Michael Barbella, Managing Editor06.13.19
Chris Troyanos knows his place among marathoners.
For more than four decades, he’s walked the delicate line between love and hate, acting as the yin to runners’ yang, the moon to their sun. He’s friend and foe wrapped in one—a prize to some, a persona non grata to others.
Troyanos, 62, owns Sports Medicine Consultants Inc., a 23-year-old Plymouth, Mass.-based firm that provides medical management, onsite medical care, and volunteer recruitment services for large-scale athletic events. A certified athletic trainer, Troyanos has coordinated and supervised medical coverage for dozens of sizable races throughout the United States and Canada, including the grueling 26.2-mile Boston Marathon.
Coordinating medical coverage for New England’s most widely viewed sporting event is quite the undertaking: Besides preparing for almost any running-related injury (shattered femurs, included) Troyanos also supervises 1,700 volunteer clinicians and the 28 medical tents along the eight-town route. The job requires some mastery of psychology as well, since no sprinter really wants to see Troyanos on race day.
“Runners need to understand what their limitations are and not try to push them,” Troyanos told Boston Magazine last month. “Boston has a tendency for people to want to do their best. We get that. But if you’re aware of your body’s limitations and how you’re feeling and don’t push that, the likelihood is you’re not going to see us in the medical tent.”
Sound advice for sure, but Troyanos will likely always have company in the tents, thanks to a bipolar Mother Nature and an incredibly strong human spirit. Both forces have detoured droves of marathoners in recent years, forcing them to retreat under heated blankets or in ice baths, or to rejuvenate themselves via Gatorade or a bag of potato chips.
This year’s casualty count was down from past races, despite the meteorological mayhem that ensued throughout the day (a bit of everything: clouds, wind, rain, lightning, sun). The Boston Athletic Association reported that 2,095 runners received medical attention along the route, with 950 of those participants undergoing treatment after the finish line and 77 runners eventually transported to area hospitals. Most of the injuries sustained during the race were minor (blisters, cramps, dehydration, hypothermia), though the sun’s unexpected appearance generated a rash of heatstroke victims within a two-and-a-half-hour period.
“[When the] sun came out, all of a sudden [there were] probably about 30 or 40 exertional heat strokes on the course and in this tent,” Troyanos divulged to WBZ-TV (CBS) after the April 15 race. “That’s pretty significant. Those are very serious illnesses. We handled them well, everyone is doing great.”
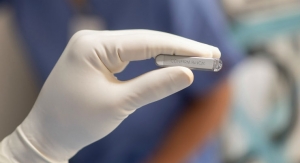
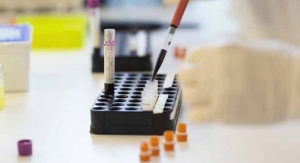
To ensure such a favorable prognosis, Troyanos used a handheld blood analyzation system from Abbott that produces lab-quality results within minutes from virtually any location. The company’s i-STAT technology measures 26 different analytes (substances in the body), including cardiac markers, gases, chemistries/electrolytes, coagulation, lactate, and hematology, from just two to three drops of blood.
First released in 1992, the i-STAT system is designed specifically for use at various points of care—from radiology and emergency departments to the intensive care unit and the operating room. More than 75,000 i-STAT systems are currently in operation, according to Abbott, with one in three U.S. hospitals employing the device to increase efficiency and quality of care.
The technology, however, has become fairly prevalent outside the hospital too—the paradigm shift to value-based healthcare has helped propel the i-STAT’s spread to outpatient clinics, doctor’s offices, long-term care facilities, emergency services vehicles, and rural institutions. The U.S. and Indian governments have used the device as well.
Athletic trainers and sporting event coordinators are perhaps the i-STAT system’s biggest fans, as the tool—evidenced by its presence at the Boston Marathon and numerous Abbott World Marathon Majors races—has helped legions of competitors return to play quicker than they would with conventional treatment.
“Without this portable tool, these runners showing signs of severe dehydration or hypernatremia would likely be transported to the hospital for diagnostic tests and follow up,” said Dr. Paul Jarvis, director of global medical affairs for Abbott’s point-of-care (POC) diagnostic business. “The fact that we can bring diagnostics right to the runner’s side is truly the power of [the i-STAT]. It’s wonderful for runners to have access to a tool like this that can help with very quick treatment. The power to finish the race is priceless. It’s a big win for runners.”
It’s also been a big win for Abbott, adding hundreds of millions of dollars to the multinational’s coffers and bolstering its share of the fast-growing global POC diagnostics/testing market, valued at $18.09 billion last year. Grand View Research data show this sector expanding 3.3 percent annually through 2025, with cancer marker testing growing at a higher pace over the forecast period due to a sharp rise in malignancies and the development of home testing kits.
Those same factors are expected to shape the POC diagnostics/testing market for years to come as an aging world population grapples with the various hellions of maturity—namely, cardiac disease, diabetes, Parkinson’s disease, and Alzheimer’s. Demand for home-based diagnostic devices and wearables as well as technological advancements that beget faster, easier-to-use products also are likely to drive market growth.
Such advancements are quickly coming to bear. Abbott, for example, has released newer, improved i-STAT models with Cloud connectivity, 2D barcode tracking ability, customizable range settings, and upgraded software. One of the versions—the i-STAT Alinity—was specifically designed for clinicians: The easy-grip handled tool offers on-screen guidance during the testing process and can identify both problems and potential solutions in real time; additionally, audio and light cues alert users to critical range test results that require immediate action. Moreover, the i-STAT Alinity is built for future sustainability.
“It was built to be ‘future-proofed,’ meaning it’s compatible with a variety of testing cartridges, including those designed for our current i-STAT platform, as well as tests that will be developed in the future,” Narendra Soman, Ph.D., R&D director for Abbott’s Point of Care Diagnostics business, noted in a company-sponsored Q&A about the Alinity. “In fact, we’ve designed i-STAT Alinity with the ability to add more testing cartridges and capabilities over time, such as ones that may be developed in the future, like [those] for concussion, blood clotting, or molecular tests, allowing healthcare providers to conduct an even broader range of important tests with one handheld platform.”
Abbott currently is working on one such future assay, having charged more than 120 of its scientists to develop a brain injury blood test for the Alinity. The test would measure levels of glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase L1 (UCH-L1), biomarkers typically released from the brain into the bloodstream after a head injury. Plasma concentrations of these proteins can indicate whether patients with head injuries suffer from intracranial lesions (brain bleeds).
Currently, intracranial lesions are detectable only through computed tomography (CT) scans—effective yet costly procedures that produce potentially harmful ionizing radiation. Blood tests like the FDA-cleared Brain Trauma Indicator from Alachua, Fla.-based Banyan Biomarkers Inc. certainly can help reduce healthcare costs and radiation exposure risk, but they are not without their own limitations. Case in point: Banyan’s test is nearly 99 percent accurate but must be administered within the first 12 hours after injury, and results take three to four hours.
Contrarily, Abbott’s proposed solution would produce results within minutes. Ironically, Abbott has enlisted Banyan’s help with its solution, licensing the smaller company’s traumatic brain injury (TBI) blood biomarkers—UCH-L1 and GFAP—for use on its core laboratory instruments. The U.S. Department of Defense (DoD) and the Transforming Research and Clinical Knowledge in Traumatic Brain Injury (TRACK-TBI) Network are lending support as well: Both organizations have partnered with Abbott on a clinical trial to evaluate the efficacy of the company’s POC blood testing technology. As part of the trial, researchers will evaluate suspected TBI patients within 24 hours of injury and compare their blood test results against traditional clinical assessments, CT scans, magnetic resonance imaging scans, and clinical outcomes.
“Whether on the battlefield or in the emergency room, we need quick and accurate information to help assess a person who may have sustained brain injury,” Geoffrey T. Manley, M.D., Ph.D., principal investigator of TRACK-TBI, neurosurgeon and neurosurgery professor at the University of California-San Francisco, said upon announcing the network’s partnership with Abbott in late April. “Our goal with this partnership is to validate the scientific rigor behind new technologies like this blood test, and how they can help ensure the best care for our troops and patients.”
Those validation efforts are a bit belated, though. Boston-based BioDirection Inc. already has proven the efficacy of its Tbit POC blood test for concussion diagnosis and earlier this year secured FDA breakthrough device designation for the prediction of positive CT scans after TBI. Like Abbott, BioDirection’s assay measures protein biomarker levels after head trauma, but calibrates the amount through patented biosensing nanowire technology. The company claims its system is highly sensitive and can deliver objective results within 90 seconds from a single drop of blood.
A preclinical study completed in 2017 concluded the Tbit System provides actionable outcomes in POC settings and can potentially reduce the number of unnecessary CT scans by more than 40 percent. The trial evaluated the Tbit System’s ability to screen TBI patients for a CT positive or negative test; results showed a 100 percent sensitivity with no false negative results, and a 41 percent specificity level.
“Blunt trauma injuries that impact the head and brain require rapid identification of comorbidities to rule out or confirm the potential of intracranial hemorrhage that may require some form of surgical intervention,” Brian McGlynn, founder and chief technical officer of BioDirection, said when the Tbit System received its Breakthrough Device Designation. “The Tbit System has the potential to deliver actionable information to the physician where minutes matter. Longer term, our technology has the potential to support a full continuum of care ranging from stratification of injury, prognosis and return to play and activity.”
Indeed, injury classification, rapid diagnosis, and lifestyle maintenance wield major clout in the POC product development arena, as they help empower real-time caregiver collaboration, improve treatment access, and enrich patients’ overall experience. These Muses also can engender optimized workflows, better healthcare delivery accountability/transparency, and lower costs—factors that are key to survival (and success) in today’s value-based ecosystem.
POC technologies reduce healthcare expenditures in various ways, the most obvious of which is speedy diagnoses. Quick (sometimes instantaneous) results spare hospitals and clinics from incurring equipment expenses (no syringes, blood collection tubes needed), as well as transportation, storage, and separation (centrifuging) costs.
POC systems also can save healthcare dollars by alleviating the economic burden of disease and increasing patient survival rates in resource-poor areas. The Massachusetts Medical Device Development Center (M2D2) is addressing the former issue (disease burden), having created a new center to help U.S. entrepreneurs develop and commercialize POC technologies for heart, lung, blood, and sleep disorders; industry data show these conditions account for 41 percent of U.S. deaths and cost the nation’s healthcare system $400 billion annually.
Supported by a five-year, $7.9 million grant from the National Heart, Lung and Blood Institute, the new entity—appropriately dubbed the Center for Advancing Point-of-Care Technologies (CAPCaT)—will facilitate entrepreneurial partnerships between private industry and academia (UMass-Lowell, UMass Medical School). The center is one of four being launched with National Institutes of Health support to improve patient care and will provide inventors with both seed funding and access to numerous stakeholders, including patients, caregivers, and clinicians.
Examples of innovations to be developed at the center include portable devices and sensors to measure medication amounts in blood, products employing smart devices to record heart or respiratory rates, and technologies that alert patients to health changes such as worsening asthma. The center also seeks to support technologies that promote holistic methods to managing diseases, including devices that measure stress levels or promote mindfulness.
“The new business accelerator will facilitate collaborations between faculty researchers and private industry to commercialize new technologies more quickly and efficiently,” M2D2 Co-Director Nate Hafer, Ph.D., director of operations at UMass Center for Clinical and Translational Science at UMass Medical School, said in unveiling the CAPCaT initiative last fall. “The goal of these new tools is to help clinicians and healthcare systems deliver higher quality care at a lower cost.”
That’s quite a tall order, considering the two are still somewhat mutually exclusive. But better care certainly can help lower overall health expenses, particularly in remote areas of the world with limited access to medical testing and treatment.
In sub-Saharan Africa, for example, resources to diagnose and treat conditions like HIV, sickle cell disease, tuberculosis, hepatitis B, and emerging infections (Ebola, SARS) are virtually non-existent. POC technologies, however, can offer healthcare practitioners a relatively inexpensive way to screen and treat large swaths of underserved populations, thereby helping to mitigate global disease epidemics and ease the financial strain on government health systems.
Los Angeles-based diagnostics developer Silver Lake Research Corporation is targeting the sickle cell scourge with its HemoType SC, an assay that works similar to a pregnancy test. HemoType features test strips containing proteins that can detect abnormal amounts of hemoglobin polypeptides in whole blood. The test also can identify the sickle cell trait, which is present in people who inherit one hemoglobin S or C gene and one normal hemoglobin A gene.
Abbott, meanwhile, is focusing its disease control efforts on hepatitis B and HIV, recently winning CE mark and World Health Organization (WHO) sanctioning of POC diagnostic tests for both diseases. The company received CE mark approval in February for its Determine HBsAg 2, a rapid diagnostic test for hepatitis B virus detection in serum, plasma, or whole blood. Boasting an analytical sensitivity of 0.1 IU/mL, Determine HBsAg 2 verifies the presence of HBV infection in 15 minutes.
Abbott is confident its Determine BHsAG 2 assay will be a critical part of the global battle plan against hepatitis B. WHO has set baseline targets to diagnose 30 percent of HBV-infected individuals by 2020 and 90 percent by 2030.
The agency has set equally ambitious targets in its fight against HIV: It recommends those receiving antiretroviral therapy (ART) undergo a viral load test at six and 12 months, and annually thereafter to monitor for treatment failure. Such monitoring is difficult for people in developing countries or remote areas, though.
Abbott, however, is attempting to increase access to ART monitoring through its CE-marked m-PIMA HIV-1/2 VL, a portable test designed specifically for use in “resource-limited settings.” Granted Prequalification approval by WHO in early May, the assay is a quantitative nucleic acid amplification test for viral load measurement of HIV type 1 groups M/N and O, and HIV-2 in plasma samples. The test produces results in 70 minutes, which allows for immediate treatment decisions and reduces the number of patients lost to follow-up, according to Abbott.
“In vitro and point-of-care diagnostic tests are becoming more critical to the healthcare space generally. The developing world and the need for lower-cost [diagnostic] tests are going to drive the market,” noted David Franta, leader of the Microfluidics Global Business in 3M’s Medical Solutions Division. “Molecular diagnostic testing is of particular interest right now. It has quite a bit of capability from a specificity standpoint and sensitivity that other tests don’t, so there’s a lot of people looking into molecular testing and saying, ‘That may be an area where there will be some significant growth.’ Molecular tests can include infectious disease testing such as flu A/B or other pathogens—there is a big market already there. With molecular diagnostics, the technology can be applied in multiple places, and depending on the probes you are looking to develop, it can be applied to many different disease states.”
Molecular diagnostics technology can be applied in various ways, too. One approach entails microfluidics— the study of fluid manipulation in “microchannels.” This discipline increasingly is being summoned for POC innovations due largely to its portability, low sample and reagent consumption, high throughput and sensitivity, large surface-to-volume ratio, and other miniaturization-related benefits.
Some of the more predominant POC microfluidic applications are lab-on-a-chip/lab-on-a-disc and blood glucose test strips. Though various types of strips have been developed over the last several decades, they all work the same way: Blood reacts with an enzyme (glucose oxidase) on the strip to produce gluconic acid as the glucometer sends a current through the portion of the strip attached to the device. Using the electric terminals on the strip, the meter measures the current; the amount of gluconic acid produced from blood glucose determines the amount of current generated between the terminals. The meter then uses an algorithm to calculate the blood glucose level based on the difference in current.
Microfluidic test strips and cartridges must be precisely manufactured and cost-efficient to compete and survive in the market. The largest expenses for microfluidic technology are material and machining costs.
Preco Inc. is reducing machining costs through its FlexStacker, a custom-build piece of equipment introduced last year that laminates and assembles multiple layers to create an aligned, precisely registered and stacked microfluidics testing device (lab-on-a-chip). The system removes any discrepancies encountered in hand or blind lamination, and removes many of the necessary steps involved in single robot stacking.
“With four stack-up stations, an inline press, and three cameras per placement head, the FlexStacker was specifically designed with microfluidics in mind,” explained Kim Hill and Terry Rusch, director of medical sales and senior project engineer, respectively, at Preco, a Lenexa, Kan.-based contract manufacturing service provider offering laser cutting, scoring, perforating, welding, cladding, heat treating, and die cutting services.
“Cards can be fully assembled in an enclosed clean environment via four different robotic placement heads. Each of these placement heads has three cameras to achieve the best alignment possible between layers,” the pair continued. “If the card is larger then four layers, we simply assemble the first four layers and run it through the FlexStacker as needed until all the needed layers are assembled. Cost efficiencies are a major factor and is equal in importance to performance requirements.”
Early design input is just as important, though, as it can help accelerate time to market and provide device manufacturers with new ways to treat age-old health issues.
“With some of the leading-edge device designs, the sooner that 3M is involved in the process, the better the outcome could be,” 3M’s Franta advised. “A lot of times a customer may have our material and has created a device that works, but if they involve us earlier in the process we may help with differentiated solutions. Early on in the development process is key, because there’s usually more than one way to solve a problem. 3M can work with a designer to either come up with work-arounds to make a device perform, or come up with a novel idea that is a differentiated solution and could help improve the performance or reduce costs.”
For more than four decades, he’s walked the delicate line between love and hate, acting as the yin to runners’ yang, the moon to their sun. He’s friend and foe wrapped in one—a prize to some, a persona non grata to others.
Troyanos, 62, owns Sports Medicine Consultants Inc., a 23-year-old Plymouth, Mass.-based firm that provides medical management, onsite medical care, and volunteer recruitment services for large-scale athletic events. A certified athletic trainer, Troyanos has coordinated and supervised medical coverage for dozens of sizable races throughout the United States and Canada, including the grueling 26.2-mile Boston Marathon.
Coordinating medical coverage for New England’s most widely viewed sporting event is quite the undertaking: Besides preparing for almost any running-related injury (shattered femurs, included) Troyanos also supervises 1,700 volunteer clinicians and the 28 medical tents along the eight-town route. The job requires some mastery of psychology as well, since no sprinter really wants to see Troyanos on race day.
“Runners need to understand what their limitations are and not try to push them,” Troyanos told Boston Magazine last month. “Boston has a tendency for people to want to do their best. We get that. But if you’re aware of your body’s limitations and how you’re feeling and don’t push that, the likelihood is you’re not going to see us in the medical tent.”
Sound advice for sure, but Troyanos will likely always have company in the tents, thanks to a bipolar Mother Nature and an incredibly strong human spirit. Both forces have detoured droves of marathoners in recent years, forcing them to retreat under heated blankets or in ice baths, or to rejuvenate themselves via Gatorade or a bag of potato chips.
This year’s casualty count was down from past races, despite the meteorological mayhem that ensued throughout the day (a bit of everything: clouds, wind, rain, lightning, sun). The Boston Athletic Association reported that 2,095 runners received medical attention along the route, with 950 of those participants undergoing treatment after the finish line and 77 runners eventually transported to area hospitals. Most of the injuries sustained during the race were minor (blisters, cramps, dehydration, hypothermia), though the sun’s unexpected appearance generated a rash of heatstroke victims within a two-and-a-half-hour period.
“[When the] sun came out, all of a sudden [there were] probably about 30 or 40 exertional heat strokes on the course and in this tent,” Troyanos divulged to WBZ-TV (CBS) after the April 15 race. “That’s pretty significant. Those are very serious illnesses. We handled them well, everyone is doing great.”
To ensure such a favorable prognosis, Troyanos used a handheld blood analyzation system from Abbott that produces lab-quality results within minutes from virtually any location. The company’s i-STAT technology measures 26 different analytes (substances in the body), including cardiac markers, gases, chemistries/electrolytes, coagulation, lactate, and hematology, from just two to three drops of blood.
First released in 1992, the i-STAT system is designed specifically for use at various points of care—from radiology and emergency departments to the intensive care unit and the operating room. More than 75,000 i-STAT systems are currently in operation, according to Abbott, with one in three U.S. hospitals employing the device to increase efficiency and quality of care.
The technology, however, has become fairly prevalent outside the hospital too—the paradigm shift to value-based healthcare has helped propel the i-STAT’s spread to outpatient clinics, doctor’s offices, long-term care facilities, emergency services vehicles, and rural institutions. The U.S. and Indian governments have used the device as well.
Athletic trainers and sporting event coordinators are perhaps the i-STAT system’s biggest fans, as the tool—evidenced by its presence at the Boston Marathon and numerous Abbott World Marathon Majors races—has helped legions of competitors return to play quicker than they would with conventional treatment.
“Without this portable tool, these runners showing signs of severe dehydration or hypernatremia would likely be transported to the hospital for diagnostic tests and follow up,” said Dr. Paul Jarvis, director of global medical affairs for Abbott’s point-of-care (POC) diagnostic business. “The fact that we can bring diagnostics right to the runner’s side is truly the power of [the i-STAT]. It’s wonderful for runners to have access to a tool like this that can help with very quick treatment. The power to finish the race is priceless. It’s a big win for runners.”
It’s also been a big win for Abbott, adding hundreds of millions of dollars to the multinational’s coffers and bolstering its share of the fast-growing global POC diagnostics/testing market, valued at $18.09 billion last year. Grand View Research data show this sector expanding 3.3 percent annually through 2025, with cancer marker testing growing at a higher pace over the forecast period due to a sharp rise in malignancies and the development of home testing kits.
Those same factors are expected to shape the POC diagnostics/testing market for years to come as an aging world population grapples with the various hellions of maturity—namely, cardiac disease, diabetes, Parkinson’s disease, and Alzheimer’s. Demand for home-based diagnostic devices and wearables as well as technological advancements that beget faster, easier-to-use products also are likely to drive market growth.
Such advancements are quickly coming to bear. Abbott, for example, has released newer, improved i-STAT models with Cloud connectivity, 2D barcode tracking ability, customizable range settings, and upgraded software. One of the versions—the i-STAT Alinity—was specifically designed for clinicians: The easy-grip handled tool offers on-screen guidance during the testing process and can identify both problems and potential solutions in real time; additionally, audio and light cues alert users to critical range test results that require immediate action. Moreover, the i-STAT Alinity is built for future sustainability.
“It was built to be ‘future-proofed,’ meaning it’s compatible with a variety of testing cartridges, including those designed for our current i-STAT platform, as well as tests that will be developed in the future,” Narendra Soman, Ph.D., R&D director for Abbott’s Point of Care Diagnostics business, noted in a company-sponsored Q&A about the Alinity. “In fact, we’ve designed i-STAT Alinity with the ability to add more testing cartridges and capabilities over time, such as ones that may be developed in the future, like [those] for concussion, blood clotting, or molecular tests, allowing healthcare providers to conduct an even broader range of important tests with one handheld platform.”
Abbott currently is working on one such future assay, having charged more than 120 of its scientists to develop a brain injury blood test for the Alinity. The test would measure levels of glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase L1 (UCH-L1), biomarkers typically released from the brain into the bloodstream after a head injury. Plasma concentrations of these proteins can indicate whether patients with head injuries suffer from intracranial lesions (brain bleeds).
Currently, intracranial lesions are detectable only through computed tomography (CT) scans—effective yet costly procedures that produce potentially harmful ionizing radiation. Blood tests like the FDA-cleared Brain Trauma Indicator from Alachua, Fla.-based Banyan Biomarkers Inc. certainly can help reduce healthcare costs and radiation exposure risk, but they are not without their own limitations. Case in point: Banyan’s test is nearly 99 percent accurate but must be administered within the first 12 hours after injury, and results take three to four hours.
Contrarily, Abbott’s proposed solution would produce results within minutes. Ironically, Abbott has enlisted Banyan’s help with its solution, licensing the smaller company’s traumatic brain injury (TBI) blood biomarkers—UCH-L1 and GFAP—for use on its core laboratory instruments. The U.S. Department of Defense (DoD) and the Transforming Research and Clinical Knowledge in Traumatic Brain Injury (TRACK-TBI) Network are lending support as well: Both organizations have partnered with Abbott on a clinical trial to evaluate the efficacy of the company’s POC blood testing technology. As part of the trial, researchers will evaluate suspected TBI patients within 24 hours of injury and compare their blood test results against traditional clinical assessments, CT scans, magnetic resonance imaging scans, and clinical outcomes.
“Whether on the battlefield or in the emergency room, we need quick and accurate information to help assess a person who may have sustained brain injury,” Geoffrey T. Manley, M.D., Ph.D., principal investigator of TRACK-TBI, neurosurgeon and neurosurgery professor at the University of California-San Francisco, said upon announcing the network’s partnership with Abbott in late April. “Our goal with this partnership is to validate the scientific rigor behind new technologies like this blood test, and how they can help ensure the best care for our troops and patients.”
Those validation efforts are a bit belated, though. Boston-based BioDirection Inc. already has proven the efficacy of its Tbit POC blood test for concussion diagnosis and earlier this year secured FDA breakthrough device designation for the prediction of positive CT scans after TBI. Like Abbott, BioDirection’s assay measures protein biomarker levels after head trauma, but calibrates the amount through patented biosensing nanowire technology. The company claims its system is highly sensitive and can deliver objective results within 90 seconds from a single drop of blood.
A preclinical study completed in 2017 concluded the Tbit System provides actionable outcomes in POC settings and can potentially reduce the number of unnecessary CT scans by more than 40 percent. The trial evaluated the Tbit System’s ability to screen TBI patients for a CT positive or negative test; results showed a 100 percent sensitivity with no false negative results, and a 41 percent specificity level.
“Blunt trauma injuries that impact the head and brain require rapid identification of comorbidities to rule out or confirm the potential of intracranial hemorrhage that may require some form of surgical intervention,” Brian McGlynn, founder and chief technical officer of BioDirection, said when the Tbit System received its Breakthrough Device Designation. “The Tbit System has the potential to deliver actionable information to the physician where minutes matter. Longer term, our technology has the potential to support a full continuum of care ranging from stratification of injury, prognosis and return to play and activity.”
Indeed, injury classification, rapid diagnosis, and lifestyle maintenance wield major clout in the POC product development arena, as they help empower real-time caregiver collaboration, improve treatment access, and enrich patients’ overall experience. These Muses also can engender optimized workflows, better healthcare delivery accountability/transparency, and lower costs—factors that are key to survival (and success) in today’s value-based ecosystem.
POC technologies reduce healthcare expenditures in various ways, the most obvious of which is speedy diagnoses. Quick (sometimes instantaneous) results spare hospitals and clinics from incurring equipment expenses (no syringes, blood collection tubes needed), as well as transportation, storage, and separation (centrifuging) costs.
POC systems also can save healthcare dollars by alleviating the economic burden of disease and increasing patient survival rates in resource-poor areas. The Massachusetts Medical Device Development Center (M2D2) is addressing the former issue (disease burden), having created a new center to help U.S. entrepreneurs develop and commercialize POC technologies for heart, lung, blood, and sleep disorders; industry data show these conditions account for 41 percent of U.S. deaths and cost the nation’s healthcare system $400 billion annually.
Supported by a five-year, $7.9 million grant from the National Heart, Lung and Blood Institute, the new entity—appropriately dubbed the Center for Advancing Point-of-Care Technologies (CAPCaT)—will facilitate entrepreneurial partnerships between private industry and academia (UMass-Lowell, UMass Medical School). The center is one of four being launched with National Institutes of Health support to improve patient care and will provide inventors with both seed funding and access to numerous stakeholders, including patients, caregivers, and clinicians.
Examples of innovations to be developed at the center include portable devices and sensors to measure medication amounts in blood, products employing smart devices to record heart or respiratory rates, and technologies that alert patients to health changes such as worsening asthma. The center also seeks to support technologies that promote holistic methods to managing diseases, including devices that measure stress levels or promote mindfulness.
“The new business accelerator will facilitate collaborations between faculty researchers and private industry to commercialize new technologies more quickly and efficiently,” M2D2 Co-Director Nate Hafer, Ph.D., director of operations at UMass Center for Clinical and Translational Science at UMass Medical School, said in unveiling the CAPCaT initiative last fall. “The goal of these new tools is to help clinicians and healthcare systems deliver higher quality care at a lower cost.”
That’s quite a tall order, considering the two are still somewhat mutually exclusive. But better care certainly can help lower overall health expenses, particularly in remote areas of the world with limited access to medical testing and treatment.
In sub-Saharan Africa, for example, resources to diagnose and treat conditions like HIV, sickle cell disease, tuberculosis, hepatitis B, and emerging infections (Ebola, SARS) are virtually non-existent. POC technologies, however, can offer healthcare practitioners a relatively inexpensive way to screen and treat large swaths of underserved populations, thereby helping to mitigate global disease epidemics and ease the financial strain on government health systems.
Los Angeles-based diagnostics developer Silver Lake Research Corporation is targeting the sickle cell scourge with its HemoType SC, an assay that works similar to a pregnancy test. HemoType features test strips containing proteins that can detect abnormal amounts of hemoglobin polypeptides in whole blood. The test also can identify the sickle cell trait, which is present in people who inherit one hemoglobin S or C gene and one normal hemoglobin A gene.
Abbott, meanwhile, is focusing its disease control efforts on hepatitis B and HIV, recently winning CE mark and World Health Organization (WHO) sanctioning of POC diagnostic tests for both diseases. The company received CE mark approval in February for its Determine HBsAg 2, a rapid diagnostic test for hepatitis B virus detection in serum, plasma, or whole blood. Boasting an analytical sensitivity of 0.1 IU/mL, Determine HBsAg 2 verifies the presence of HBV infection in 15 minutes.
Abbott is confident its Determine BHsAG 2 assay will be a critical part of the global battle plan against hepatitis B. WHO has set baseline targets to diagnose 30 percent of HBV-infected individuals by 2020 and 90 percent by 2030.
The agency has set equally ambitious targets in its fight against HIV: It recommends those receiving antiretroviral therapy (ART) undergo a viral load test at six and 12 months, and annually thereafter to monitor for treatment failure. Such monitoring is difficult for people in developing countries or remote areas, though.
Abbott, however, is attempting to increase access to ART monitoring through its CE-marked m-PIMA HIV-1/2 VL, a portable test designed specifically for use in “resource-limited settings.” Granted Prequalification approval by WHO in early May, the assay is a quantitative nucleic acid amplification test for viral load measurement of HIV type 1 groups M/N and O, and HIV-2 in plasma samples. The test produces results in 70 minutes, which allows for immediate treatment decisions and reduces the number of patients lost to follow-up, according to Abbott.
“In vitro and point-of-care diagnostic tests are becoming more critical to the healthcare space generally. The developing world and the need for lower-cost [diagnostic] tests are going to drive the market,” noted David Franta, leader of the Microfluidics Global Business in 3M’s Medical Solutions Division. “Molecular diagnostic testing is of particular interest right now. It has quite a bit of capability from a specificity standpoint and sensitivity that other tests don’t, so there’s a lot of people looking into molecular testing and saying, ‘That may be an area where there will be some significant growth.’ Molecular tests can include infectious disease testing such as flu A/B or other pathogens—there is a big market already there. With molecular diagnostics, the technology can be applied in multiple places, and depending on the probes you are looking to develop, it can be applied to many different disease states.”
Molecular diagnostics technology can be applied in various ways, too. One approach entails microfluidics— the study of fluid manipulation in “microchannels.” This discipline increasingly is being summoned for POC innovations due largely to its portability, low sample and reagent consumption, high throughput and sensitivity, large surface-to-volume ratio, and other miniaturization-related benefits.
Some of the more predominant POC microfluidic applications are lab-on-a-chip/lab-on-a-disc and blood glucose test strips. Though various types of strips have been developed over the last several decades, they all work the same way: Blood reacts with an enzyme (glucose oxidase) on the strip to produce gluconic acid as the glucometer sends a current through the portion of the strip attached to the device. Using the electric terminals on the strip, the meter measures the current; the amount of gluconic acid produced from blood glucose determines the amount of current generated between the terminals. The meter then uses an algorithm to calculate the blood glucose level based on the difference in current.
Microfluidic test strips and cartridges must be precisely manufactured and cost-efficient to compete and survive in the market. The largest expenses for microfluidic technology are material and machining costs.
Preco Inc. is reducing machining costs through its FlexStacker, a custom-build piece of equipment introduced last year that laminates and assembles multiple layers to create an aligned, precisely registered and stacked microfluidics testing device (lab-on-a-chip). The system removes any discrepancies encountered in hand or blind lamination, and removes many of the necessary steps involved in single robot stacking.
“With four stack-up stations, an inline press, and three cameras per placement head, the FlexStacker was specifically designed with microfluidics in mind,” explained Kim Hill and Terry Rusch, director of medical sales and senior project engineer, respectively, at Preco, a Lenexa, Kan.-based contract manufacturing service provider offering laser cutting, scoring, perforating, welding, cladding, heat treating, and die cutting services.
“Cards can be fully assembled in an enclosed clean environment via four different robotic placement heads. Each of these placement heads has three cameras to achieve the best alignment possible between layers,” the pair continued. “If the card is larger then four layers, we simply assemble the first four layers and run it through the FlexStacker as needed until all the needed layers are assembled. Cost efficiencies are a major factor and is equal in importance to performance requirements.”
Early design input is just as important, though, as it can help accelerate time to market and provide device manufacturers with new ways to treat age-old health issues.
“With some of the leading-edge device designs, the sooner that 3M is involved in the process, the better the outcome could be,” 3M’s Franta advised. “A lot of times a customer may have our material and has created a device that works, but if they involve us earlier in the process we may help with differentiated solutions. Early on in the development process is key, because there’s usually more than one way to solve a problem. 3M can work with a designer to either come up with work-arounds to make a device perform, or come up with a novel idea that is a differentiated solution and could help improve the performance or reduce costs.”