01.20.14
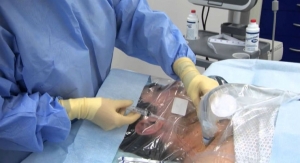
W.L. Gore & Associates Inc. has launched its Acuseal Vascular Graft for vascular access. Designed for early cannulation within 24 hours after implantation, the federally approved Gore Acuseal Vascular Graft expands treatment options for earlier removal and/or possible prevention of a central venous catheter –-- a major source of infection for hemodialysis patients.
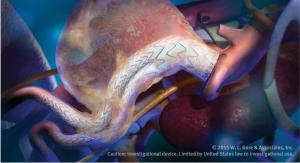
The graft's tri-layer design features a low-bleed elastomeric middle membrane between inner and outer layers of expanded polytetrafluoroethylene (ePTFE). The middle membrane hinders suture line and cannulation needle bleeding and may reduce the risk of seroma formation and pseudoaneurysm, the company claims. The Acuseal's design is kink resistant and flexible around curves while allowing for precise suturing and anastomotic tailoring.
A prospective, non-randomized, multi-center U.S. clinical trial involving 138 patients indicated the six-month cumulative patency of the Gore Acuseal graft is comparable to other arteriovenous grafts with 84 percent patency compared to 75 percent patency in the historical control. Results also showed that within 28 days of graft implantation, 75.6 percent of the implanted Gore Acuseal Vascular Grafts had successfully been cannulated three consecutive times, allowing for the potential for the central venous catheter to be removed.
“With early cannulation, physicians can reduce the number of days patients use tunnelled catheters for dialysis, which in turn will reduce the morbidity and mortality for these patients,” said Marc Glickman, M.D., principal investigator and chief of Vascular Services for Sentara Healthcare in Norfolk, Va. “The results of our trial demonstrate that the Gore Acuseal Vascular Graft does allow for early cannulation within 72 hours of implantation without the risk of cannulation-related complications such as infection and without reducing the patency of the graft.”
The ePTFE luminal surface of the Acuseal graft incorporates the Carmeda BioActive Surface (CBAS Surface). This proprietary end-point covalently bonded heparin technology is anchored to the graft surface and imparts thromboresistant properties to the vascular graft while achieving sustained anti-thrombotic bioactivity on the graft surface for an extended period of time. CBAS Surface is the same technology introduced with Gore's Propaten Vascular Graft.
“The new Gore Acuseal Vascular Graft represents Gore’s commitment to developing graft products that improve patient care,” said Cress Whitfield, sales leader, Surgical Vascular, Gore Medical. “By offering a graft that allows for early cannulation, physicians can provide patients with better treatment options.”
Based in Flagstaff, Ariz., privately held W.L. Gore & Associates develops electronics, fabrics, industrial and medical products. Its medical portfolio includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.
The graft's tri-layer design features a low-bleed elastomeric middle membrane between inner and outer layers of expanded polytetrafluoroethylene (ePTFE). The middle membrane hinders suture line and cannulation needle bleeding and may reduce the risk of seroma formation and pseudoaneurysm, the company claims. The Acuseal's design is kink resistant and flexible around curves while allowing for precise suturing and anastomotic tailoring.
A prospective, non-randomized, multi-center U.S. clinical trial involving 138 patients indicated the six-month cumulative patency of the Gore Acuseal graft is comparable to other arteriovenous grafts with 84 percent patency compared to 75 percent patency in the historical control. Results also showed that within 28 days of graft implantation, 75.6 percent of the implanted Gore Acuseal Vascular Grafts had successfully been cannulated three consecutive times, allowing for the potential for the central venous catheter to be removed.
“With early cannulation, physicians can reduce the number of days patients use tunnelled catheters for dialysis, which in turn will reduce the morbidity and mortality for these patients,” said Marc Glickman, M.D., principal investigator and chief of Vascular Services for Sentara Healthcare in Norfolk, Va. “The results of our trial demonstrate that the Gore Acuseal Vascular Graft does allow for early cannulation within 72 hours of implantation without the risk of cannulation-related complications such as infection and without reducing the patency of the graft.”
The ePTFE luminal surface of the Acuseal graft incorporates the Carmeda BioActive Surface (CBAS Surface). This proprietary end-point covalently bonded heparin technology is anchored to the graft surface and imparts thromboresistant properties to the vascular graft while achieving sustained anti-thrombotic bioactivity on the graft surface for an extended period of time. CBAS Surface is the same technology introduced with Gore's Propaten Vascular Graft.
“The new Gore Acuseal Vascular Graft represents Gore’s commitment to developing graft products that improve patient care,” said Cress Whitfield, sales leader, Surgical Vascular, Gore Medical. “By offering a graft that allows for early cannulation, physicians can provide patients with better treatment options.”
Based in Flagstaff, Ariz., privately held W.L. Gore & Associates develops electronics, fabrics, industrial and medical products. Its medical portfolio includes vascular grafts, endovascular and interventional devices, surgical meshes for hernia repair, soft tissue reconstruction, staple line reinforcement and sutures for use in vascular, cardiac, and general surgery.