Sam Brusco, Associate Editor05.01.20
In the early 1990s, the Stanford Research Institute (SRI) developed a prototype that caught the attention of the Defense Advanced Research Projects Administration (DARPA). DARPA thought the technology would be a welcome tool to perform remote surgeries on soldiers wounded on the battlefield.
Dr. Frederick Moll, a surgeon, had non-military applications in mind when he first saw the surgical robotic system in 1994. He became acquainted with venture capitalist John Freund a year later. Freund was able to negotiate a technology license for SRI’s robotic surgical system. Together with engineer Robert Younge, Dr. Moll and Freund began their enterprise to improve minimally invasive surgery—Intuitive Surgical.
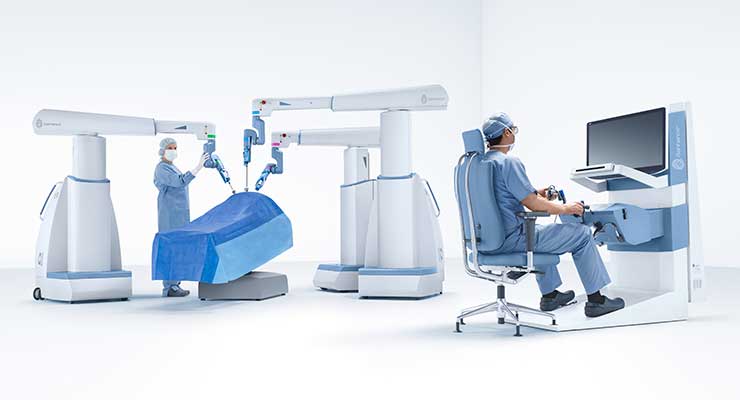
The now renowned da Vinci surgical system was commercialized in 1999 in Europe, and a year later came to the U.S. In June 2000, Intuitive went public with a $46 million initial public offering. The company was immediately sued for patent infringement in August 2000 by close competitor Computer Motion, an early entrant to the market with its ZEUS robotic surgical system. (Intuitive acquired Computer Motion in 2003.)
da Vinci’s first FDA approval was for general laparoscopic surgery to treat gallbladder and gastroesophageal diseases. Over the years, the FDA has approved da Vinci and its iterations for prostate surgery, thorascopic surgery, certain cardiac procedures, and gynecologic procedures.
In May 2018, da Vinci took a major step thanks to FDA clearance for the da Vinci SP (single port) surgical system for urologic procedures. Its ability to enter the body through a single, small incision aids surgeons in performing more complex procedures. Intuitive plans to pursue further regulatory clearances for transoral, transanal, and extraperitoneal procedures.
“Our da Vinci SP compliments da Vinci X and Xi systems by enabling surgeons to access narrow workspaces while maintaining high quality vision, precision, and control that surgeons have come to trust from da Vinci systems,” Intuitive CEO Gary Guthart told the press.
The system features three, multi-jointed, wristed instruments and a fully wristed 3D HD camera. The instruments and the camera emerge through a single cannula and are properly triangulated around target anatomy to avoid external instrument collisions. Surgeons control the fully articulating instruments and camera on the same surgeon console as the da Vinci X and Xi systems.
The company’s second product franchise, the Ion flexible robotic catheter, gained an FDA nod last February for minimally invasive peripheral lung biopsy. But Intuitive wasn’t first to market on this occasion. Auris Health, a company cofounded by Dr. Moll after he left Intuitive in 2003, secured FDA clearance for a device to perform lung biopsies. (Johnson & Johnson went on to buy Auris Health for $3.4 billion last April.)
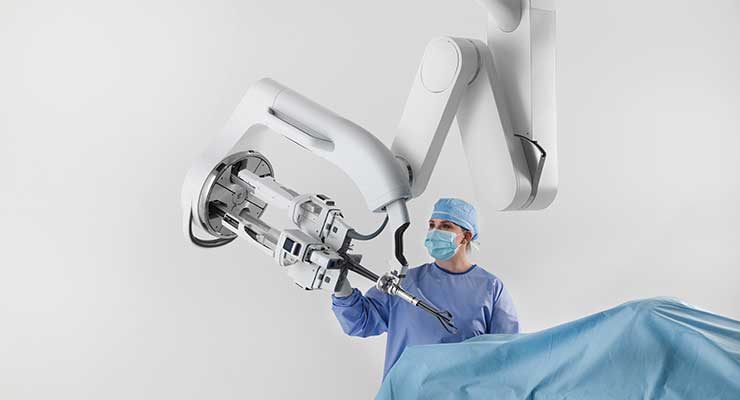
After nearly two decades holding a near monopoly on the surgical robotics market, new competitors are emerging for Intuitive. Morrisville, N.C.-based TransEnterix Surgical Inc. won FDA approval for its Senhance Surgical Robotic System in 2017. At the time, it was the first new entrant into the abdominal surgical robotic field since 2000. The Senhance augments laparoscopic surgical instrument and endoscopic camera precision, visualization, and control. It utilizes an open architecture so hospitals and surgeons can leverage existing technology in the OR with the Senhance. The FDA cleared 3mm instruments for the Senhance in 2018, which according to president and CEO Anthony Fernando, made it the first digital system to use instruments that small on a robotic interface.
“Taken together, the Senhance Surgical System improves laparoscopic surgery by allowing for maximum precision of instruments and surgeon control of their camera and tools,” said Fernando. “It minimizes invasiveness by offering the smallest instruments and also minimizing forces generated during surgery. It allows for surgeons to operate in long, complex cases with minimal fatigue when seated at a console. The goal is to reduce variability in laparoscopic procedures with digital assistance at a reasonable cost, thanks to reusable instrumentation.”
According to TransEnterix, the Senhance is the first digital surgery system with haptic feedback to measure and limit forces generated on the instruments or the patient’s abdominal wall. It also boosts ergonomics by allowing surgeons to sit comfortably in an ergonomic position during long, complex surgeries. Surgeons have long been approaching the boundaries of minimally invasive procedures using handheld, manual instruments and cameras.
“We seek to reduce the physical and cognitive load on surgeons through application of digital and robotic assistance,” said Fernando. “The first area is ergonomics. By allowing a surgeon to sit upright and always keep their hands in a comfortable position, the system keeps a surgeon in an ergonomic position while performing complex surgery. The system filters tremor, stabilizes instruments, and scales motion to improve control in small, precise motions. They can easily control the endoscopic camera by having the system track their eyes to not interrupt the flow of surgery. The new augmented intelligence system moves the camera with a new level of ease by tracking instruments to allow focus on the surgical task with optimized camera movement.”
The company obtained FDA clearance for its Intelligent Surgical Unit (ISU) to enable machine vision capabilities on the Senhance this past March. Machine vision drives surgeon camera control by responding to commands and recognizing certain objects and locations in the surgical field. The ISU hardware will be compatible with planned future augmented intelligence features like scene cognition and surgical image analytics. The ISU is also compatible with third-party vision systems the Senhance currently supports.
“At its core, the ISU lets a computer see, process, and augment surgical images,” said Fernando. “The set of features enabled by the initial approval are related to camera control augmented by machine vision. Now a surgeon can have the system recognize various instruments in the field and track the camera to their movement. The system can quickly zoom in and out on a particular point, and can move the camera to a desired location with one click. When using an off-angle scope, zooming in and out will maintain the image horizon with a constant image center throughout the zooming process, eliminating the need to pan the camera in addition to zooming. These features combine with eye-tracking to make Senhance a sophisticated and full-featured camera control mechanism for digital surgery.”
Since its launch, the Senhance’s indications have been expanded to include laparoscopic inguinal hernia and laparoscopic cholecystectomy (gallbladder removal) surgery in the U.S., and the ability to treat pediatric patients in Europe. The Senhance Ultrasonic System gained FDA clearance last January as well, enabling use of instruments that deliver controlled energy to ligate and divide tissue and minimize thermal injury to surrounding structures.
Multi-port robotic surgery requiring multiple incisions has been practiced for about 20 years. It’s becoming the new care standard in several surgical disciplines. Unlike traditional laparoscopic surgeries, today’s robotic technologies provide surgical precision, tremor-free dexterity, 3D HD visualization and natural user interface, while allowing more comfortable operation than manual laparoscopic surgery.
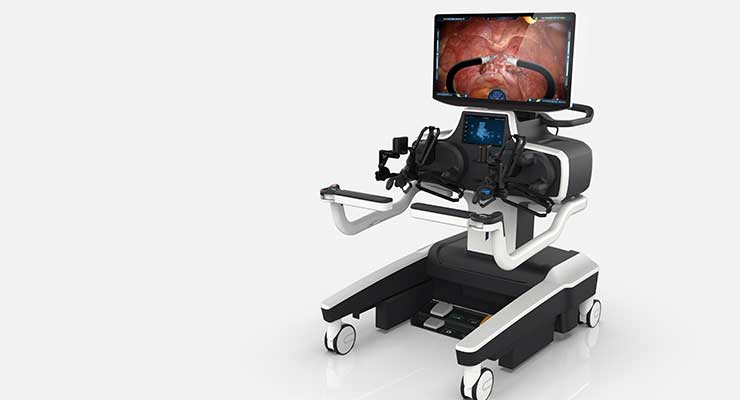
Toronto, Ontario-based Titan Medical’s single-port robotic surgery system is still under development. Last year, the company successfully completed two critical tasks for the system’s FDA IDE filing: acute and chronic procedures performed under Good Laboratory Practice principles and a human factors evaluation.
“Similar to traditional laparoscopic surgeries, most of today’s robotic surgeries still require at least three and as many as six different incisions,” said David McNally, president and CEO of Titan Medical. “Our single-port robotic system was designed with surgeon input from the very beginning, improves upon existing multi-port systems, and offers minimally invasive surgical options through only a single incision while maintaining the primary surgeon benefits afforded by robotic technology. By enabling surgeons to minimize the number of incisions, our robot potentially offers reduced trauma, faster recovery times, and minimal scarring.”
The Titan Medical single-port surgery system’s multi-articulated instruments have single-use, replaceable tips for grasping, suturing, cutting, and coagulation. Its workstation (which is maneuvered on swiveling coasters) features a flatscreen, 3D high definition display, a multi-articulated handle interface, and configurable elbow rest and foot pedal positioning. It has a single-arm mobile patient cart and 25mm-diameter camera insertion tube. The single-arm configuration has no external moving parts to ease simple setup and assistant-friendly procedures.
Ultimately, Titan Medical hopes to expand robotic surgery into currently underserved areas like general abdominal, gynecologic, urologic, and colorectal.
“Our three primary goals for this year are to secure adequate financing to resume product development in coordination with our outsourcing partners; execute planned improvements to the instruments, camera systems, and accessories based on additional surgeon input received during our GLP studies; and perform software development, verification, and validation in preparation for our planned IDE studies.”
Munich, Germany-based Brainlab creates and integrates software and systems for various surgical procedure stages: pre-planning, surgical navigation, data management and manipulation, and intraoperative imaging and surgical follow-up. Surgical navigation—also known as image-guided surgery, computer-assisted surgery, or software-guided surgery—continuously tracks instrumentation in relation to patient anatomy and displays the relationship in real-time of a screen during surgery.
“Turning time-consuming human interaction into automatic digital processes is the best way to decrease surgical time and increase surgical productivity,” said Meraj Khan, director of Brainlab’s neurosurgery portfolio. “After integrating thousands of ORs across the globe for the last twenty years, we’ve identified four key ways which hospitals should digitalize processes to enhance efficiency: consolidating data influx into a single control platform, streamlining documentation flow, improving communication in and outside the OR, and facilitating planning from anywhere.”
“As we focus on the operating room setting in the coming year, our technology continues to excite users,” Khan continued. “We bring value in clinical flexibility by integrating all types of pre-operative and intra-operative imaging modalities. Brainlab has a diverse automatic image registration option to connect an intraoperative imaging device whether it is a 2D C-arm, a 3D C-arm/cone beam CT (such as the Medtronic O-Arm, Siemens CIOS Spin, or Ziehm RFD3D), true intraoperative CT (such as Airo), or our newly launched Loop-X* Mobile Imaging Robot.”
The company’s suite of spine surgery products helps to effectively plan and execute spine procedures and place pedicle screws more accurately while minimizing the surgical team and patient’s X-ray exposure. Brainlab Spinal Navigation can address tumors and deformities, as well as routing cervical, thoracic, or lumbar spine surgeries. Instrument-tracking software helps avoid critical structures. Image registration and compatible imaging device options are broad so the procedure and imaging devices can be tailored to the surgeon’s needs.
“Brainlab spine surgery planning software has been designed for automated functionalities enabling effective and straightforward treatment planning. The Brainlab Elements 3D Viewer, for example, allows fast, easy access to medical image data, quick and intuitive interaction with DICOM images, and direct transfer to Brainlab Spine Navigation. Our software capabilities include specific enrichment tools powered by AI and big data algorithms, such as Elements Rigid and Elastic Image Fusion, Elements Smartbrush, and intraoperative planning for complex and degenerative spinal cases.”
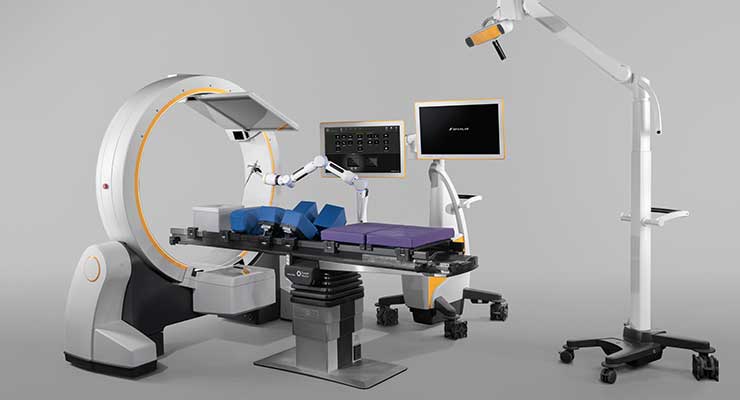
The company’s Loop-X mobile intraoperative imaging robot was unveiled at last September’s North American Spine Society (NASS) meeting. It automates imaging workflow steps and moves robotically with the procedure to be in sync with other robotic arms, the surgeon, and staff. Non-isocentric movement means Loop-X can image much larger and smaller structures than what’s possible with a typical CT or 3D C-arm. It can be integrated with Brainlab or third-party technology through an open interface.
“Smart laser projections remove the intraoperative imaging ‘fiddle factor,’” said Khan. “By defining the ‘level of interest’ on a 2D image, Loop-X robotically supports localization and projects both the incision start and end points directly onto the patient’s skin. The detector and imaging source move independently, enabling non-isocentric imaging. Patients don’t need to be positioned in the center of the gantry since the system moves the scan area to the region of interest. Screw placement verification is a critical yet often time-consuming task in surgery; instrument-guided imaging allows Loop-X to robotically follow the pointer or a preplanned screw and position itself for AP, lateral, or even oblique 2D verification scans.”
Brainlab has partnered with Johnson & Johnson and global orthopedic device maker DePuy Synthes for almost 20 years to jointly develop DePuy Synthes navigated instruments and Brainlab navigation applications. The firms hope to advance 3D navigation through integrated instrumentation and software for open and minimally invasive (MIS) spine surgery.
“Together with DePuy Synthes, we’ve launched a powerful new professional education and training mobile lab initiative that will roll out nationwide this year. ‘Experience the Future of Digital Surgery with DePuy Synthes and Brainlab,’ offers hands-on training to healthcare professionals on the comprehensive and synergistic Brainlab and DePuy Synthes spine portfolios. The Enabling Technology Mobile Lab will train on the full procedural solution for complex cervical, MIS TLIF, lateral, and deformity procedures. Clinicians can attend the mobile lab training to learn about implant solutions, pre-operative planning, visualization, navigation, and robotics. Full surgical teams can train and review process flow together, allowing them to be more efficient and productive.”
The Nuts and Bolts
Dozens of sensors are necessary for a surgical robot to function and respond to its environment. These include position and torque sensors for articulations, gyroscopes and accelerometers for positioning and moving parts, pressure sensors, and image sensors. They can generally be divided into two groups: enabler sensors at the human-machine interface, and sensors not specifically developed for medical robotics applications. Medical robotics is opening a new wave of opportunities for sensor makers. Haptics (force sensing) is also a highly sought technical feature requested by physicians in surgical robotics. Surgeons want technology to “feel” the body’s tissue remotely using haptic sensing, as well as better image quality. Both can be addressed using haptic sensors and high-resolution CCD or CMOS cameras.
“Optical slotted switches and encoders, hollow shaft sensors, and precision potentiometers are necessary to accurately position and track movement for the precise outcome so crucial in surgical robotic equipment,” said Shahab Shahbaz, business development manager at TT Electronics, a Woking, U.K.-based provider of engineered electronics for performance critical applications.
Companies entering the surgical robotic market must ensure their systems are designed to very high quality and safety standards. This means those working on the project must understand the importance of each component in the system, particularly power sources, supplies and components. For example, hospital environments have increasingly complex electrical requirements, and AC power alone isn’t always reliable, designing in a back-up power source early in the process is necessary.
“We also provide the power sources that run these instruments via discretely implemented power supplies or components—transformers, inductors, EMI filters, resistors, and connectors. Our IoT division can integrate connectivity into these systems, meeting the medical community’s increasing demand for remote operation and monitoring.”
A manufacturing partner with experience in these areas can help OEMs in the surgical robotic field meet the high standards necessary for a safe, effective, and reliable surgical robot.
“We can customize each component, sub-assembly, or sub-system to meet any surgical robotic application’s requirements,” said Shahbaz. “This assures the precision necessary for tracking and positioning, torque, speed, weight, and size. Our expert engineers can also identify potential failure modes and address them with the latest products and solutions—improving uptime and safeguarding smooth equipment operation.”
Precision motor and drive systems are equally as important for a successful surgical robot. Robotic devices need increasingly greater types of motors for different applications. For example, over a dozen high-performance brushless motors might be used in a single surgical robot. Stepper, brush and brushless DC motors, gearmotors, mechanical actuators, and drives are all key components for this type of automated equipment. Drive components for surgical robotic applications have extremely demanding requirements. Precision, sterilizability, smooth running, long service life, and low heat buildup in DC and EC drives are essential.
“In dedicated areas like the brain, the drive’s precision is an essential component for performing a successful surgery,” said Carsten Horn, applications engineering manager at maxon , a Taunton, Mass.-based developer and builder of high precision electric drive systems. “For in-body robots, small and compact size are the key factors driving the success while maintaining enough space for surgeons to work effectively. The smaller the drives, actuators, and surgical insertion, the faster the patient’s recovery. These successes are based on having the necessary robotic surgical power and reliability during the procedure.”
To date, a number of different actuators have been used as the main means of surgical robot motion and force transmission, like cable system (tendon, wire), fluidic actuators, and magnetic actuators. Smart materials like shape memory alloy and piezoelectricity are also preferred.
“Typical laparoscopic surgical tool actuators are limited to four degrees of freedom,” said Horn. “The goal is to increase the degrees of freedom to seven to allow for new and more complex surgeries A laparoscopic surgical robot has at least two arms, while a support robot typically only has one arm. Laparoscopic robots contain remote-controlled actuators and vision systems (tele-robotics) and robotic support systems that help position the patient or support positioning tools in place. The number of actuators, the small size, and the OR’s sterile requirements support using small, powerful, and very precise electrical driven actuators like those we produce.”
“Other key factors for successful robotic surgical procedures include noise level, weight, and accuracy,” Horn went on. “Miniature driven laparoscopic actuators may address this using direct-, wire-, or mechanical transfer (chains, pusher, leverages, etc.)-driven drivetrain power density and controllability.”
Surgical robotics is advancing from externally actuated systems to miniature in-vivo robotics. However, this miniaturization provokes a tradeoff between the size of the robot and its capability. Slow actuation, low load capacity, sterilization difficulties, leaking electricity and transferring produced heat to tissues, and high cost are among the key limitations for electric motors in in- vivo applications. Manufacturers are attempting to respond to this with even smaller, more powerful drive components.
“We see the market moving toward mobile single insertion, or even in-body robots. This is driving us to design even smaller drive components with a power density closer to the physical limits.”
Firms that specialize in manufacturing sensing, motion control, or the myriad other surgical robotic components—along with other relevant value-added services—can be essential partners for device makers looking to enter this field.
“We fully embrace outsourced design and development for our single-port robotic technology,” said McNally. “Our outsourcing strategy is an asset—even a competitive advantage—as it is key to accessing deep subject matter expertise within the broad array of technologies required to produce a complex robotic surgical system. Our development-stage, state-of the-art, robotic surgical system encompasses customized precision electro-mechanical systems, extensive kinematic software, 3D HD camera technology, highly dexterous instruments, and modular industrial design for ease-of-use and transportability in and among ORs.”
*pending commercial availability
Dr. Frederick Moll, a surgeon, had non-military applications in mind when he first saw the surgical robotic system in 1994. He became acquainted with venture capitalist John Freund a year later. Freund was able to negotiate a technology license for SRI’s robotic surgical system. Together with engineer Robert Younge, Dr. Moll and Freund began their enterprise to improve minimally invasive surgery—Intuitive Surgical.
The now renowned da Vinci surgical system was commercialized in 1999 in Europe, and a year later came to the U.S. In June 2000, Intuitive went public with a $46 million initial public offering. The company was immediately sued for patent infringement in August 2000 by close competitor Computer Motion, an early entrant to the market with its ZEUS robotic surgical system. (Intuitive acquired Computer Motion in 2003.)
da Vinci’s first FDA approval was for general laparoscopic surgery to treat gallbladder and gastroesophageal diseases. Over the years, the FDA has approved da Vinci and its iterations for prostate surgery, thorascopic surgery, certain cardiac procedures, and gynecologic procedures.
In May 2018, da Vinci took a major step thanks to FDA clearance for the da Vinci SP (single port) surgical system for urologic procedures. Its ability to enter the body through a single, small incision aids surgeons in performing more complex procedures. Intuitive plans to pursue further regulatory clearances for transoral, transanal, and extraperitoneal procedures.
“Our da Vinci SP compliments da Vinci X and Xi systems by enabling surgeons to access narrow workspaces while maintaining high quality vision, precision, and control that surgeons have come to trust from da Vinci systems,” Intuitive CEO Gary Guthart told the press.
The system features three, multi-jointed, wristed instruments and a fully wristed 3D HD camera. The instruments and the camera emerge through a single cannula and are properly triangulated around target anatomy to avoid external instrument collisions. Surgeons control the fully articulating instruments and camera on the same surgeon console as the da Vinci X and Xi systems.
The company’s second product franchise, the Ion flexible robotic catheter, gained an FDA nod last February for minimally invasive peripheral lung biopsy. But Intuitive wasn’t first to market on this occasion. Auris Health, a company cofounded by Dr. Moll after he left Intuitive in 2003, secured FDA clearance for a device to perform lung biopsies. (Johnson & Johnson went on to buy Auris Health for $3.4 billion last April.)
After nearly two decades holding a near monopoly on the surgical robotics market, new competitors are emerging for Intuitive. Morrisville, N.C.-based TransEnterix Surgical Inc. won FDA approval for its Senhance Surgical Robotic System in 2017. At the time, it was the first new entrant into the abdominal surgical robotic field since 2000. The Senhance augments laparoscopic surgical instrument and endoscopic camera precision, visualization, and control. It utilizes an open architecture so hospitals and surgeons can leverage existing technology in the OR with the Senhance. The FDA cleared 3mm instruments for the Senhance in 2018, which according to president and CEO Anthony Fernando, made it the first digital system to use instruments that small on a robotic interface.
“Taken together, the Senhance Surgical System improves laparoscopic surgery by allowing for maximum precision of instruments and surgeon control of their camera and tools,” said Fernando. “It minimizes invasiveness by offering the smallest instruments and also minimizing forces generated during surgery. It allows for surgeons to operate in long, complex cases with minimal fatigue when seated at a console. The goal is to reduce variability in laparoscopic procedures with digital assistance at a reasonable cost, thanks to reusable instrumentation.”
According to TransEnterix, the Senhance is the first digital surgery system with haptic feedback to measure and limit forces generated on the instruments or the patient’s abdominal wall. It also boosts ergonomics by allowing surgeons to sit comfortably in an ergonomic position during long, complex surgeries. Surgeons have long been approaching the boundaries of minimally invasive procedures using handheld, manual instruments and cameras.
“We seek to reduce the physical and cognitive load on surgeons through application of digital and robotic assistance,” said Fernando. “The first area is ergonomics. By allowing a surgeon to sit upright and always keep their hands in a comfortable position, the system keeps a surgeon in an ergonomic position while performing complex surgery. The system filters tremor, stabilizes instruments, and scales motion to improve control in small, precise motions. They can easily control the endoscopic camera by having the system track their eyes to not interrupt the flow of surgery. The new augmented intelligence system moves the camera with a new level of ease by tracking instruments to allow focus on the surgical task with optimized camera movement.”
The company obtained FDA clearance for its Intelligent Surgical Unit (ISU) to enable machine vision capabilities on the Senhance this past March. Machine vision drives surgeon camera control by responding to commands and recognizing certain objects and locations in the surgical field. The ISU hardware will be compatible with planned future augmented intelligence features like scene cognition and surgical image analytics. The ISU is also compatible with third-party vision systems the Senhance currently supports.
“At its core, the ISU lets a computer see, process, and augment surgical images,” said Fernando. “The set of features enabled by the initial approval are related to camera control augmented by machine vision. Now a surgeon can have the system recognize various instruments in the field and track the camera to their movement. The system can quickly zoom in and out on a particular point, and can move the camera to a desired location with one click. When using an off-angle scope, zooming in and out will maintain the image horizon with a constant image center throughout the zooming process, eliminating the need to pan the camera in addition to zooming. These features combine with eye-tracking to make Senhance a sophisticated and full-featured camera control mechanism for digital surgery.”
Since its launch, the Senhance’s indications have been expanded to include laparoscopic inguinal hernia and laparoscopic cholecystectomy (gallbladder removal) surgery in the U.S., and the ability to treat pediatric patients in Europe. The Senhance Ultrasonic System gained FDA clearance last January as well, enabling use of instruments that deliver controlled energy to ligate and divide tissue and minimize thermal injury to surrounding structures.
Multi-port robotic surgery requiring multiple incisions has been practiced for about 20 years. It’s becoming the new care standard in several surgical disciplines. Unlike traditional laparoscopic surgeries, today’s robotic technologies provide surgical precision, tremor-free dexterity, 3D HD visualization and natural user interface, while allowing more comfortable operation than manual laparoscopic surgery.
Toronto, Ontario-based Titan Medical’s single-port robotic surgery system is still under development. Last year, the company successfully completed two critical tasks for the system’s FDA IDE filing: acute and chronic procedures performed under Good Laboratory Practice principles and a human factors evaluation.
“Similar to traditional laparoscopic surgeries, most of today’s robotic surgeries still require at least three and as many as six different incisions,” said David McNally, president and CEO of Titan Medical. “Our single-port robotic system was designed with surgeon input from the very beginning, improves upon existing multi-port systems, and offers minimally invasive surgical options through only a single incision while maintaining the primary surgeon benefits afforded by robotic technology. By enabling surgeons to minimize the number of incisions, our robot potentially offers reduced trauma, faster recovery times, and minimal scarring.”
The Titan Medical single-port surgery system’s multi-articulated instruments have single-use, replaceable tips for grasping, suturing, cutting, and coagulation. Its workstation (which is maneuvered on swiveling coasters) features a flatscreen, 3D high definition display, a multi-articulated handle interface, and configurable elbow rest and foot pedal positioning. It has a single-arm mobile patient cart and 25mm-diameter camera insertion tube. The single-arm configuration has no external moving parts to ease simple setup and assistant-friendly procedures.
Ultimately, Titan Medical hopes to expand robotic surgery into currently underserved areas like general abdominal, gynecologic, urologic, and colorectal.
“Our three primary goals for this year are to secure adequate financing to resume product development in coordination with our outsourcing partners; execute planned improvements to the instruments, camera systems, and accessories based on additional surgeon input received during our GLP studies; and perform software development, verification, and validation in preparation for our planned IDE studies.”
Munich, Germany-based Brainlab creates and integrates software and systems for various surgical procedure stages: pre-planning, surgical navigation, data management and manipulation, and intraoperative imaging and surgical follow-up. Surgical navigation—also known as image-guided surgery, computer-assisted surgery, or software-guided surgery—continuously tracks instrumentation in relation to patient anatomy and displays the relationship in real-time of a screen during surgery.
“Turning time-consuming human interaction into automatic digital processes is the best way to decrease surgical time and increase surgical productivity,” said Meraj Khan, director of Brainlab’s neurosurgery portfolio. “After integrating thousands of ORs across the globe for the last twenty years, we’ve identified four key ways which hospitals should digitalize processes to enhance efficiency: consolidating data influx into a single control platform, streamlining documentation flow, improving communication in and outside the OR, and facilitating planning from anywhere.”
“As we focus on the operating room setting in the coming year, our technology continues to excite users,” Khan continued. “We bring value in clinical flexibility by integrating all types of pre-operative and intra-operative imaging modalities. Brainlab has a diverse automatic image registration option to connect an intraoperative imaging device whether it is a 2D C-arm, a 3D C-arm/cone beam CT (such as the Medtronic O-Arm, Siemens CIOS Spin, or Ziehm RFD3D), true intraoperative CT (such as Airo), or our newly launched Loop-X* Mobile Imaging Robot.”
The company’s suite of spine surgery products helps to effectively plan and execute spine procedures and place pedicle screws more accurately while minimizing the surgical team and patient’s X-ray exposure. Brainlab Spinal Navigation can address tumors and deformities, as well as routing cervical, thoracic, or lumbar spine surgeries. Instrument-tracking software helps avoid critical structures. Image registration and compatible imaging device options are broad so the procedure and imaging devices can be tailored to the surgeon’s needs.
“Brainlab spine surgery planning software has been designed for automated functionalities enabling effective and straightforward treatment planning. The Brainlab Elements 3D Viewer, for example, allows fast, easy access to medical image data, quick and intuitive interaction with DICOM images, and direct transfer to Brainlab Spine Navigation. Our software capabilities include specific enrichment tools powered by AI and big data algorithms, such as Elements Rigid and Elastic Image Fusion, Elements Smartbrush, and intraoperative planning for complex and degenerative spinal cases.”
The company’s Loop-X mobile intraoperative imaging robot was unveiled at last September’s North American Spine Society (NASS) meeting. It automates imaging workflow steps and moves robotically with the procedure to be in sync with other robotic arms, the surgeon, and staff. Non-isocentric movement means Loop-X can image much larger and smaller structures than what’s possible with a typical CT or 3D C-arm. It can be integrated with Brainlab or third-party technology through an open interface.
“Smart laser projections remove the intraoperative imaging ‘fiddle factor,’” said Khan. “By defining the ‘level of interest’ on a 2D image, Loop-X robotically supports localization and projects both the incision start and end points directly onto the patient’s skin. The detector and imaging source move independently, enabling non-isocentric imaging. Patients don’t need to be positioned in the center of the gantry since the system moves the scan area to the region of interest. Screw placement verification is a critical yet often time-consuming task in surgery; instrument-guided imaging allows Loop-X to robotically follow the pointer or a preplanned screw and position itself for AP, lateral, or even oblique 2D verification scans.”
Brainlab has partnered with Johnson & Johnson and global orthopedic device maker DePuy Synthes for almost 20 years to jointly develop DePuy Synthes navigated instruments and Brainlab navigation applications. The firms hope to advance 3D navigation through integrated instrumentation and software for open and minimally invasive (MIS) spine surgery.
“Together with DePuy Synthes, we’ve launched a powerful new professional education and training mobile lab initiative that will roll out nationwide this year. ‘Experience the Future of Digital Surgery with DePuy Synthes and Brainlab,’ offers hands-on training to healthcare professionals on the comprehensive and synergistic Brainlab and DePuy Synthes spine portfolios. The Enabling Technology Mobile Lab will train on the full procedural solution for complex cervical, MIS TLIF, lateral, and deformity procedures. Clinicians can attend the mobile lab training to learn about implant solutions, pre-operative planning, visualization, navigation, and robotics. Full surgical teams can train and review process flow together, allowing them to be more efficient and productive.”
The Nuts and Bolts
Dozens of sensors are necessary for a surgical robot to function and respond to its environment. These include position and torque sensors for articulations, gyroscopes and accelerometers for positioning and moving parts, pressure sensors, and image sensors. They can generally be divided into two groups: enabler sensors at the human-machine interface, and sensors not specifically developed for medical robotics applications. Medical robotics is opening a new wave of opportunities for sensor makers. Haptics (force sensing) is also a highly sought technical feature requested by physicians in surgical robotics. Surgeons want technology to “feel” the body’s tissue remotely using haptic sensing, as well as better image quality. Both can be addressed using haptic sensors and high-resolution CCD or CMOS cameras.
“Optical slotted switches and encoders, hollow shaft sensors, and precision potentiometers are necessary to accurately position and track movement for the precise outcome so crucial in surgical robotic equipment,” said Shahab Shahbaz, business development manager at TT Electronics, a Woking, U.K.-based provider of engineered electronics for performance critical applications.
Companies entering the surgical robotic market must ensure their systems are designed to very high quality and safety standards. This means those working on the project must understand the importance of each component in the system, particularly power sources, supplies and components. For example, hospital environments have increasingly complex electrical requirements, and AC power alone isn’t always reliable, designing in a back-up power source early in the process is necessary.
“We also provide the power sources that run these instruments via discretely implemented power supplies or components—transformers, inductors, EMI filters, resistors, and connectors. Our IoT division can integrate connectivity into these systems, meeting the medical community’s increasing demand for remote operation and monitoring.”
A manufacturing partner with experience in these areas can help OEMs in the surgical robotic field meet the high standards necessary for a safe, effective, and reliable surgical robot.
“We can customize each component, sub-assembly, or sub-system to meet any surgical robotic application’s requirements,” said Shahbaz. “This assures the precision necessary for tracking and positioning, torque, speed, weight, and size. Our expert engineers can also identify potential failure modes and address them with the latest products and solutions—improving uptime and safeguarding smooth equipment operation.”
Precision motor and drive systems are equally as important for a successful surgical robot. Robotic devices need increasingly greater types of motors for different applications. For example, over a dozen high-performance brushless motors might be used in a single surgical robot. Stepper, brush and brushless DC motors, gearmotors, mechanical actuators, and drives are all key components for this type of automated equipment. Drive components for surgical robotic applications have extremely demanding requirements. Precision, sterilizability, smooth running, long service life, and low heat buildup in DC and EC drives are essential.
“In dedicated areas like the brain, the drive’s precision is an essential component for performing a successful surgery,” said Carsten Horn, applications engineering manager at maxon , a Taunton, Mass.-based developer and builder of high precision electric drive systems. “For in-body robots, small and compact size are the key factors driving the success while maintaining enough space for surgeons to work effectively. The smaller the drives, actuators, and surgical insertion, the faster the patient’s recovery. These successes are based on having the necessary robotic surgical power and reliability during the procedure.”
To date, a number of different actuators have been used as the main means of surgical robot motion and force transmission, like cable system (tendon, wire), fluidic actuators, and magnetic actuators. Smart materials like shape memory alloy and piezoelectricity are also preferred.
“Typical laparoscopic surgical tool actuators are limited to four degrees of freedom,” said Horn. “The goal is to increase the degrees of freedom to seven to allow for new and more complex surgeries A laparoscopic surgical robot has at least two arms, while a support robot typically only has one arm. Laparoscopic robots contain remote-controlled actuators and vision systems (tele-robotics) and robotic support systems that help position the patient or support positioning tools in place. The number of actuators, the small size, and the OR’s sterile requirements support using small, powerful, and very precise electrical driven actuators like those we produce.”
“Other key factors for successful robotic surgical procedures include noise level, weight, and accuracy,” Horn went on. “Miniature driven laparoscopic actuators may address this using direct-, wire-, or mechanical transfer (chains, pusher, leverages, etc.)-driven drivetrain power density and controllability.”
Surgical robotics is advancing from externally actuated systems to miniature in-vivo robotics. However, this miniaturization provokes a tradeoff between the size of the robot and its capability. Slow actuation, low load capacity, sterilization difficulties, leaking electricity and transferring produced heat to tissues, and high cost are among the key limitations for electric motors in in- vivo applications. Manufacturers are attempting to respond to this with even smaller, more powerful drive components.
“We see the market moving toward mobile single insertion, or even in-body robots. This is driving us to design even smaller drive components with a power density closer to the physical limits.”
Firms that specialize in manufacturing sensing, motion control, or the myriad other surgical robotic components—along with other relevant value-added services—can be essential partners for device makers looking to enter this field.
“We fully embrace outsourced design and development for our single-port robotic technology,” said McNally. “Our outsourcing strategy is an asset—even a competitive advantage—as it is key to accessing deep subject matter expertise within the broad array of technologies required to produce a complex robotic surgical system. Our development-stage, state-of the-art, robotic surgical system encompasses customized precision electro-mechanical systems, extensive kinematic software, 3D HD camera technology, highly dexterous instruments, and modular industrial design for ease-of-use and transportability in and among ORs.”
*pending commercial availability