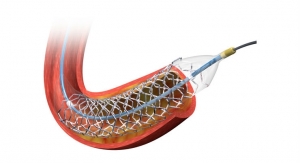
BioFreedom is the latest development in Biosensors' stent technology. It includes a micro-structured abluminal surface that permits the controlled release of a drug without the use of a polymer or other carrier. The drug used is highly lipophilic and anti-restenotic, and was developed by Biosensors specifically for use with stents. In its first-in-man study, treatment with BioFreedom demonstrated excellent 12-month late lumen loss and sustained safety up to four years, including absence of definite and/or probable stent thrombosis, company officials report.
The company believes this is the first clinical trial within the United States to evaluate a polymer-free DCS. Results from this study will support a future pivotal IDE study in the United States.
The trial design is multi-center and prospective, enrolling up to 100 patients at up to 10 centers. Due to the unique features of the BioFreedom DCS, the U.S. Food and Drug Administration (FDA) has approved a post-implant strategy requiring only three months of dual anti-platelet therapy (DAPT) for this trial. The initial patient has been enrolled in the trial by principal investigator, Ron Waksman, M.D., at the MedStar Washington Hospital Center in Washington D.C.
Biosensors received conditional IDE approval from the FDA to conduct a U.S.-based clinical trial of BioFreedom in May. Enrollment in this study is anticipated to last around four months from initial patient enrollment.
"I am very excited to be involved in this first U.S. study of a drug-coated stent," said Waksman. "This will hopefully prove to be the first step toward FDA approval of BioFreedom, offering U.S. patients all the benefits of a drug-eluting stent but with a shorter DAPT requirement."
The BioFreedom U.S. IDE feasibility trial is enrolling patients with symptomatic ischemic heart disease due to de novo native coronary artery lesions. The primary safety endpoint of the study is the occurrence of MACE (a composite of cardiac death, myocardial infarction, target lesion revascularization and definite stent thrombosis) within nine months following stent implantation. The primary efficacy endpoint is in-stent late lumen loss at nine months compared with a historical control.
Another important BioFreedom trial, LEADERS FREE, is currently applying a one-month DAPT strategy in patients at high risk of bleeding to further develop the product's safety and efficacy profile. The goal is to demonstrate that treatment with BioFreedom delivers the safety profile of a bare-metal stent with the anti-restenotic benefit of a drug-eluting stent.
BioFreedom received CE Mark approval in January 2013 and is currently available in select markets, but not the United States.
Biosensors is headquartered in Singapore.
BioFreedom is the latest development in Biosensors' stent technology. It includes a micro-structured abluminal surface that permits the controlled release of a drug without the use of a polymer or other carrier. The drug used is highly lipophilic and anti-restenotic, and was developed by Biosensors specifically for use with stents. In its first-in-man study, treatment with BioFreedom demonstrated excellent 12-month late lumen loss and sustained safety up to four years, including absence of definite and/or probable stent thrombosis, company officials report.
The company believes this is the first clinical trial within the United States to evaluate a polymer-free DCS. Results from this study will support a future pivotal IDE study in the United States.
The trial design is multi-center and prospective, enrolling up to 100 patients at up to 10 centers. Due to the unique features of the BioFreedom DCS, the U.S. Food and Drug Administration (FDA) has approved a post-implant strategy requiring only three months of dual anti-platelet therapy (DAPT) for this trial. The initial patient has been enrolled in the trial by principal investigator, Ron Waksman, M.D., at the MedStar Washington Hospital Center in Washington D.C.
Biosensors received conditional IDE approval from the FDA to conduct a U.S.-based clinical trial of BioFreedom in May. Enrollment in this study is anticipated to last around four months from initial patient enrollment.
"I am very excited to be involved in this first U.S. study of a drug-coated stent," said Waksman. "This will hopefully prove to be the first step toward FDA approval of BioFreedom, offering U.S. patients all the benefits of a drug-eluting stent but with a shorter DAPT requirement."
The BioFreedom U.S. IDE feasibility trial is enrolling patients with symptomatic ischemic heart disease due to de novo native coronary artery lesions. The primary safety endpoint of the study is the occurrence of MACE (a composite of cardiac death, myocardial infarction, target lesion revascularization and definite stent thrombosis) within nine months following stent implantation. The primary efficacy endpoint is in-stent late lumen loss at nine months compared with a historical control.
Another important BioFreedom trial, LEADERS FREE, is currently applying a one-month DAPT strategy in patients at high risk of bleeding to further develop the product's safety and efficacy profile. The goal is to demonstrate that treatment with BioFreedom delivers the safety profile of a bare-metal stent with the anti-restenotic benefit of a drug-eluting stent.
BioFreedom received CE Mark approval in January 2013 and is currently available in select markets, but not the United States.
Biosensors is headquartered in Singapore.