11.23.13
OncoSil Medical Ltd. has been awarded a U.S. patent covering its OncoSil nuclear medical device that treats locally advanced pancreatic cancer.
The Australian company plans to initiate a pivotal study for the device in the first quarter of 2014 with 100 to 300 patients.
Patent 13/314,239, titled “Devices and Methods for the Treatment of Cancer” was awarded by the U.S. Patent and Trademark Office and extends to 2022.
OncoSil is an active implantable medical device that provides a localised radiation therapy for tumours, avoiding the systemic side effects of external radiation treatment.
The firm already has two successful Phase II studies under Enigma Therapeutics, which OncoSil acquired in April this year.
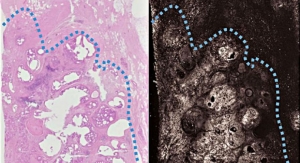
Analysis of patients in the first pilot trial had demonstrated significant tumouricial (cancer cell destroying) activity with a disease control rate of 82 percent along with a reduction in pain.
A second study confirmed the 400Gy radiation dose was well tolerated and that no adverse events with the device occurred. This also demonstrated a tumour control rate was 100 percent, and confirmed stabilisation of tumour growth but was stopped at an early stage due to a change in focus by the previous owner.
OncoSil Medical develops radiation therapeutic delivery methods for tumors.
The Australian company plans to initiate a pivotal study for the device in the first quarter of 2014 with 100 to 300 patients.
Patent 13/314,239, titled “Devices and Methods for the Treatment of Cancer” was awarded by the U.S. Patent and Trademark Office and extends to 2022.
OncoSil is an active implantable medical device that provides a localised radiation therapy for tumours, avoiding the systemic side effects of external radiation treatment.
The firm already has two successful Phase II studies under Enigma Therapeutics, which OncoSil acquired in April this year.
Analysis of patients in the first pilot trial had demonstrated significant tumouricial (cancer cell destroying) activity with a disease control rate of 82 percent along with a reduction in pain.
A second study confirmed the 400Gy radiation dose was well tolerated and that no adverse events with the device occurred. This also demonstrated a tumour control rate was 100 percent, and confirmed stabilisation of tumour growth but was stopped at an early stage due to a change in focus by the previous owner.
OncoSil Medical develops radiation therapeutic delivery methods for tumors.