Mark Crawford, Contributing Writer02.07.22
A Class III medical device classification is for implanted medical devices that are used to support life or prevent the potential risk of illness or injury. These sterile devices or components are placed directly into the human body and must be biocompatible.
Roughly 10 percent of all medical devices fall under this category. Examples of Class III devices include orthopedic implants, pacemakers, cardiovascular stents, and respiratory ventilators.
Compromised packaging for these sterile devices is a significant risk to patient safety. Once bacteria or other contamianants enter the container, the device is no longer sterile and cannot be used. An undetected breach in a seal presents a significant health concern, perhaps at a critical moment of use, and can lead to surgical delays or adverse patient outcomes that can result in litigation or recall.
Medical devices have a broad range of package and delivery systems, ranging from porous flexible packaging to non-porous rigid containers. The variety in product types and packaging options create different inspection challenges that must be met to ensure seal strength, sterility, and quality.
Seal-Testing Methods
To maintain the quality of packaged Class III products, package and seal integrity is absolutely critical. There are two ways to approach package integrity testing:
A variety of seal-testing methods are available to medical device manufacturers (MDMs), such as dye testing, peel tests, burst tests, and seal strength, but none of these are a perfect solution for seal testing—for example, these methods are often destructive or time-consuming (dye testing can take up to 48 hours).
Even with these testing technologies available, many MDMs still prefer a 100-percent, in-line manual visual inspection by an operator for CCI testing. However, this is also one of the least reliable methods for ensuring quality and detection of defects. Failure in manual visual inspection is predominantly attributable to human biases, fatigue, and the fact that not all physical defects can be seen.
According to ASTM Method F1886, manual visual inspection has the capability to detect a 75-micron channel defect 60-100 percent of the time, a wide range that presents a higher level of risk for Class III medical devices compared to newer testing methods. The ability to visually detect defects also diminishes the longer an inspector works on the line and becomes increasingly tired. Manual visual inspection is also less effective depending on certain materials, such as multi-layered foil for some package formats.
Airborne Ultrasound Technology
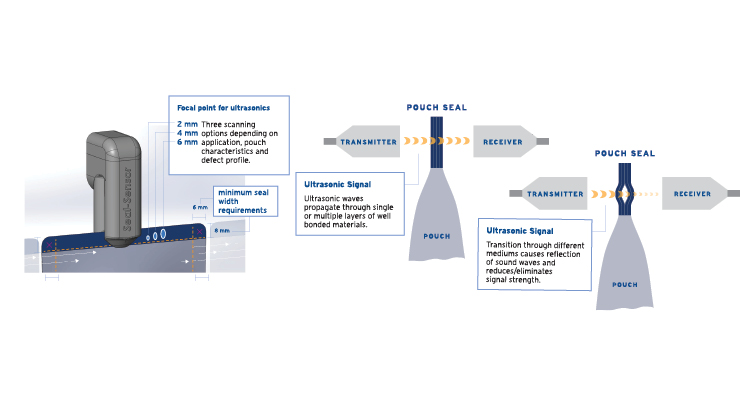
Inspection methods that provide faster and more reliable, sensitive, and traceable data are in high demand by MDMs as they try to improve efficiencies, reduce risk, streamline approvals, and control costs. One of the best ways to achieve this is using airborne ultrasound technology, which tests seals with a quick, accurate, non-destructive, and non-invasive method that provides high levels of sensitivity and reliability. Airborne ultrasound is ideal for a wide range of material types, including Tyvek, paper, foil, film, aluminum, plastic, and poly laminates.
The process works by passing the pouch seal in a direct line between an ultrasonic transmitter and receiver. Ultrasound waves propagate through the package seal, causing the reflection of sound waves. Defects are identified by measuring the variation of the reflected signal strength. Airborne ultrasound is a rapid-fire sensor pulsing sound through the seal at 1,000 pulses per second. Tyvek pouches and other flexible packaging systems can have the final seal scanning rate of 20 inches/second, with the ability to detect even the smallest seal defects and seal quality issues.
Any material fold, foreign media inclusion, or even a microscopic air gap between layers of pouch material can cause significant reflection, resulting in a transmitted signal. A small cut, abrasion, or missing layer in a material causes less reflection, so the transmitted signal is higher. Therefore, the level of a signal received after passing through the seal is a function of the seal quality. Various types of defects—visible and invisible, leaking and non-leaking, process-related and random—are detectable.
Airborne ultrasound can be automated in several ways. It can be mounted into the production flow, capturing seal quality as the pouches exit a band-sealer, or transfer through a production line.
Inline or Offline
Airborne ultrasound can be deployed within many production settings with a high reliability of detecting critical defects.
As an inline tool on the production line, it can test seals for defects non-destructively 100 percent inline, including incomplete seals, partial or weak areas in seals, and common seal defects that affect product quality and shelf life. A single linear scan of the seal takes less than one second and produces a pass/fail result as well as quantitative, traceable data. The technology can be integrated into pouch sealing systems and continuous band sealers.
Airborne ultrasound can also be used as an offline laboratory analytical tool for seal quality inspection, testing individual pouch seals or materials one sample at a time. The semi-automatic inspection system with x-y drive can non-destructively identify and evaluate seal defects, seal characterization, and material analysis. Advanced digital imaging software tools for process control provide in-depth seal quality analysis, including a pixel-by-pixel evaluation of seals in a high-resolution image that characterizes overall quality and uniformity of the seal.
Online and offline airborne ultrasound can be used together to not only identify defects, but also to determine where it is occurring in the line, which enables quick correction of the problem.
Take Your Testing to the Next Level
Airborne ultrasound has been established as one of the most effective methods for non-destructive testing of flexible packages and was given ASTM Test Method F3004-13 status in 2013, which is also an FDA Recognized Consensus Standard for seal quality inspection. Airborne ultrasound is a referenced technology in the USP <1207> Chapter Guidance as a deterministic test method for seal quality testing.
FDA warning letters to MDMs often relate to seal defects in medical device packaging, which ultimately results in delays and higher operational costs.
Airborne ultrasound technologies are non-destructive, non-subjective, and provide MDMs with a highly accurate seal-testing solution that improves quality, throughput, and process control, while greatly reducing product waste and cost, as well as risks for defects and recalls.
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.
Roughly 10 percent of all medical devices fall under this category. Examples of Class III devices include orthopedic implants, pacemakers, cardiovascular stents, and respiratory ventilators.
Compromised packaging for these sterile devices is a significant risk to patient safety. Once bacteria or other contamianants enter the container, the device is no longer sterile and cannot be used. An undetected breach in a seal presents a significant health concern, perhaps at a critical moment of use, and can lead to surgical delays or adverse patient outcomes that can result in litigation or recall.
Medical devices have a broad range of package and delivery systems, ranging from porous flexible packaging to non-porous rigid containers. The variety in product types and packaging options create different inspection challenges that must be met to ensure seal strength, sterility, and quality.
Seal-Testing Methods
To maintain the quality of packaged Class III products, package and seal integrity is absolutely critical. There are two ways to approach package integrity testing:
- 100 percent in-line testing—All products in a batch are tested at an acceptable level of container closure integrity (CCI) assurance in a production line at high speed.
- Small-batch testing—A statistically significant batch size is tested for specific quality attributes and extrapolated to reflect the quality of the production lot.
A variety of seal-testing methods are available to medical device manufacturers (MDMs), such as dye testing, peel tests, burst tests, and seal strength, but none of these are a perfect solution for seal testing—for example, these methods are often destructive or time-consuming (dye testing can take up to 48 hours).
Even with these testing technologies available, many MDMs still prefer a 100-percent, in-line manual visual inspection by an operator for CCI testing. However, this is also one of the least reliable methods for ensuring quality and detection of defects. Failure in manual visual inspection is predominantly attributable to human biases, fatigue, and the fact that not all physical defects can be seen.
According to ASTM Method F1886, manual visual inspection has the capability to detect a 75-micron channel defect 60-100 percent of the time, a wide range that presents a higher level of risk for Class III medical devices compared to newer testing methods. The ability to visually detect defects also diminishes the longer an inspector works on the line and becomes increasingly tired. Manual visual inspection is also less effective depending on certain materials, such as multi-layered foil for some package formats.
Airborne Ultrasound Technology
Inspection methods that provide faster and more reliable, sensitive, and traceable data are in high demand by MDMs as they try to improve efficiencies, reduce risk, streamline approvals, and control costs. One of the best ways to achieve this is using airborne ultrasound technology, which tests seals with a quick, accurate, non-destructive, and non-invasive method that provides high levels of sensitivity and reliability. Airborne ultrasound is ideal for a wide range of material types, including Tyvek, paper, foil, film, aluminum, plastic, and poly laminates.
The process works by passing the pouch seal in a direct line between an ultrasonic transmitter and receiver. Ultrasound waves propagate through the package seal, causing the reflection of sound waves. Defects are identified by measuring the variation of the reflected signal strength. Airborne ultrasound is a rapid-fire sensor pulsing sound through the seal at 1,000 pulses per second. Tyvek pouches and other flexible packaging systems can have the final seal scanning rate of 20 inches/second, with the ability to detect even the smallest seal defects and seal quality issues.
Any material fold, foreign media inclusion, or even a microscopic air gap between layers of pouch material can cause significant reflection, resulting in a transmitted signal. A small cut, abrasion, or missing layer in a material causes less reflection, so the transmitted signal is higher. Therefore, the level of a signal received after passing through the seal is a function of the seal quality. Various types of defects—visible and invisible, leaking and non-leaking, process-related and random—are detectable.
Airborne ultrasound can be automated in several ways. It can be mounted into the production flow, capturing seal quality as the pouches exit a band-sealer, or transfer through a production line.
Inline or Offline
Airborne ultrasound can be deployed within many production settings with a high reliability of detecting critical defects.
As an inline tool on the production line, it can test seals for defects non-destructively 100 percent inline, including incomplete seals, partial or weak areas in seals, and common seal defects that affect product quality and shelf life. A single linear scan of the seal takes less than one second and produces a pass/fail result as well as quantitative, traceable data. The technology can be integrated into pouch sealing systems and continuous band sealers.
Airborne ultrasound can also be used as an offline laboratory analytical tool for seal quality inspection, testing individual pouch seals or materials one sample at a time. The semi-automatic inspection system with x-y drive can non-destructively identify and evaluate seal defects, seal characterization, and material analysis. Advanced digital imaging software tools for process control provide in-depth seal quality analysis, including a pixel-by-pixel evaluation of seals in a high-resolution image that characterizes overall quality and uniformity of the seal.
Online and offline airborne ultrasound can be used together to not only identify defects, but also to determine where it is occurring in the line, which enables quick correction of the problem.
Take Your Testing to the Next Level
Airborne ultrasound has been established as one of the most effective methods for non-destructive testing of flexible packages and was given ASTM Test Method F3004-13 status in 2013, which is also an FDA Recognized Consensus Standard for seal quality inspection. Airborne ultrasound is a referenced technology in the USP <1207> Chapter Guidance as a deterministic test method for seal quality testing.
FDA warning letters to MDMs often relate to seal defects in medical device packaging, which ultimately results in delays and higher operational costs.
Airborne ultrasound technologies are non-destructive, non-subjective, and provide MDMs with a highly accurate seal-testing solution that improves quality, throughput, and process control, while greatly reducing product waste and cost, as well as risks for defects and recalls.
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.