Maria Shepherd, President and Founder, Medi-Vantage10.12.16
For those of us in the medtech space, the Comprehensive Care for Joint Replacement Model1 (CJR) is a sneak preview of what CMS has in store for all of us. CMS has developed this model to provide improved and efficient care for Medicare beneficiaries receiving common (and expensive) inpatient surgeries. Starting with hip and knee joint replacements, CMS is testing bundled payments and quality measurements for the entire episode of care associated with these procedures. The goal is to persuade hospitals, clinicians, and post-procedure care providers to align care and improve the coordination and quality of treatment patients receive—from pre-procedure through recovery.
Why This Is Important
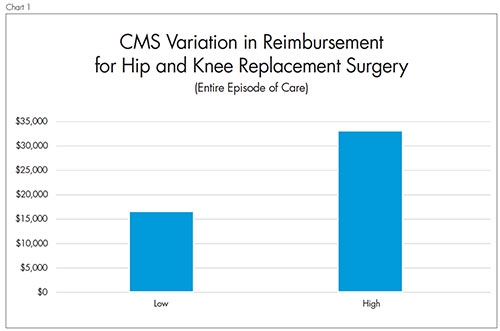
The CJR is mandatory for approximately 800 hospitals in 67 urban geographic areas of the United States.2 CMS chose hip and knee joint replacements because they are the most common inpatient surgeries for CMS beneficiaries and frequently entail lengthy recovery and rehabilitation. CMS reports that in 2014, there were more than 400,000 hip and knee joint replacement procedures. The bill for the 400,000 hospital stays cost CMS more than $7 billion. Worse, the quality and cost of care varied greatly among healthcare providers. The rate of complications such as infections or implant failures post-surgery can be three times greater at some hospitals, increasing the chance that the patient will be readmitted. Average CMS spending for surgery, hospitalization, and recovery ranges from $16,500 to $33,000 across the United States (Chart 1).
Who’s Next?
CJR hospitals are now reimbursed a set amount for the entire 90-day episode of care. Next in line for bundled payments are heart attacks and bypass surgery (and stroke treatments associated with these procedures), because these diseases cause one in three deaths and result in more than $300 billion in healthcare costs each year.3
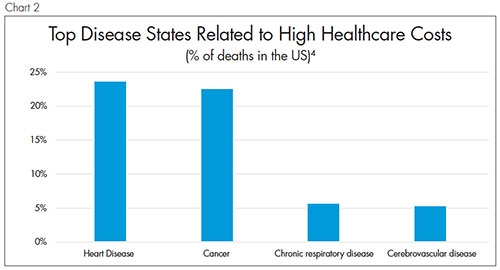
After that, expect CMS to move on to procedures related to the top causes of death (and high healthcare costs) per year (Chart 2).
What Can a Medical Device Company Do?
Quite a bit, actually. Many medtech companies are responding with powerful programs, and not just in the orthopedic space.
In 2009, Stryker Corp. assessed the risk and created an evolving program called Performance Solutions that offers predictive analytics, episode-of-care performance management tools, and other services to help healthcare providers meet CMS expectations and reduce the cost of care. Medtronic plc launched the Diabetes Service and Solutions Business Unit to optimize clinical outcomes and enhance clinician and patient experience with its products. Medtronic is focused on innovation in products like infusion sets to transform how data is collected, analyzed, and presented through the Medtronic CareLink system.
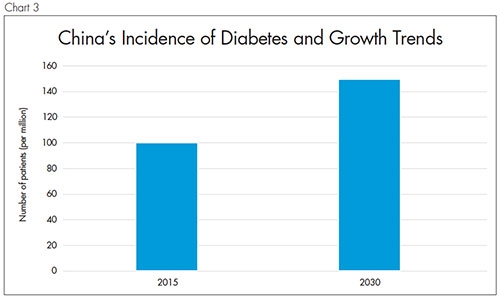
The reason Medtronic is investing so much in this space is demonstrated in Chart 3. It is estimated that in 2015, in China alone, 100 million patients suffered from diabetes. For the sickest patients, there were 1.5 million deaths—the highest number of diabetes cases and mortality in any country in the world. It is predicted that the number of patients will to rise to 150 million by 2030.4
What Is Your Company Doing to Respond to the CMS Challenge?
There is no doubt about it. With the exception of truly innovative technology and very small disease states, CMS will be developing bundled payments for all of its Medicare beneficiaries. Its goal is to have 50 percent of traditional Medicare payments flowing through these payment models by 2018. What services can your company provide that will help your providers meet this challenge and help your organization stay relevant?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Data Decision Group, now re-branded as Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses, and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 ext. 102 or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com
Why This Is Important
The CJR is mandatory for approximately 800 hospitals in 67 urban geographic areas of the United States.2 CMS chose hip and knee joint replacements because they are the most common inpatient surgeries for CMS beneficiaries and frequently entail lengthy recovery and rehabilitation. CMS reports that in 2014, there were more than 400,000 hip and knee joint replacement procedures. The bill for the 400,000 hospital stays cost CMS more than $7 billion. Worse, the quality and cost of care varied greatly among healthcare providers. The rate of complications such as infections or implant failures post-surgery can be three times greater at some hospitals, increasing the chance that the patient will be readmitted. Average CMS spending for surgery, hospitalization, and recovery ranges from $16,500 to $33,000 across the United States (Chart 1).

Who’s Next?
CJR hospitals are now reimbursed a set amount for the entire 90-day episode of care. Next in line for bundled payments are heart attacks and bypass surgery (and stroke treatments associated with these procedures), because these diseases cause one in three deaths and result in more than $300 billion in healthcare costs each year.3
After that, expect CMS to move on to procedures related to the top causes of death (and high healthcare costs) per year (Chart 2).

What Can a Medical Device Company Do?
Quite a bit, actually. Many medtech companies are responding with powerful programs, and not just in the orthopedic space.
In 2009, Stryker Corp. assessed the risk and created an evolving program called Performance Solutions that offers predictive analytics, episode-of-care performance management tools, and other services to help healthcare providers meet CMS expectations and reduce the cost of care. Medtronic plc launched the Diabetes Service and Solutions Business Unit to optimize clinical outcomes and enhance clinician and patient experience with its products. Medtronic is focused on innovation in products like infusion sets to transform how data is collected, analyzed, and presented through the Medtronic CareLink system.
The reason Medtronic is investing so much in this space is demonstrated in Chart 3. It is estimated that in 2015, in China alone, 100 million patients suffered from diabetes. For the sickest patients, there were 1.5 million deaths—the highest number of diabetes cases and mortality in any country in the world. It is predicted that the number of patients will to rise to 150 million by 2030.4

What Is Your Company Doing to Respond to the CMS Challenge?
There is no doubt about it. With the exception of truly innovative technology and very small disease states, CMS will be developing bundled payments for all of its Medicare beneficiaries. Its goal is to have 50 percent of traditional Medicare payments flowing through these payment models by 2018. What services can your company provide that will help your providers meet this challenge and help your organization stay relevant?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Data Decision Group, now re-branded as Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses, and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 ext. 102 or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com