02.17.15
Flagstaff, Ariz.-based W.L. Gore & Associates Inc. has completed primary enrollment in the Gore Excluder Iliac Branch Clinical Study a prospective, multi-center, non-randomized, single-arm study designed to assess the safety and effectiveness of the Gore Excluder iliac branch endoprosthesis in treating common iliac artery aneurysms or aorto-iliac aneurysms.
A total of 62 patients have received the device, which completed the required enrollment of 60 set by the U.S. Food and Drug Administration (FDA). The FDA also approved a Continued Access Protocol (CAP) allowing an additional 140 patients to be treated with the device. The CAP also includes use of the Gore Excluder iliac branch endoprosthesis for bilateral placement.
“An estimated 20 to 40 percent of patients with abdominal aortic aneurysms have common iliac arteries that also require repair, yet no device has been approved for this treatment indication in the United States,” said Darren Schneider, M.D., chief of vascular and endovascular surgery at the Weill Cornell Medical College and principal investigator for the national trial. “This iliac branch device provides a wider range of patients with access to minimally invasive, endovascular treatment options.”
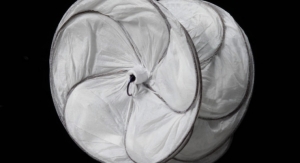
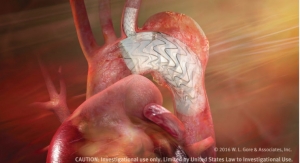
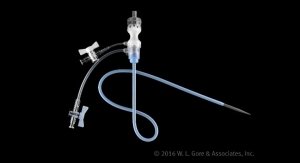
The Gore Excluder Iliac Branch Device (iliac branch component and internal iliac component) is used in conjunction with the Gore Excluder AAA (abdominal aortic aneurysm) endoprosthesis to isolate the common iliac artery from systemic blood flow and preserve blood flow in the external iliac and internal iliac arteries.
“Completing primary enrollment marks a significant step toward achieving FDA approval for the Gore Excluder iliac branch device,” said Ryan Takeuchi, aortic business unit leader at Gore. “If approved, the iliac branch endoprosthesis would become the first and only device indicated to treat common iliac aneurysms in the United States.”
In October 2013, the Gore Excluder iliac branch endoprosthesis earned the CE mark.
A total of 62 patients have received the device, which completed the required enrollment of 60 set by the U.S. Food and Drug Administration (FDA). The FDA also approved a Continued Access Protocol (CAP) allowing an additional 140 patients to be treated with the device. The CAP also includes use of the Gore Excluder iliac branch endoprosthesis for bilateral placement.
“An estimated 20 to 40 percent of patients with abdominal aortic aneurysms have common iliac arteries that also require repair, yet no device has been approved for this treatment indication in the United States,” said Darren Schneider, M.D., chief of vascular and endovascular surgery at the Weill Cornell Medical College and principal investigator for the national trial. “This iliac branch device provides a wider range of patients with access to minimally invasive, endovascular treatment options.”
The Gore Excluder Iliac Branch Device (iliac branch component and internal iliac component) is used in conjunction with the Gore Excluder AAA (abdominal aortic aneurysm) endoprosthesis to isolate the common iliac artery from systemic blood flow and preserve blood flow in the external iliac and internal iliac arteries.
“Completing primary enrollment marks a significant step toward achieving FDA approval for the Gore Excluder iliac branch device,” said Ryan Takeuchi, aortic business unit leader at Gore. “If approved, the iliac branch endoprosthesis would become the first and only device indicated to treat common iliac aneurysms in the United States.”
In October 2013, the Gore Excluder iliac branch endoprosthesis earned the CE mark.