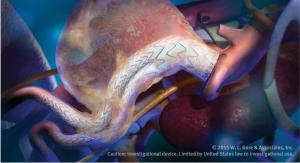
The U.S. Food and Drug Administration (FDA) has approved a device from W.L. Gore & Associates Inc. for the endovascular repair of acute and chronic Type B dissections of the descending thoracic aorta. The company claims its Conformable Gore Tag Thoracic Endoprosthesis is the only product to receive FDA approval for aneurysm, trauma and dissection.
An aortic dissection is the tearing of the innermost layer of the aortic wall, allowing blood to flow between the inner and outer layers of the vessel wall causing separation. If not stabilized, the dissection can increase the risk of rupture, cause complications with blood flow to the lower body and harm nearby organs. Until now, the only approved treatment options were medical management or open surgical repair.
"Vascular surgeons have for some time awaited an approved indication for treating certain Type B aortic dissection patients since the consensus has been that endovascular stent graft repair has emerged as the treatment of choice," said Richard Cambria, M.D., chief of the Division of Vascular and Endovascular Surgery and co-director of the Thoracic Aortic Center at Massachusetts General Hospital's Vascular Center in Boston. "Now, surgeons will be able to present patients with a minimally invasive treatment option and the exchange of information and experience among surgeons will be more acceptable now that the procedure is approved."
"Having an FDA-approved Type B dissection indication is very important, as the impact this has on the endovascular community and the eligible patient population is huge," said Joseph Bavaria, M.D., Roberts/Measey Professor of Surgery, vice chairman of the Division of Cardiovascular Surgery and director of the Thoracic Aortic Surgery Program at the University of Pennsylvania.
The Conformable GORE TAG Device offers conformability and ease of use, while accommodating tortuous and tapered anatomy. The device resists compression and has a broad oversizing window ranging from 6 percent to 33 percent. Physicians are able to choose the appropriate oversizing for the patient anatomy, which is particularly important in dissection patients due to the delicate nature of the disease.
The Conformable GORE TAG Thoracic Endoprosthesis is delivered via catheter and inserted into the femoral artery through a small incision in the groin and carefully guided up the leg artery through the abdomen into the chest and to the implantation site. The device is available in diameters of 21-45 mm, allowing for the treatment of patients with aortic diameters of 16-42 mm. Tapered device configurations are also available.
"[This] approval further advances the use of endovascular devices as standard of care and allows more patients to benefit from this preferred treatment option," said Ryan Takeuchi, Gore's Aortic Business Leader. "Gore was the first to gain approval for thoracic aortic aneurysm followed by aortic traumas using our Conformable GORE TAG Device. We are proud to offer surgeons the only FDA-approved, minimally invasive treatment for Type B dissections."
Gore develops electronics, fabrics, industrial items and medical products.