Michael Barbella, Managing Editor09.09.19
If only the human body wasn’t so...well, human.
Imagine, for a moment, the possibilities: Organs and joints that never fail, bones that never break, neurons that never misfire, muscles that never tear, and cells that never damage. Invincibility at its very best.
Mankind is well on its way to achieving such immortality with science-fiction-inspired technological advancements like 3D-printed tissue, artificial hearts, smartphone-controlled arms, spray-on skin, exoskeletons, and brain-computer interface devices.
The latter innovation, supported by a growing cache of scientific research, fulfills humankind’s decades-long dream to harness cerebral electricity to directly control machinery and/or non-working body parts. Though still in its early stages, the technology has already been used to help a paralyzed teenager kick a soccer ball, and a quadriplegic man regain control of his hand, wrist, and fingers. “It’s certainly a huge challenge to restore movement to a paralyzed limb,” quadriplegic Ian Burkhart acknowledges in a video posted to the Battelle Memorial Institute website. The 28-year-old Ohio resident was paralyzed in a 2010 diving accident.
“The biggest thing is it has to be speculative because that’s how you make the big leaps forward,” Burkhart says in the Battelle video. “It has to be seen as something that can’t be done. Yes, there might be something that’s way too far-fetched and isn’t realistic, but if you put the right team together...I think it’s something that can be done. Take those wild ideas so that way you can push science forward.”
Burkhart has helped push science forward by participating in a five-year study of Battelle’s BCI technology (NeuroLife). Conducted in collaboration with Ohio State University Wexner Medical Center doctors, the analysis proved it possible for paralyzed patients to move their hands and fingers via mere thought.
Such mental prowess (and promise) is substantiated by various other BCI devices in development: Carnegie Mellon and University of Minnesota researchers, for example, have built a mind-controlled robotic arm capable of continuously tracking and following a computer cursor, while BrainGate Consortium’s brain-computer interface technology has proven its worth with tetraplegic study participants.
BrainGate’s investigational device features an internal neural signal sensor that detects the signs presaging intended movements in the brain’s cortex. Those signals are then decoded and routed to an external device. The implantable sensor houses a baby aspirin-sized chip with 100 electrode sensors—each thinner than a human hair—that can detect the brain’s electrical activity.
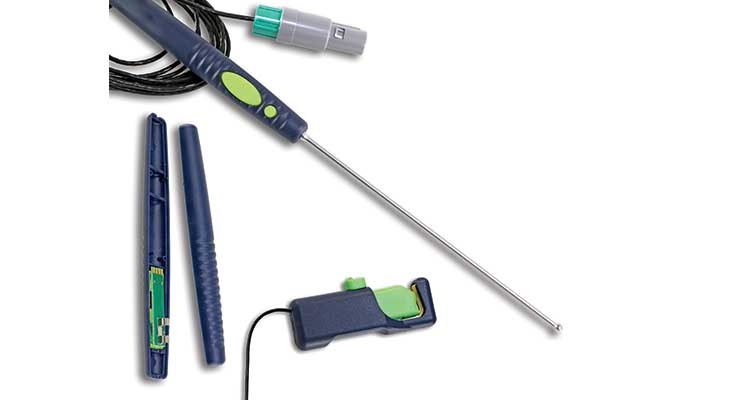
Developing electronic components small enough for human implantation requires a proficiency in miniaturization, custom design, enhanced functionality, and material selection, among others. It also demands tremendous flexibility, as the Internet of Things, big data, and analytics continue to pervade the $400 billion global medtech market.
To better understand the factors and trends influencing the custom medical electronics market (both now and in the future), Medical Product Outsourcing spoke with numerous industry professionals over the last few weeks. Input came from:
Farshid Allahakbari, head of the electrical engineering team at Irvine, Calif.-based FUTEK Advanced Sensor Technology Inc., a manufacturer of load cells, torque sensors, pressure sensors, multi-axis sensors, and related instruments/software.
Drew Bratton, sales manager at Plastics One Inc., a Roanoke, Va.-headquartered medical contract manufacturer.
Brian Burke, sales manager at BAYCABLE, a custom cable and cable assembly solutions provider located in Fremont, Calif.
David Cianciolo and Patrick Kinyanjui, engineering director and principal engineer, respectively, at Fischer Connectors, a global manufacturer of high-performance push-pull circular connectors and cable assemblies.
Christian Fritz, sales director, Motion Control & Electronics, at maxon group, a Swiss manufacturer and supplier of high-precision drive systems.
Bala Kashi, business development manager, Medical Sensors, at TE Connectivity Ltd., a global manufacturer of connectivity and sensor solutions for various industries, including energy, medical technology, data communications, transportation, industrial applications, and the home.
Mike Labbe, R&D director at Valtronic, an electronics manufacturing services developer of highly complex printed circuit boards, hybrid circuits, and complete medical devices.
Michael Barbella: What factors must be taken into consideration when designing custom electronic components for medical devices?
Drew Bratton: When designing custom electronics components, there are many attributes to focus on. To list some of the inputs to our design and development process; functional, electrical, software, environmental performances, physical characteristics, user and patient safety considerations, and target cost. Plastics One has invested in developing an integrated system to guide our development of complete custom electrode following ISO 13485 guidelines to ensure no inputs are not reviewed.
Brian Burke: Are the devices going in the body? Will autoclave be needed? (Multi-use cleaning procedures). Can the market bear another player in the chosen segment? Is the idea manufacturable and repeatable in production? Are the customer’s engineers open to our knowledge and know-how?
David Cianciolo: When dealing with a connectivity solution in medical devices, you have to know what Medical Device Class of the equipment is. That will determine the risk level and the degree of validation required. Higher Device Classes will require more scrutiny, testing, and higher validation quantities
Patrick Kinyanjui: Device Class 1 or 2 materials may not be acceptable for a Class 3 device. Most connectors do not normally meet the requirements for implantable applications. Class 3 devices also require a higher level of traceability down to the materials at every level of components.
Christian Fritz: While the medical device industry is subject to similar market pressures as other industries, there are a couple of unique challenges that impact the design of custom electronics and the development of low-level firmware and application specific software for medical applications. Innovation, tight time-to-market requirements, and the need to provide a cost-effective solution are common factors to consider when designing custom electronics. In the medical device industry, it is very important to consider the type of customer the device is targeting. The requirements are vastly different if the device is designed for a service provider (hospital, laboratories) or the end user (patient). On top of those economic factors the medical industry requires an increased level of certifiable safety and reliability, which has a major impact on the design. Regulatory requirements need to be factored in from the very early stages of prototyping throughout the life-cycle of the hardware and software design.
Bala Kashi: When designing custom electronic components for medical devices, there are many factors that need to be considered. Not only do factors such as understanding the application, validations, parameters, material selection, standards to be met, cleanliness, and cost targets need to be acknowledged but there are other considerations. Some of these other considerations include the ruggedness, size, accuracy, and scalability of the sensors since medical components can be exposed to various harsh conditions and media. For example, if the sensor is being used for an implant application, it is required to be more critical for biocompatibility since it will have long-term contact with body parts. Additionally, the sensor will need to function long-term with almost no probability of fail.
Mike Labbe: There are many specific factors to consider in designing medical devices. First are the appropriate regulations that must be met for that product to get approved for market. There are many FDA and ISO standards related to electrical safety, EMC compliance, human factors and usability, risk management, HIPAA, and cybersecurity concerns that must be addressed in the design process if the product is to be cleared for distribution, and these must be incorporated into the design process from the very beginning.
Time to market for a medical device can be much longer than consumer products. Often there are regulatory clearances, agency testing, and clinical trials that must be completed, which can take five years or more. With this in mind, selection of components in the design phase is critical, otherwise components selected in the initial design will be obsolete before the product ever gets to production. Fast-changing technologies such as displays, batteries, memory, and other electronics used in mass consumer devices are especially important to focus on in the selection process to find parts that will have an adequate life to support product launch.
A third critical factor to consider in customer electronic medical devices is Design for Manufacturability (DFM). Understandably, the focus during product development is often on functionality and clinical effectivity. However, to have a successful product, the end result must be a design that can be manufactured in a consistent manner, at an appropriate cost and with high quality. Focus on getting DFM input during the design and prototyping phase from an experienced manufacturing team in the processes required to build the product is essential.
Barbella: Please discuss some of the challenges in designing and manufacturing custom electronic components for medical devices. How has your company overcome these challenges?
Farshid Allahakbari: To meet the requirements of any custom medical projects, a designer usually faces several obstacles. Typically, we classify the anticipated constraints into two disciplines: design vs. manufacturing. Design phase challenges are reliant on experience and knowledge of the designers. Medical applications typically require a miniature board with limitations. These constraints vary and consist of the following:
Manufacturing challenges that are usually overcome by:
Bratton: Many of the challenges we face in designing and manufacturing custom electronic components related to the seamless integration of multiple manufacturers and processes. Projects we are working on now require much more than just a molded solution, rather various levels of manufacturing, packaging, and sterilization. Our company has overcome these challenges by qualifying a diverse group of vendors and providing turnkey manufacturing solutions (dock to stock).
Burke: Some of the [previously mentioned] thoughts.
Engineering staffs and resource allocation both at the customer and for us as well. Everything is getting smaller and smaller and miniaturization presents challenges trying to extrude insulation and mold parts that are smaller and have tighter tolerances. We listen to what the customer needs and see if it fits in our capabilities and discern if they are a viable potential customer and partner. We then sit with our engineering gang and see if we can meet these requirements in the timeline required. Often we need to do some experimental runs to test the range of capability for newer custom parts that are outside of our current capability. We can run various iterations and work with our customers throughout this process of collaborative design.
Cianciolo: Due to sterilization, requirements sealing of a connector present a bigger challenge in the medical industry. When working on a new medical cable, we always inquire about the end-user’s sterilization methods. We may need to use higher temperature materials such as silicone, FEP, PPSU, etc., for the construction of the assembly.
We also look at isolation characteristics; Medical Device Classification Type B, BF, or CF, that are unique to each medical device. Type BF and CF require extra tests that Type B doesn’t require.
We ask many questions to make sure we know exactly how and where the equipment is being used and what the expected sterilization protocol is. That initial interview is needed so design time is reduced.
Fritz: Over the last couple of years, the medical device industry has been facing a changing landscape of regulatory requirements. This put strain on the design teams who had to build up knowledge and refine existing processes while executing a growing number of projects. To increase their engineering efficiency, many medical device companies turned to third parties and components vendors for some of the vertical expertise needed to complete their devices. As a response, maxon, a key provider of small high-precision motors and motor control electronics, has increased its service offering and is collaborating closely with medical device builders for their electronics needs. Leveraging the existing building blocks of their off-the-shelf motor controllers, maxon can quickly provide a customized solution and help with the documentation needed for medical certifications.
Kashi: The design and manufacture of medical electronic components, including sensors, involves many challenges. Material selection, validation tests, quality systems, scaling, and custom parametric tests are all examples of some of the challenges faced during this process. Since sterilization is a high priority within the medical industry, an additional challenge is the requirements regarding how components are to be manufactured—meaning, the space that they are developed within needs to follow specific guidelines to ensure a certain level of cleanliness required by the medical industry.
Labbe: As mentioned, understanding the expected life cycle of components selected in the design phase is critical to assure consistent supply. Valtronic works closely with the suppliers of these components to choose options that have an assured product life through the first several years of manufacture. Wherever possible, alternate components are also included on the approved BOM to allow for supply chain flexibility. Additionally, Valtronic continuously monitors lead times and orders components in advance to avoid manufacturing disruption.
Design for Manufacturing is also critical in medical device design. Once a product has been designed, prototyped, tested, and cleared through regulatory agencies, changes to the design and manufacturing can be time consuming and expensive. It is critical that products built once the device is in full series production meet all of the quality, performance, and safety specifications of the initial units used in product verification and clinical validation studies. To achieve this, input from experts in the processes required for production should be included in the design cycle from the beginning.
Barbella: What are customers demanding or expecting in their custom electronic components?
Allahakbari: Customers mostly dictate the size, performance, and cost of the components. Typically, the first size and performance conflict is with cost. A direct dialogue and transparency with the customer is essential to understand what requirements are crucial. A straightforward custom design could turn to a very complicated design if requirements aren’t received up front.
Bratton: Customers are now demanding more complex solutions than ever before. With new technology and software developments, the components have had to shift in culture to support the vast array of modern technology. Many years ago, cables and connectors were very simplistic and just a means to connect items. Many of our clients are now looking to combine multiple cables, electrodes, and circuitry all into one complete system, whereas in the past it may have been various items working in tandem.
Burke: Regulatory help and sourcing expertise. Custom manufacturing requirements to curb costs and lead times.
Cianciolo: Customers are also looking for connectivity solutions that integrate sensors to count the number of procedures, or total time used, keeping patients safer. Design engineers are definitely reimagining connectivity solutions to gather data and make the devices smarter and safer.
Kinyanjui: Our customers are looking for smarter products that include electronic identification. This gives the customer the ability to offer a system that will preset the device settings for its unique procedure. Others go further and demand that the connectivity solution identifies the exact device. This increasing demand for traceability means that we work with our customers to design in ID technology. This can be done by using a specific sub-component integrated into the cable assembly.
Fritz: While the design needs to meet the technical requirements and provide the performance that the medical device requires, it is important to provide enough flexibility to allow for adjustments that might become necessary during prototyping stages or clinical trials. At the same time, it is important to consider the certification needs early on. Providers of custom electronics need to apply documented development processes and comprehensive test documentation that aids a smooth certification process.
Kashi: The medical industry requires electronic component options for many different medical applications. However, there are some demands from customers that remain consistent since some sensing components measure various applications that relate to human life. For example, the accuracy and functionality of medical electronic components for life support devices is critical for that application. In addition, medical sensing components need to be reliable and meet all medical standards.
Labbe: There are a couple of shifts Valtronic has been observing in the medical electronics CM industry in the last few years. First, many more customers are becoming more educated in the SMT and manufacturing process technologies, and are demanding these processes be used on their products. Automated Solder Paste Inspection (SPI), closed-loop Automated Optical Inspection (AOI), Artificial Intelligence systems, and other advanced technologies are expected by more educated customers.
Secondly, more customers are requesting full turn-key services. Customers are outsourcing more aspects of design and DFM review before transferring to contract manufacturers. Additionally, there is more demand to perform full turnkey manufacturing, from supply chain management, sub-assembly manufacture to full device assembly, test, packaging, and even distribution. Providing more value-added services has become more critical for partnerships with OEM customers in today’s marketplace.
Barbella: How is IoT (Internet of Things) influencing custom electronic component development?
Bratton: The most significant shift we have seen in the past five to 10 years is the integration of home healthcare and data collection. Many products are shifting to allow them to be used through small home devices such as a smartphone or a tablet. Diagnostic or monitoring tests that have historically been conducted in a hospital or clinic are now completed within the comfort of your own home. Many of these devices are connected through the internet, and data can be uploaded to cloud storage for further review and analysis.
Burke: The chip and big connector guys are so in love with that segment, that sometimes it’s hard to get attention on a legacy product—i.e., power and signal standard products.
Fritz: There is an increased interest for leveraging IoT technologies (communication interfaces, security, increased data collection and analytics) that allow medical device builders to broaden their services offering, increase the uptime of their systems, and lower the maintenance cost. This is especially true for devices targeted at medical service providers. When it comes to medical devices, it is important to evaluate these technologies with a strong focus on security and data protection.
Kashi: There is a push toward developing sensing components that are suitable for IoT applications since sensors are the means to gather the data needed for successful deployment of these IoT applications. The data collected by sensors can help medical professionals understand critical situations faster and more accurately and enable patients to be more informed about their conditions and improvements. For example, if the sensor is being used within an in-home care application, it might have a wireless transmitter to send the data remotely to the cloud. Physicians or the patients can have the ability to download the data for analysis. Additionally, the sensor devices are required to be accurate, precise, and reliable to avoid false triggering of any event. From blood pressure sensing to dialysis equipment monitoring, invasive and non-invasive sensors are providing localized data and information to the cloud to better serve patients and the medical industry.
Labbe: IoT is becoming ubiquitous in the industry. This shift requires expanded expertise in RF and wireless technologies, software and data management, as well as protocols for security and encryption. These are rapidly developing and changing technologies, and there are many groups popping up with specific expertise to stay on top of these developments. It is critical to have in-house expertise, or relationships with external experts to stay on top of these technologies.
Barbella: How is big data influencing custom electronic component development?
Allahakbari: In general, big data analysis requires fast processors, sophisticated digital systems, and high-power communication that could make the custom designs complicated. We cannot process big data through traditional software, so companies have implemented cloud-based tools to mine massive amounts of data and deliver the data successfully. A lot of development, automation, and safety features are required to be considered during the data-gathering processes. This makes the custom designs even more complicated for medical applications.
Bratton: Within contract manufacturing, we do not experience the direct influence of big data. We do, however, work with medical device OEM companies which relay many of the results of these studies and data collections to Plastics One for review. How does this benefit a manufacturer? The more useful data that is collected helps manufacturers such as Plastics One make products that will last longer and perform at a higher level within their field of use.
Burke: Data analytics and auction sites cut down on how customer service levels and past performance affect procurement strategies. You can perform spectacularly for years, but if procurement departments go to auction or purely data/prices-based decision-making, all that good will is never baked into the cake.
Kashi: Big data is influencing custom electronic component development in a positive way. The feedback gathered by big data can influence the functionality and the parameters monitored. For example, since the patient data will get stored remotely, it is possible to analyze this big data to understand the predictability prior to the occurrence of an event. The big data analysis can check for effectiveness of the treatment by age, gender, race, location, and patient habits. Therefore, with this information, the treatments can be more customized to specific patients. In the future, there will also be an increasing emphasis on smart sensing, using on-board sensor capabilities to analyze data and transmit vital data rather than simply “big” data.
Labbe: Big data has large impacts on the requirements for software development. Hardware to collect, transmit, and manage big data is fairly well-established. Especially in medical devices, there is an increasing trend to treating patients away from the hospitals. This has led to many devices that collect data from the patient, and upload to the cloud. Manufacturers or external services providers then need to sort through all this data in near real time, and implement algorithms to detect events or critical data and provide this digested information to the physicians and caregivers for action. This requires significant upfront investment in software systems for cybersecurity, algorithm development, and HIPAA compliant processes. Regulatory agencies are also beginning to review and regulate these aspects more critically so a comprehensive strategy to manage and protect this data is required.
Barbella: How is AI (artificial intelligence) influencing custom electronic component development?
Allahakbari: In general, AI aims for the design and implementation of processes that, when run on physical architectures, trigger the maximization of desired results by focusing on productivity enhancement. Some of the lines of work of the research intelligence are: design and prototyping of electronic boards and advanced devices for integration in medical equipment and robot surgeries; ARM solution designs for advanced data processing to support medical applications; and design of intelligent systems for precision agriculture with feedback and self-fault detect features for medical applications.
Bratton: Many of our customers are beginning to integrate “smart” components and circuitry electronics within the cables and connectors we manufacture. Recent integrations we have worked with include applications to limit the number of uses for disposable medical devices and automatic device profile downloads to the medical instruments. These developments help reduce the patient risk preventing the products from being misused in a surgical setting.
Burke: Only the big chip and connector guys have the resources to utilize AI to any degree.
Kinyanjui: Right now, integrating AI into medical devices is a goal, not a reality. The FDA needs to update rules before AI will be fully designed for the medical community. We know it’s coming, and we are looking at it. It’s going to be an interesting ride the next few years as we figure out how AI can ethically and practically be introduced.
Kashi: Similar to the positive impacts of big data, AI allows for predictability before any patient event. Based on patient patterns, AI not only enables the ability to predict a patient event but act on that event. For example, AI can provide notifications to paramedics or the physician if the medical machine is failing or if the patient is showing certain symptoms. Additionally, AI can interact with other devices that are hooked to the patient.
Barbella: Is the trend toward miniaturization of medical devices driving a need for flexible custom electronic components?
Allahakbari: Physical limitations and constraints in medical applications require custom designs to have more advanced and complicated layouts. For example, a rigid/flex design is usually required to provide a smaller package with a more complicated assembly. The connectors and mounting provisions used in medical applications drive the need for flexible low-profile designs. Usually the assembly needs to be folded to fit in the allocated area. This process forces the designer to consider more design rules during the PCB design and makes the manufacturing more expensive. It also forces the designer to be more quality-minded when it comes to selecting fabrication houses.
Bratton: Medical devices are continually undergoing design changes and improvements to reduce size. As size reduces, many performance characteristics must be reviewed and tested before production implementation. Plastics One’s pre-clinical division has been at the forefront of miniaturization, working with university students and doctors on cannulas and electrodes used in drug delivery systems. We have also invested in tool-making technology to help facilitate mold fabrication on a miniature scale.
Burke: Yes, and that entails endless hours of research as the products get smaller, the labor is more specialized, and there is less apt for big ramps. Miniaturization of wire and connectors results in a need for automating the termination process, which requires much research on how to automate, as most applications are extremely specialized.
Kinyanjui: Weight is always a factor in handheld devices, due to the fatigue factor in the OR, as procedures can easily run for hours at a times. Fortunately, new technology allows connectors to be smaller with more dense connections. Size and weight become a bigger factor with wearables. We have a plastic lightweight connector that is specifically designed for wearables. We’ve reduced weight by changing the materials used in manufacturing. We actually see a greater focus on miniaturization and weight in the military since it’s been a focus for so long. We find we are able to apply what we’ve learned in that market to our medical customers.
Fritz: There is clearly a trend towards miniaturization, not only in regard to electronic components but also for mechanical and electromechanical components and subsystems.
As a result, maxon increased their offering regarding product customization and value-add services changing the role they play in the development process. An active collaboration on the development of highly customized components or integrated subsystems allows for compact design, maximum performance and helps address the increasing complexity of medical devices.
Kashi: Wearable and portable devices require smaller, lighter, and less power-hungry devices that allow for more sensing functionality within one device. Devices such as these require more resources to develop and are more expensive to build. Therefore, during the design phase, there is a need to consider multiple other potential applications and try to address them as well.
Labbe: Historically the need for miniaturization of medical devices led to a significant use of bare and stacked die, flip, and wire-bonded assemblies to minimize the component size. Today’s packaged components and ICs are becoming so optimized for size that these more complex processes are required less often. Placement of packaged components down to 01005 size and lower, barely visible to the naked eye, has resulted in increased need for automated monitoring and inspection processes for controlling quality. Bottom terminated assemblies such as BGA and QFN packaging require more sophisticated assembly and reflow profile development and optimization, as well as X-ray and other inspection techniques to monitor processes. In-process and final functional testing at the manufacturing location are more critical than ever to detect and correct issues in real time and provide feedback to the process.
Valtronic is also seeing an increasing use of flexible and rigid-flex assemblies. These approaches allow for using more standard manufacturing techniques in production, and then folding and packing electronics in the final assembly stage to minimize size and space of finished devices. This presents a different set of challenges in the design, manufacture, and supplier selection for these more complex flexible components.
Barbella: How else is miniaturization impacting custom electronic component development?
Allahakbari: Miniaturization usually adds more unknowns to the equation as far as the component selection, the PCB layout, and the manufacturing process. Sometimes, miniaturization limits force the designer to fit the same amount of components that require a large amount of room in a smaller area. The limitations cause additional challenges such as heat dissipation because there isn’t enough surface area to dissipate the heat. In this case, the design would require more attention to the design approach, component selection, power optimization, testing, and validation processes.
Bratton: With a reduction in size, many solutions are becoming tailored to each specific project’s end use versus an off-the-shelf approach. Historically many manufacturers, including Plastics One, have had a line card of the products they can produce. We have seen a shift in this mindset where many solutions require specific and proprietary electronic components.
Burke: Smaller doesn’t always mean cheaper, and there is a need to develop packaging to assure no damage in any of the processes. New test fixtures that mirror the smaller products. Automation and research factors go up exponentially.
Kashi: With the drive toward developing miniaturized components, there have been impacts within the development process. Time and cost associated with development has increased due to the increased resources and the additional testing needed to confirm products work efficiently. There is also the need to fit all the same functionality into a smaller package. For example, shrinking sensor devices will take a lot of engineering. It will be costly since new designs may be required in addition to using expensive material and precise components. There will also be the need for highly accurate manufacturing techniques since the devices will be so small. To ensure performance won’t be compromised, the new devices may be required to go through extensive validation tests, third party certifications, etc.
Barbella: In what ways is the changing regulatory environment impacting custom electronic component development?
Allahakbari: While our component-level sensors and electronics are not subject to the more stringent FDA and European finished medical device regulations, they are subject to an ever-changing materials restrictions and electronic emission/susceptibility landscape of regulations. Many our design choices are governed by the limited availability of materials that must conform to RoHS and REACH Directives that change almost every year, as well as emission and susceptibility testing regulations that are sometimes revised with new acceptance criteria. At best, this mandates retesting and, at worst, a redesign of mature products to ensure continued compliance. As electronic component manufacturers switched over from non-RoHS to RoHS components, there was an interruption in the supply chain when they stopped producing the obsolete parts before the new RoHS components became readily available. We continue to face challenges with the availability of RoHS-compliant components today.
Bratton: To list a few of the significant regulations we follow: RoHS, Reach, Health Canada, Latex Free, and Animal Derived Materials. Each of these regulations is constantly updating standards, requiring manufacturers to review updates every year. The main effect this has on products is with some of the initiatives, certain elements are either reduced or limited in percentage content. As the regulations change, manufacturers are required to revalidate the product if the raw components shift in high proportion.
Burke: The non-revenue generating aspect of compliance is a constant battle every manufacturer and supplier is dealing with. The time spent on compliance must be covered along with engineering, manufacturing, and all other SG&A costs. The big guys factor in these costs and skirt them by advising on standards committees and feel they have done their part, leaving smaller manufacturers to bear the brunt of a lot of those un-recoverable costs.
Cianciolo: There are some recent changes in IEC 60601-1: 4th edition that require reduced radiated emission and immunity in medical devices. This is being accomplished by adding EMI filtering components to a cable assembly. These changes add componentry and manufacturing time to an assembly. IEC 60601-1: 4th edition has been mandatory in Europe since January 2019, and is being implemented in the U.S.
Kashi: Regulatory practices will add a layer of safety for the product and application. The evolving regulatory environment has been impacting development and manufacture of electronic components. Because of these regulatory impacts, unit cost of products is likely to increase due to the need to perform new and incremental testing to meet these new standards. In addition, some custom parts may warrant modification or redesign.
Barbella: How are requests for low-power design impacting custom electronic component development?
Allahakbari: Low power designs mean more restrictions on the power rails, current draw, and power consumption of the entire system. Sometimes a particular process should be done in a very short interval. This method would add more limitations to the design because more efficient code needs to be developed and executed to meet the timing requirements. Technically, low power designs mean less processing power that would limit the designer to meet the requirements. Sometimes the designer cannot meet the requirements due to lack of component capabilities and should come up with an innovative and creative approach to bring down the average power by shutting down the system and bring it back on when necessary.
Bratton: Low-power electronics include devices such as cardiac pacemakers, cardiac defibrillators, neurological stimulators, muscle stimulation devices, hearing aids, bone conduction amplifiers, tinnitus blockers, and cochlear implants. Plastics One has worked with OEM companies to help develop many of these solutions. We treat power requirements as an input to the overall project success and either verify or validate the performance criteria for each.
Burke: New chemistry and designs are needed to keep up with low power and high power ends of the spectrum.
Cianciolo: Low power requirements allow the use of a smaller connector with smaller contacts with higher density. Creepage and clearance becomes less of an issue when you are running low voltage and current. Smaller connectors lead to smaller devices, which are more manageable in certain situations.
Fritz: Low-power design is an important topic, especially for the growing number of applications that require hand-held and/or battery-powered operation. Coming up with the most efficient design requires a holistic look at the whole system.
Kashi: The need for low-power design is increasing with the advancements in the IoT application space. Therefore, the impacts have included digitizing sensing components and designing them in a way that allows the capability to gather data on demand rather than continuously taking measurements. In addition, there is a shift to including signal processing at the receiver rather than at the measuring device.
Labbe: The trend to smaller, portable, home use devices has definitely driven changes to electronics design. With the widespread use of cell phones and other consumer electronics, users have become accustomed to managing devices and daily recharging requirements, with expectations of long product life in a small device. Designing low-power electronics, managing lithium battery recharge requirements, and even powering devices remotely using induction or ultrasonic technologies is now a requirement for medical device development engineers. Additionally, users expect high quality displays with intuitive user interface designs, which increases the need for up front human factors design and more creative software techniques to manage these requirements in a low power platform.
Barbella: Is your company impacted by green initiatives from medtech customers in Europe or the U.S.?
Allahakbari: We have been designing and manufacturing medical electronics over the last decade under the RoHS, REACH, SVHC, Conflict Material, Prop-65 directives and regulations. Those requirements are well understood, although frequently changing. New requirements are surfacing encouraging green (reduced carbon footprint) initiatives from manufacturers. This focuses not only on the manufactured products that we produce, but the processes employed in manufacturing as well. We’re facing a new and challenging manufacturing environment where the engineers must “Design for Green Manufacturing” to minimize the carbon footprint throughout the entire product lifecycle.
Bratton: Plastics One is a worldwide manufacturer. Our compliance team has not only to review U.S. FDA regulations but keep up with many other countries and compliance programs such as the EU FDA. The main effect this has on products is with some of the initiatives certain elements are either reduced or limited in percentage content. As the regulations change, manufacturers are required to revalidate the product if the raw components shift an insignificant proportion.
Burke: Yes, the regulatory needs of RoHS, REACH, and all the other bodies need resources that are very much hard costs and most OEMs deem them as part of SG&A and standard operating costs. Regulatory concerns eat into resource management and profits, because if the ticket to the dance is getting all the data needed for an OEM’s regulatory requirements, we cannot bill our customers for these costs. As a company, we love the environmental side of complying with all the regulatory bodies, but it gets harder every day to do that and not drain talent away from designing the solutions our customers have asked for originally.
Cianciolo: It’s a global market, so we’ve seen a few requests for more eco-friendly materials required in Europe that aren’t required elsewhere. It hasn’t had a huge impact on our business so far, but it could change at any moment.
Kashi: With the shift in focus to considering environmental regulations and adopting green approaches, TE Connectivity (TE) has taken steps to provide more environmental-friendly products. For example, TE has taken steps to replacing some materials that were noncompliant with environmental regulations. TE is also in the process of identifying some alternative materials that can be integrated into existing and new products.
Labbe: Requirements such as RoHS, REACH, and others have definitely driven changes in the electronics industry. New materials, suppliers, and increased supplier controls are required to meet these regulations. At this point, these have become a standard part of our everyday processes.
Barbella: Are there any other trends you are noticing in custom electronic component development for medical devices?
Allahakbari: Studying and researching advanced topics and new technologies are usually considered for supporting medical applications. Designers should be motivated to go beyond their comfort zone and develop, and evaluate something new and unproven, within reason. In some cases, the company needs to initiate the effort in the industry to support specific applications that could be very costly, and it may result in open-ended projects. Some complicated medical applications rely on new publications that do not have enough data to support the claim and technically they are just theoretical approaches that may or may not result in real-life applications.
Bratton: The primary trend we have been following is the complexity of devices. As [previously] mentioned, there has been a shift from simplistic connections to complete custom systems. Many OEM medical companies are not only designing and manufacturing surgical tools but the cables, connectors, and software boxes that drive their operations.
Burke: A lack of standards in competing designs for certain segments—i.e., connectors that you’ll find in any operating room in the world and every device has either a custom application or five different systems when one could carry the power and signals needed for the data a surgical team needs to assure patient health and vitals.
Kinyanjui: You can’t talk trends in medical without talking about robotics. Like in other industries, the medical industry continues to expand the use of robots. Connectivity solutions are driving the ability to reduce size, with faster data processing. When we work on connectivity solutions for medical robotics, we are always very specific about the user’s environment and sterilization needs to create a low-risk device.
Kashi: Many of the advances will be incremental. There will continue to be advancements in miniaturization, performance, artificial intelligence, and price for digital and wireless sensing. With the developments within the Internet of Medical Things (IoMT), there will also be an increasing emphasis on portable, wearable devices to monitor patients to enhance remote operating capabilities.
Imagine, for a moment, the possibilities: Organs and joints that never fail, bones that never break, neurons that never misfire, muscles that never tear, and cells that never damage. Invincibility at its very best.
Mankind is well on its way to achieving such immortality with science-fiction-inspired technological advancements like 3D-printed tissue, artificial hearts, smartphone-controlled arms, spray-on skin, exoskeletons, and brain-computer interface devices.
The latter innovation, supported by a growing cache of scientific research, fulfills humankind’s decades-long dream to harness cerebral electricity to directly control machinery and/or non-working body parts. Though still in its early stages, the technology has already been used to help a paralyzed teenager kick a soccer ball, and a quadriplegic man regain control of his hand, wrist, and fingers. “It’s certainly a huge challenge to restore movement to a paralyzed limb,” quadriplegic Ian Burkhart acknowledges in a video posted to the Battelle Memorial Institute website. The 28-year-old Ohio resident was paralyzed in a 2010 diving accident.
“The biggest thing is it has to be speculative because that’s how you make the big leaps forward,” Burkhart says in the Battelle video. “It has to be seen as something that can’t be done. Yes, there might be something that’s way too far-fetched and isn’t realistic, but if you put the right team together...I think it’s something that can be done. Take those wild ideas so that way you can push science forward.”
Burkhart has helped push science forward by participating in a five-year study of Battelle’s BCI technology (NeuroLife). Conducted in collaboration with Ohio State University Wexner Medical Center doctors, the analysis proved it possible for paralyzed patients to move their hands and fingers via mere thought.
Such mental prowess (and promise) is substantiated by various other BCI devices in development: Carnegie Mellon and University of Minnesota researchers, for example, have built a mind-controlled robotic arm capable of continuously tracking and following a computer cursor, while BrainGate Consortium’s brain-computer interface technology has proven its worth with tetraplegic study participants.
BrainGate’s investigational device features an internal neural signal sensor that detects the signs presaging intended movements in the brain’s cortex. Those signals are then decoded and routed to an external device. The implantable sensor houses a baby aspirin-sized chip with 100 electrode sensors—each thinner than a human hair—that can detect the brain’s electrical activity.
Developing electronic components small enough for human implantation requires a proficiency in miniaturization, custom design, enhanced functionality, and material selection, among others. It also demands tremendous flexibility, as the Internet of Things, big data, and analytics continue to pervade the $400 billion global medtech market.
To better understand the factors and trends influencing the custom medical electronics market (both now and in the future), Medical Product Outsourcing spoke with numerous industry professionals over the last few weeks. Input came from:
Farshid Allahakbari, head of the electrical engineering team at Irvine, Calif.-based FUTEK Advanced Sensor Technology Inc., a manufacturer of load cells, torque sensors, pressure sensors, multi-axis sensors, and related instruments/software.
Drew Bratton, sales manager at Plastics One Inc., a Roanoke, Va.-headquartered medical contract manufacturer.
Brian Burke, sales manager at BAYCABLE, a custom cable and cable assembly solutions provider located in Fremont, Calif.
David Cianciolo and Patrick Kinyanjui, engineering director and principal engineer, respectively, at Fischer Connectors, a global manufacturer of high-performance push-pull circular connectors and cable assemblies.
Christian Fritz, sales director, Motion Control & Electronics, at maxon group, a Swiss manufacturer and supplier of high-precision drive systems.
Bala Kashi, business development manager, Medical Sensors, at TE Connectivity Ltd., a global manufacturer of connectivity and sensor solutions for various industries, including energy, medical technology, data communications, transportation, industrial applications, and the home.
Mike Labbe, R&D director at Valtronic, an electronics manufacturing services developer of highly complex printed circuit boards, hybrid circuits, and complete medical devices.
Michael Barbella: What factors must be taken into consideration when designing custom electronic components for medical devices?
Drew Bratton: When designing custom electronics components, there are many attributes to focus on. To list some of the inputs to our design and development process; functional, electrical, software, environmental performances, physical characteristics, user and patient safety considerations, and target cost. Plastics One has invested in developing an integrated system to guide our development of complete custom electrode following ISO 13485 guidelines to ensure no inputs are not reviewed.
Brian Burke: Are the devices going in the body? Will autoclave be needed? (Multi-use cleaning procedures). Can the market bear another player in the chosen segment? Is the idea manufacturable and repeatable in production? Are the customer’s engineers open to our knowledge and know-how?
David Cianciolo: When dealing with a connectivity solution in medical devices, you have to know what Medical Device Class of the equipment is. That will determine the risk level and the degree of validation required. Higher Device Classes will require more scrutiny, testing, and higher validation quantities
Patrick Kinyanjui: Device Class 1 or 2 materials may not be acceptable for a Class 3 device. Most connectors do not normally meet the requirements for implantable applications. Class 3 devices also require a higher level of traceability down to the materials at every level of components.
Christian Fritz: While the medical device industry is subject to similar market pressures as other industries, there are a couple of unique challenges that impact the design of custom electronics and the development of low-level firmware and application specific software for medical applications. Innovation, tight time-to-market requirements, and the need to provide a cost-effective solution are common factors to consider when designing custom electronics. In the medical device industry, it is very important to consider the type of customer the device is targeting. The requirements are vastly different if the device is designed for a service provider (hospital, laboratories) or the end user (patient). On top of those economic factors the medical industry requires an increased level of certifiable safety and reliability, which has a major impact on the design. Regulatory requirements need to be factored in from the very early stages of prototyping throughout the life-cycle of the hardware and software design.
Bala Kashi: When designing custom electronic components for medical devices, there are many factors that need to be considered. Not only do factors such as understanding the application, validations, parameters, material selection, standards to be met, cleanliness, and cost targets need to be acknowledged but there are other considerations. Some of these other considerations include the ruggedness, size, accuracy, and scalability of the sensors since medical components can be exposed to various harsh conditions and media. For example, if the sensor is being used for an implant application, it is required to be more critical for biocompatibility since it will have long-term contact with body parts. Additionally, the sensor will need to function long-term with almost no probability of fail.
Mike Labbe: There are many specific factors to consider in designing medical devices. First are the appropriate regulations that must be met for that product to get approved for market. There are many FDA and ISO standards related to electrical safety, EMC compliance, human factors and usability, risk management, HIPAA, and cybersecurity concerns that must be addressed in the design process if the product is to be cleared for distribution, and these must be incorporated into the design process from the very beginning.
Time to market for a medical device can be much longer than consumer products. Often there are regulatory clearances, agency testing, and clinical trials that must be completed, which can take five years or more. With this in mind, selection of components in the design phase is critical, otherwise components selected in the initial design will be obsolete before the product ever gets to production. Fast-changing technologies such as displays, batteries, memory, and other electronics used in mass consumer devices are especially important to focus on in the selection process to find parts that will have an adequate life to support product launch.
A third critical factor to consider in customer electronic medical devices is Design for Manufacturability (DFM). Understandably, the focus during product development is often on functionality and clinical effectivity. However, to have a successful product, the end result must be a design that can be manufactured in a consistent manner, at an appropriate cost and with high quality. Focus on getting DFM input during the design and prototyping phase from an experienced manufacturing team in the processes required to build the product is essential.
Barbella: Please discuss some of the challenges in designing and manufacturing custom electronic components for medical devices. How has your company overcome these challenges?
Farshid Allahakbari: To meet the requirements of any custom medical projects, a designer usually faces several obstacles. Typically, we classify the anticipated constraints into two disciplines: design vs. manufacturing. Design phase challenges are reliant on experience and knowledge of the designers. Medical applications typically require a miniature board with limitations. These constraints vary and consist of the following:
- Designing efficient low power products or providing digital power management to add flexibility to the design structure.
- Providing precise, low power, low noise, mixed-signal design for medical measurement systems.
- Designing PCB layouts presents challenges since the designs are usually expected to be dense and compact, and the designers need to maintain proper placement and partitioning disciplines to minimize the effect of noise cross-coupling and injection. Most of the medical custom designs require safety features to be implemented to immunize the system from failures while providing fault detection feedback to the master system. Meeting the requirements of these types of applications are challenging.
- Selecting components can that meet design requirements can be challenging.
- Meeting deadlines for digital designs is an obstacle we face as there can be delays in the digital systems for control feedback. Defining mounting provisions is a complicated process since the allocated spaces are tight and come with limitations.
Manufacturing challenges that are usually overcome by:
- Qualifying the right vendors.
- Continually monitoring and evaluating the outcome of the manufacturing and production lines.
- Providing a proper test procedure for production.
- Evaluating the quality of reports frequently.
- Providing test scenarios to catch failures prior to shipping the final product to the customers.
- Automating the systems in order to avoid human errors in production.
- Providing the proper ESD protected and temperature-controlled environment for the testing and calibration.
- Training and educating operators on the production floor to support the manufacturing process.
- Providing a thorough traceable production system.
Bratton: Many of the challenges we face in designing and manufacturing custom electronic components related to the seamless integration of multiple manufacturers and processes. Projects we are working on now require much more than just a molded solution, rather various levels of manufacturing, packaging, and sterilization. Our company has overcome these challenges by qualifying a diverse group of vendors and providing turnkey manufacturing solutions (dock to stock).
Burke: Some of the [previously mentioned] thoughts.
Engineering staffs and resource allocation both at the customer and for us as well. Everything is getting smaller and smaller and miniaturization presents challenges trying to extrude insulation and mold parts that are smaller and have tighter tolerances. We listen to what the customer needs and see if it fits in our capabilities and discern if they are a viable potential customer and partner. We then sit with our engineering gang and see if we can meet these requirements in the timeline required. Often we need to do some experimental runs to test the range of capability for newer custom parts that are outside of our current capability. We can run various iterations and work with our customers throughout this process of collaborative design.
Cianciolo: Due to sterilization, requirements sealing of a connector present a bigger challenge in the medical industry. When working on a new medical cable, we always inquire about the end-user’s sterilization methods. We may need to use higher temperature materials such as silicone, FEP, PPSU, etc., for the construction of the assembly.
We also look at isolation characteristics; Medical Device Classification Type B, BF, or CF, that are unique to each medical device. Type BF and CF require extra tests that Type B doesn’t require.
We ask many questions to make sure we know exactly how and where the equipment is being used and what the expected sterilization protocol is. That initial interview is needed so design time is reduced.
Fritz: Over the last couple of years, the medical device industry has been facing a changing landscape of regulatory requirements. This put strain on the design teams who had to build up knowledge and refine existing processes while executing a growing number of projects. To increase their engineering efficiency, many medical device companies turned to third parties and components vendors for some of the vertical expertise needed to complete their devices. As a response, maxon, a key provider of small high-precision motors and motor control electronics, has increased its service offering and is collaborating closely with medical device builders for their electronics needs. Leveraging the existing building blocks of their off-the-shelf motor controllers, maxon can quickly provide a customized solution and help with the documentation needed for medical certifications.
Kashi: The design and manufacture of medical electronic components, including sensors, involves many challenges. Material selection, validation tests, quality systems, scaling, and custom parametric tests are all examples of some of the challenges faced during this process. Since sterilization is a high priority within the medical industry, an additional challenge is the requirements regarding how components are to be manufactured—meaning, the space that they are developed within needs to follow specific guidelines to ensure a certain level of cleanliness required by the medical industry.
Labbe: As mentioned, understanding the expected life cycle of components selected in the design phase is critical to assure consistent supply. Valtronic works closely with the suppliers of these components to choose options that have an assured product life through the first several years of manufacture. Wherever possible, alternate components are also included on the approved BOM to allow for supply chain flexibility. Additionally, Valtronic continuously monitors lead times and orders components in advance to avoid manufacturing disruption.
Design for Manufacturing is also critical in medical device design. Once a product has been designed, prototyped, tested, and cleared through regulatory agencies, changes to the design and manufacturing can be time consuming and expensive. It is critical that products built once the device is in full series production meet all of the quality, performance, and safety specifications of the initial units used in product verification and clinical validation studies. To achieve this, input from experts in the processes required for production should be included in the design cycle from the beginning.
Barbella: What are customers demanding or expecting in their custom electronic components?
Allahakbari: Customers mostly dictate the size, performance, and cost of the components. Typically, the first size and performance conflict is with cost. A direct dialogue and transparency with the customer is essential to understand what requirements are crucial. A straightforward custom design could turn to a very complicated design if requirements aren’t received up front.
Bratton: Customers are now demanding more complex solutions than ever before. With new technology and software developments, the components have had to shift in culture to support the vast array of modern technology. Many years ago, cables and connectors were very simplistic and just a means to connect items. Many of our clients are now looking to combine multiple cables, electrodes, and circuitry all into one complete system, whereas in the past it may have been various items working in tandem.
Burke: Regulatory help and sourcing expertise. Custom manufacturing requirements to curb costs and lead times.
Cianciolo: Customers are also looking for connectivity solutions that integrate sensors to count the number of procedures, or total time used, keeping patients safer. Design engineers are definitely reimagining connectivity solutions to gather data and make the devices smarter and safer.
Kinyanjui: Our customers are looking for smarter products that include electronic identification. This gives the customer the ability to offer a system that will preset the device settings for its unique procedure. Others go further and demand that the connectivity solution identifies the exact device. This increasing demand for traceability means that we work with our customers to design in ID technology. This can be done by using a specific sub-component integrated into the cable assembly.
Fritz: While the design needs to meet the technical requirements and provide the performance that the medical device requires, it is important to provide enough flexibility to allow for adjustments that might become necessary during prototyping stages or clinical trials. At the same time, it is important to consider the certification needs early on. Providers of custom electronics need to apply documented development processes and comprehensive test documentation that aids a smooth certification process.
Kashi: The medical industry requires electronic component options for many different medical applications. However, there are some demands from customers that remain consistent since some sensing components measure various applications that relate to human life. For example, the accuracy and functionality of medical electronic components for life support devices is critical for that application. In addition, medical sensing components need to be reliable and meet all medical standards.
Labbe: There are a couple of shifts Valtronic has been observing in the medical electronics CM industry in the last few years. First, many more customers are becoming more educated in the SMT and manufacturing process technologies, and are demanding these processes be used on their products. Automated Solder Paste Inspection (SPI), closed-loop Automated Optical Inspection (AOI), Artificial Intelligence systems, and other advanced technologies are expected by more educated customers.
Secondly, more customers are requesting full turn-key services. Customers are outsourcing more aspects of design and DFM review before transferring to contract manufacturers. Additionally, there is more demand to perform full turnkey manufacturing, from supply chain management, sub-assembly manufacture to full device assembly, test, packaging, and even distribution. Providing more value-added services has become more critical for partnerships with OEM customers in today’s marketplace.
Barbella: How is IoT (Internet of Things) influencing custom electronic component development?
Bratton: The most significant shift we have seen in the past five to 10 years is the integration of home healthcare and data collection. Many products are shifting to allow them to be used through small home devices such as a smartphone or a tablet. Diagnostic or monitoring tests that have historically been conducted in a hospital or clinic are now completed within the comfort of your own home. Many of these devices are connected through the internet, and data can be uploaded to cloud storage for further review and analysis.
Burke: The chip and big connector guys are so in love with that segment, that sometimes it’s hard to get attention on a legacy product—i.e., power and signal standard products.
Fritz: There is an increased interest for leveraging IoT technologies (communication interfaces, security, increased data collection and analytics) that allow medical device builders to broaden their services offering, increase the uptime of their systems, and lower the maintenance cost. This is especially true for devices targeted at medical service providers. When it comes to medical devices, it is important to evaluate these technologies with a strong focus on security and data protection.
Kashi: There is a push toward developing sensing components that are suitable for IoT applications since sensors are the means to gather the data needed for successful deployment of these IoT applications. The data collected by sensors can help medical professionals understand critical situations faster and more accurately and enable patients to be more informed about their conditions and improvements. For example, if the sensor is being used within an in-home care application, it might have a wireless transmitter to send the data remotely to the cloud. Physicians or the patients can have the ability to download the data for analysis. Additionally, the sensor devices are required to be accurate, precise, and reliable to avoid false triggering of any event. From blood pressure sensing to dialysis equipment monitoring, invasive and non-invasive sensors are providing localized data and information to the cloud to better serve patients and the medical industry.
Labbe: IoT is becoming ubiquitous in the industry. This shift requires expanded expertise in RF and wireless technologies, software and data management, as well as protocols for security and encryption. These are rapidly developing and changing technologies, and there are many groups popping up with specific expertise to stay on top of these developments. It is critical to have in-house expertise, or relationships with external experts to stay on top of these technologies.
Barbella: How is big data influencing custom electronic component development?
Allahakbari: In general, big data analysis requires fast processors, sophisticated digital systems, and high-power communication that could make the custom designs complicated. We cannot process big data through traditional software, so companies have implemented cloud-based tools to mine massive amounts of data and deliver the data successfully. A lot of development, automation, and safety features are required to be considered during the data-gathering processes. This makes the custom designs even more complicated for medical applications.
Bratton: Within contract manufacturing, we do not experience the direct influence of big data. We do, however, work with medical device OEM companies which relay many of the results of these studies and data collections to Plastics One for review. How does this benefit a manufacturer? The more useful data that is collected helps manufacturers such as Plastics One make products that will last longer and perform at a higher level within their field of use.
Burke: Data analytics and auction sites cut down on how customer service levels and past performance affect procurement strategies. You can perform spectacularly for years, but if procurement departments go to auction or purely data/prices-based decision-making, all that good will is never baked into the cake.
Kashi: Big data is influencing custom electronic component development in a positive way. The feedback gathered by big data can influence the functionality and the parameters monitored. For example, since the patient data will get stored remotely, it is possible to analyze this big data to understand the predictability prior to the occurrence of an event. The big data analysis can check for effectiveness of the treatment by age, gender, race, location, and patient habits. Therefore, with this information, the treatments can be more customized to specific patients. In the future, there will also be an increasing emphasis on smart sensing, using on-board sensor capabilities to analyze data and transmit vital data rather than simply “big” data.
Labbe: Big data has large impacts on the requirements for software development. Hardware to collect, transmit, and manage big data is fairly well-established. Especially in medical devices, there is an increasing trend to treating patients away from the hospitals. This has led to many devices that collect data from the patient, and upload to the cloud. Manufacturers or external services providers then need to sort through all this data in near real time, and implement algorithms to detect events or critical data and provide this digested information to the physicians and caregivers for action. This requires significant upfront investment in software systems for cybersecurity, algorithm development, and HIPAA compliant processes. Regulatory agencies are also beginning to review and regulate these aspects more critically so a comprehensive strategy to manage and protect this data is required.
Barbella: How is AI (artificial intelligence) influencing custom electronic component development?
Allahakbari: In general, AI aims for the design and implementation of processes that, when run on physical architectures, trigger the maximization of desired results by focusing on productivity enhancement. Some of the lines of work of the research intelligence are: design and prototyping of electronic boards and advanced devices for integration in medical equipment and robot surgeries; ARM solution designs for advanced data processing to support medical applications; and design of intelligent systems for precision agriculture with feedback and self-fault detect features for medical applications.
Bratton: Many of our customers are beginning to integrate “smart” components and circuitry electronics within the cables and connectors we manufacture. Recent integrations we have worked with include applications to limit the number of uses for disposable medical devices and automatic device profile downloads to the medical instruments. These developments help reduce the patient risk preventing the products from being misused in a surgical setting.
Burke: Only the big chip and connector guys have the resources to utilize AI to any degree.
Kinyanjui: Right now, integrating AI into medical devices is a goal, not a reality. The FDA needs to update rules before AI will be fully designed for the medical community. We know it’s coming, and we are looking at it. It’s going to be an interesting ride the next few years as we figure out how AI can ethically and practically be introduced.
Kashi: Similar to the positive impacts of big data, AI allows for predictability before any patient event. Based on patient patterns, AI not only enables the ability to predict a patient event but act on that event. For example, AI can provide notifications to paramedics or the physician if the medical machine is failing or if the patient is showing certain symptoms. Additionally, AI can interact with other devices that are hooked to the patient.
Barbella: Is the trend toward miniaturization of medical devices driving a need for flexible custom electronic components?
Allahakbari: Physical limitations and constraints in medical applications require custom designs to have more advanced and complicated layouts. For example, a rigid/flex design is usually required to provide a smaller package with a more complicated assembly. The connectors and mounting provisions used in medical applications drive the need for flexible low-profile designs. Usually the assembly needs to be folded to fit in the allocated area. This process forces the designer to consider more design rules during the PCB design and makes the manufacturing more expensive. It also forces the designer to be more quality-minded when it comes to selecting fabrication houses.
Bratton: Medical devices are continually undergoing design changes and improvements to reduce size. As size reduces, many performance characteristics must be reviewed and tested before production implementation. Plastics One’s pre-clinical division has been at the forefront of miniaturization, working with university students and doctors on cannulas and electrodes used in drug delivery systems. We have also invested in tool-making technology to help facilitate mold fabrication on a miniature scale.
Burke: Yes, and that entails endless hours of research as the products get smaller, the labor is more specialized, and there is less apt for big ramps. Miniaturization of wire and connectors results in a need for automating the termination process, which requires much research on how to automate, as most applications are extremely specialized.
Kinyanjui: Weight is always a factor in handheld devices, due to the fatigue factor in the OR, as procedures can easily run for hours at a times. Fortunately, new technology allows connectors to be smaller with more dense connections. Size and weight become a bigger factor with wearables. We have a plastic lightweight connector that is specifically designed for wearables. We’ve reduced weight by changing the materials used in manufacturing. We actually see a greater focus on miniaturization and weight in the military since it’s been a focus for so long. We find we are able to apply what we’ve learned in that market to our medical customers.
Fritz: There is clearly a trend towards miniaturization, not only in regard to electronic components but also for mechanical and electromechanical components and subsystems.
As a result, maxon increased their offering regarding product customization and value-add services changing the role they play in the development process. An active collaboration on the development of highly customized components or integrated subsystems allows for compact design, maximum performance and helps address the increasing complexity of medical devices.
Kashi: Wearable and portable devices require smaller, lighter, and less power-hungry devices that allow for more sensing functionality within one device. Devices such as these require more resources to develop and are more expensive to build. Therefore, during the design phase, there is a need to consider multiple other potential applications and try to address them as well.
Labbe: Historically the need for miniaturization of medical devices led to a significant use of bare and stacked die, flip, and wire-bonded assemblies to minimize the component size. Today’s packaged components and ICs are becoming so optimized for size that these more complex processes are required less often. Placement of packaged components down to 01005 size and lower, barely visible to the naked eye, has resulted in increased need for automated monitoring and inspection processes for controlling quality. Bottom terminated assemblies such as BGA and QFN packaging require more sophisticated assembly and reflow profile development and optimization, as well as X-ray and other inspection techniques to monitor processes. In-process and final functional testing at the manufacturing location are more critical than ever to detect and correct issues in real time and provide feedback to the process.
Valtronic is also seeing an increasing use of flexible and rigid-flex assemblies. These approaches allow for using more standard manufacturing techniques in production, and then folding and packing electronics in the final assembly stage to minimize size and space of finished devices. This presents a different set of challenges in the design, manufacture, and supplier selection for these more complex flexible components.
Barbella: How else is miniaturization impacting custom electronic component development?
Allahakbari: Miniaturization usually adds more unknowns to the equation as far as the component selection, the PCB layout, and the manufacturing process. Sometimes, miniaturization limits force the designer to fit the same amount of components that require a large amount of room in a smaller area. The limitations cause additional challenges such as heat dissipation because there isn’t enough surface area to dissipate the heat. In this case, the design would require more attention to the design approach, component selection, power optimization, testing, and validation processes.
Bratton: With a reduction in size, many solutions are becoming tailored to each specific project’s end use versus an off-the-shelf approach. Historically many manufacturers, including Plastics One, have had a line card of the products they can produce. We have seen a shift in this mindset where many solutions require specific and proprietary electronic components.
Burke: Smaller doesn’t always mean cheaper, and there is a need to develop packaging to assure no damage in any of the processes. New test fixtures that mirror the smaller products. Automation and research factors go up exponentially.
Kashi: With the drive toward developing miniaturized components, there have been impacts within the development process. Time and cost associated with development has increased due to the increased resources and the additional testing needed to confirm products work efficiently. There is also the need to fit all the same functionality into a smaller package. For example, shrinking sensor devices will take a lot of engineering. It will be costly since new designs may be required in addition to using expensive material and precise components. There will also be the need for highly accurate manufacturing techniques since the devices will be so small. To ensure performance won’t be compromised, the new devices may be required to go through extensive validation tests, third party certifications, etc.
Barbella: In what ways is the changing regulatory environment impacting custom electronic component development?
Allahakbari: While our component-level sensors and electronics are not subject to the more stringent FDA and European finished medical device regulations, they are subject to an ever-changing materials restrictions and electronic emission/susceptibility landscape of regulations. Many our design choices are governed by the limited availability of materials that must conform to RoHS and REACH Directives that change almost every year, as well as emission and susceptibility testing regulations that are sometimes revised with new acceptance criteria. At best, this mandates retesting and, at worst, a redesign of mature products to ensure continued compliance. As electronic component manufacturers switched over from non-RoHS to RoHS components, there was an interruption in the supply chain when they stopped producing the obsolete parts before the new RoHS components became readily available. We continue to face challenges with the availability of RoHS-compliant components today.
Bratton: To list a few of the significant regulations we follow: RoHS, Reach, Health Canada, Latex Free, and Animal Derived Materials. Each of these regulations is constantly updating standards, requiring manufacturers to review updates every year. The main effect this has on products is with some of the initiatives, certain elements are either reduced or limited in percentage content. As the regulations change, manufacturers are required to revalidate the product if the raw components shift in high proportion.
Burke: The non-revenue generating aspect of compliance is a constant battle every manufacturer and supplier is dealing with. The time spent on compliance must be covered along with engineering, manufacturing, and all other SG&A costs. The big guys factor in these costs and skirt them by advising on standards committees and feel they have done their part, leaving smaller manufacturers to bear the brunt of a lot of those un-recoverable costs.
Cianciolo: There are some recent changes in IEC 60601-1: 4th edition that require reduced radiated emission and immunity in medical devices. This is being accomplished by adding EMI filtering components to a cable assembly. These changes add componentry and manufacturing time to an assembly. IEC 60601-1: 4th edition has been mandatory in Europe since January 2019, and is being implemented in the U.S.
Kashi: Regulatory practices will add a layer of safety for the product and application. The evolving regulatory environment has been impacting development and manufacture of electronic components. Because of these regulatory impacts, unit cost of products is likely to increase due to the need to perform new and incremental testing to meet these new standards. In addition, some custom parts may warrant modification or redesign.
Barbella: How are requests for low-power design impacting custom electronic component development?
Allahakbari: Low power designs mean more restrictions on the power rails, current draw, and power consumption of the entire system. Sometimes a particular process should be done in a very short interval. This method would add more limitations to the design because more efficient code needs to be developed and executed to meet the timing requirements. Technically, low power designs mean less processing power that would limit the designer to meet the requirements. Sometimes the designer cannot meet the requirements due to lack of component capabilities and should come up with an innovative and creative approach to bring down the average power by shutting down the system and bring it back on when necessary.
Bratton: Low-power electronics include devices such as cardiac pacemakers, cardiac defibrillators, neurological stimulators, muscle stimulation devices, hearing aids, bone conduction amplifiers, tinnitus blockers, and cochlear implants. Plastics One has worked with OEM companies to help develop many of these solutions. We treat power requirements as an input to the overall project success and either verify or validate the performance criteria for each.
Burke: New chemistry and designs are needed to keep up with low power and high power ends of the spectrum.
Cianciolo: Low power requirements allow the use of a smaller connector with smaller contacts with higher density. Creepage and clearance becomes less of an issue when you are running low voltage and current. Smaller connectors lead to smaller devices, which are more manageable in certain situations.
Fritz: Low-power design is an important topic, especially for the growing number of applications that require hand-held and/or battery-powered operation. Coming up with the most efficient design requires a holistic look at the whole system.
Kashi: The need for low-power design is increasing with the advancements in the IoT application space. Therefore, the impacts have included digitizing sensing components and designing them in a way that allows the capability to gather data on demand rather than continuously taking measurements. In addition, there is a shift to including signal processing at the receiver rather than at the measuring device.
Labbe: The trend to smaller, portable, home use devices has definitely driven changes to electronics design. With the widespread use of cell phones and other consumer electronics, users have become accustomed to managing devices and daily recharging requirements, with expectations of long product life in a small device. Designing low-power electronics, managing lithium battery recharge requirements, and even powering devices remotely using induction or ultrasonic technologies is now a requirement for medical device development engineers. Additionally, users expect high quality displays with intuitive user interface designs, which increases the need for up front human factors design and more creative software techniques to manage these requirements in a low power platform.
Barbella: Is your company impacted by green initiatives from medtech customers in Europe or the U.S.?
Allahakbari: We have been designing and manufacturing medical electronics over the last decade under the RoHS, REACH, SVHC, Conflict Material, Prop-65 directives and regulations. Those requirements are well understood, although frequently changing. New requirements are surfacing encouraging green (reduced carbon footprint) initiatives from manufacturers. This focuses not only on the manufactured products that we produce, but the processes employed in manufacturing as well. We’re facing a new and challenging manufacturing environment where the engineers must “Design for Green Manufacturing” to minimize the carbon footprint throughout the entire product lifecycle.
Bratton: Plastics One is a worldwide manufacturer. Our compliance team has not only to review U.S. FDA regulations but keep up with many other countries and compliance programs such as the EU FDA. The main effect this has on products is with some of the initiatives certain elements are either reduced or limited in percentage content. As the regulations change, manufacturers are required to revalidate the product if the raw components shift an insignificant proportion.
Burke: Yes, the regulatory needs of RoHS, REACH, and all the other bodies need resources that are very much hard costs and most OEMs deem them as part of SG&A and standard operating costs. Regulatory concerns eat into resource management and profits, because if the ticket to the dance is getting all the data needed for an OEM’s regulatory requirements, we cannot bill our customers for these costs. As a company, we love the environmental side of complying with all the regulatory bodies, but it gets harder every day to do that and not drain talent away from designing the solutions our customers have asked for originally.
Cianciolo: It’s a global market, so we’ve seen a few requests for more eco-friendly materials required in Europe that aren’t required elsewhere. It hasn’t had a huge impact on our business so far, but it could change at any moment.
Kashi: With the shift in focus to considering environmental regulations and adopting green approaches, TE Connectivity (TE) has taken steps to provide more environmental-friendly products. For example, TE has taken steps to replacing some materials that were noncompliant with environmental regulations. TE is also in the process of identifying some alternative materials that can be integrated into existing and new products.
Labbe: Requirements such as RoHS, REACH, and others have definitely driven changes in the electronics industry. New materials, suppliers, and increased supplier controls are required to meet these regulations. At this point, these have become a standard part of our everyday processes.
Barbella: Are there any other trends you are noticing in custom electronic component development for medical devices?
Allahakbari: Studying and researching advanced topics and new technologies are usually considered for supporting medical applications. Designers should be motivated to go beyond their comfort zone and develop, and evaluate something new and unproven, within reason. In some cases, the company needs to initiate the effort in the industry to support specific applications that could be very costly, and it may result in open-ended projects. Some complicated medical applications rely on new publications that do not have enough data to support the claim and technically they are just theoretical approaches that may or may not result in real-life applications.
Bratton: The primary trend we have been following is the complexity of devices. As [previously] mentioned, there has been a shift from simplistic connections to complete custom systems. Many OEM medical companies are not only designing and manufacturing surgical tools but the cables, connectors, and software boxes that drive their operations.
Burke: A lack of standards in competing designs for certain segments—i.e., connectors that you’ll find in any operating room in the world and every device has either a custom application or five different systems when one could carry the power and signals needed for the data a surgical team needs to assure patient health and vitals.
Kinyanjui: You can’t talk trends in medical without talking about robotics. Like in other industries, the medical industry continues to expand the use of robots. Connectivity solutions are driving the ability to reduce size, with faster data processing. When we work on connectivity solutions for medical robotics, we are always very specific about the user’s environment and sterilization needs to create a low-risk device.
Kashi: Many of the advances will be incremental. There will continue to be advancements in miniaturization, performance, artificial intelligence, and price for digital and wireless sensing. With the developments within the Internet of Medical Things (IoMT), there will also be an increasing emphasis on portable, wearable devices to monitor patients to enhance remote operating capabilities.