07.17.14
Aussie company OncoSil Medical Ltd. has begun work with German manufacturing partner Eckert & Ziegler in a key step to bringing its OncoSil localized radiation therapy treatment for pancreatic cancer to clinical trial stage.
The manufacturing supply chain has also been established with OncoSil securing sufficient supplies of the raw materials and intermediates required to manufacture the product for the pivotal clinical trial globally.
OncoSil officials also noted the successful commissioning and re-validation of its manufacturing process and quality system would support both the CE mark audit and the U.S. Investigational Device Exemption (IDE) submission.
Securing the CE mark would allow sales of OncoSil from its manufacturing site in Germany to the European Union while the IDE is the first step to securing U.S. Food and Drug Administration commercial approval under a premarket approval.
“We are extremely pleased to report that the manufacturing revalidation which will facilitate the production of OncoSil for use in its pivotal clinical trial and also for future sales as part of the CE mark process has now been completed,” CEO Neil Frazer, M.B.,Ch.B., FRCA, FFPM said. “This represents a major component of the trial process, as it ensures we have sufficient, quality product to meet the demands of our trial participants at various locations across the globe.”
The manufacturing process review had involved revalidation and requalification of facilities and work previously completed.This confirmed that the manufacturing and quality systems are of a standard that will support manufacture of OncoSil for the trial and also for future commercial use.
OncoSil commenced the proceedings for its pivotal clinical trial, which will enroll 150 patients across up to 20 trial sites, in March. The company plans to roll-out the trial in Australia, in parallel with trial sites in the United Kingdom, Belgium, Singapore and then the United States once IDE approval is earned. Engaging hospitals as trial sites will be a key initial focus, followed by patient recruitment and then the commencement of dosing patients under the trial. The trial will compare patients receiving standard-of-care (for pancreatic cancer it is chemotherapy treatment) with patients receiving standard-of-care plus OncoSil treatment in a randomized and controlled fashion. Patients will be randomized with 100 subjects to receive OncoSil plus chemotherapy while 50 patients will receive chemotherapy alone.
The company was awarded ethics approval for the Australian hospital sites proposed for the trial in July.
If positive, data generated by the trial may facilitate the commercialization of OncoSil, including in the United States, which is the world’s largest health care market.
The trial also has the potential to be a Global Registration Study, the company reports.
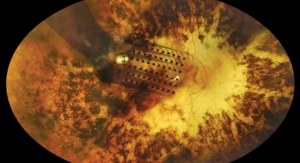
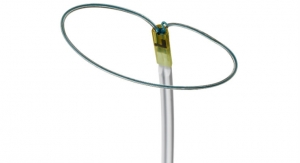
OncoSil is an implantable device that emits radiation directly into a pancreatic tumor, and the surrounding pain conducting nerves, and delivers radiation therapy locally for up to three months.
The device is inserted directly into the center of the tumor, and emits targeted beta radiation to the site in a short 15-30 minute procedure. This has been proven to kill tumor cells, and provides the major benefit of reducing systemic bodily exposure to radiation.
Based in Sydney, Australia, OncoSil is the provider of its patented beta radiation technology for the treatment of cancerous tumors.
The manufacturing supply chain has also been established with OncoSil securing sufficient supplies of the raw materials and intermediates required to manufacture the product for the pivotal clinical trial globally.
OncoSil officials also noted the successful commissioning and re-validation of its manufacturing process and quality system would support both the CE mark audit and the U.S. Investigational Device Exemption (IDE) submission.
Securing the CE mark would allow sales of OncoSil from its manufacturing site in Germany to the European Union while the IDE is the first step to securing U.S. Food and Drug Administration commercial approval under a premarket approval.
“We are extremely pleased to report that the manufacturing revalidation which will facilitate the production of OncoSil for use in its pivotal clinical trial and also for future sales as part of the CE mark process has now been completed,” CEO Neil Frazer, M.B.,Ch.B., FRCA, FFPM said. “This represents a major component of the trial process, as it ensures we have sufficient, quality product to meet the demands of our trial participants at various locations across the globe.”
The manufacturing process review had involved revalidation and requalification of facilities and work previously completed.This confirmed that the manufacturing and quality systems are of a standard that will support manufacture of OncoSil for the trial and also for future commercial use.
OncoSil commenced the proceedings for its pivotal clinical trial, which will enroll 150 patients across up to 20 trial sites, in March. The company plans to roll-out the trial in Australia, in parallel with trial sites in the United Kingdom, Belgium, Singapore and then the United States once IDE approval is earned. Engaging hospitals as trial sites will be a key initial focus, followed by patient recruitment and then the commencement of dosing patients under the trial. The trial will compare patients receiving standard-of-care (for pancreatic cancer it is chemotherapy treatment) with patients receiving standard-of-care plus OncoSil treatment in a randomized and controlled fashion. Patients will be randomized with 100 subjects to receive OncoSil plus chemotherapy while 50 patients will receive chemotherapy alone.
The company was awarded ethics approval for the Australian hospital sites proposed for the trial in July.
If positive, data generated by the trial may facilitate the commercialization of OncoSil, including in the United States, which is the world’s largest health care market.
The trial also has the potential to be a Global Registration Study, the company reports.
OncoSil is an implantable device that emits radiation directly into a pancreatic tumor, and the surrounding pain conducting nerves, and delivers radiation therapy locally for up to three months.
The device is inserted directly into the center of the tumor, and emits targeted beta radiation to the site in a short 15-30 minute procedure. This has been proven to kill tumor cells, and provides the major benefit of reducing systemic bodily exposure to radiation.
Based in Sydney, Australia, OncoSil is the provider of its patented beta radiation technology for the treatment of cancerous tumors.