Mark Paulsen, Contributing Writer02.03.16
The costs of hospital-acquired infection (HAI) are staggering. A 2009 report1 published by the Centers for Disease Control (CDC) estimated the annual national direct medical costs of HAI to range between $28.4 billion and $45 billion, and notes that approximately 20 percent of all HAIs are preventable based on current medical technology. The CDC report, citing previous studies, distinguishes device-related infection (DRI) from other vectors, noting for example, that 37 percent of bloodstream infections and 80 percent of urinary tract infections are device related. Another report2 notes more generally that more than half of HAIs are attributed to medical devices such as central venous catheters, endotracheal tubes, bladder catheters, and various surgical implants.
Silicone elastomers are in many ways an ideal biomaterial. These elastomers can be fabricated using standardized processes to produce tubing, coatings, thin membranes, and various other types of molded components. Silicones enjoy a 60-year history as an implant material and represent the gold standard for biocompatibility and biostability.
Silicones are not, however, immune to bacterial colonization. Like other hydrophobic materials, the surface of silicone medical devices allow attachment and adhesion of various micoorganisms.3 Various techniques have been investigated to minimize the attachment of bacteria to the surface of silicone medical devices and hence to reduce the incidence of surgical site (SSI) and bloodstream infection (BSI). Coatings, often containing silver-based compounds, have shown to be effective at reducing the population of certain types of bacteria. Coating processes have several drawbacks, though. The process often involves costly automated spraying or dipping equipment; analytical techniques must be developed to measure coat weight uniformity; the durability of the coating and adhesion to the silicone substrate must be proven under different conditions; and the antimicrobial efficacy of the thin coating is often of limited duration. Additionally, in the case of catheters, most coating processes cannot treat the surface of the inner lumen.
A second approach involves the addition of antibiotic active pharmaceutical ingredients (API) to silicone raw materials. Specialty Silicone Fabricators’ (SSF) lab has previously worked with several antibiotic drugs including chlorhexidine, gentamycin, xifaxin and doxycycline. In all cases, the drugs, in powder form, were combined with different silicones using numerous types of mixing equipment. After homogenization, these silicone-drug mixtures can be formed into desired shapes and vulcanized using various fabrication processes.
But this process also suffers limitations. It is known that some API will inhibit or poison the cure system of certain silicones. Also, many drugs are unstable at the elevated temperatures required for the cost-effective vulcanization of silicone components. The development of robust mixing processes often involve numerous trial iterations using highly specialized equipment. Analytical test methods must be developed to confirm the homogeneity of the drug within a mix-batch. The qualification and validation of mixing processes is performed using production-ready equipment as batch sizes are typically large and drug costs can be considerable. The particle size of the drug powder is an important variable as it affects release kinetics from the finished component. Particle size, therefore, must be maintained within a narrow window despite the tendency of many drug powders to agglomerate during storage.
Finally, post-manufacturing cleaning processes must be developed and validated to ensure that drug-contacting surfaces of both mixing and fabrication equipment are within API carry-over limits.
The work described below provides an alternative approach to manufacturing antimicrobial silicone device components that offers considerable advantages over traditional processes.
Study Methodology
The vast majority of silicone medical components are manufactured from raw material formulations containing polydimethylsiloxane (PDMS) polymers reinforced with amorphous non-crystalline silica. It is known that vulcanized PDMS elastomers easily swell when immersed in various organic solvents.
SSF’s study investigated the simultaneous transfer of two drugs from solution into pre-vulcanized silicone rubber components. The two antibiotic API investigated were Clindamycin Hydrochloride (CLIN)—mol weight = 461.45 g/mol—and Rifampicin (RIF)—mol weight = 822.94 g/mol. Study variables included 1) the total concentration of the drugs in solution; 2) the ratio of the drugs in solution; 3) immersion time of the silicone component; and 4) the durometer of the silicone test article.
Effect of Solution Concentration on Drug Impregnation
CLIN and RIF were added to chloroform to produce three unique solutions. The CLIN/RIF ratio was held constant but the overall concentration of the two drugs in solution was varied from x to 2x to 10x. Six-inch lengths of silicone tubing (50 durometer Shore A) were immersed in the solutions. Tubes were removed from solution at predetermined intervals; immersion times ranged from a few minutes to several hours. The impregnated tubing was then blotted dry and the inner lumen flushed with IPA. The test articles were then allowed to devolatilize for 24 hours.
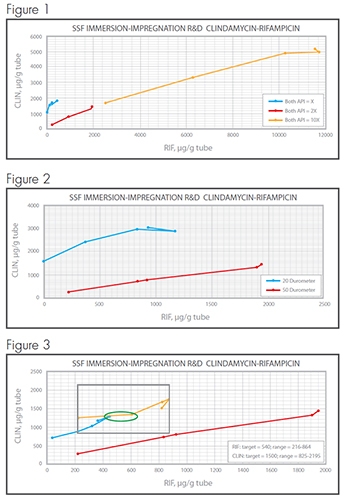
SSF’s analytical lab developed methodology to extract both drugs from the silicone test articles as well as HPLC test methods to quantify drug concentrations. Small sections of tubes were weighed and extracted. The extract was then evaluated via HPLC to determine the masses of both drugs within a known mass of tubing. Results for the drug content testing of both CLIN and RIF are expressed in the units of micrograms of drug per gram of tube. Data for tubing immersed in the three different solutions is summarized in Figure 1.
Five data points, representing different immersion times, are plotted for each of the three solutions. In general, as the concentration of API in solution increased, so too did the resultant concentration within the silicone test article. It’s also evident from the plotted data that the rate of impregnation is strongly influenced by the concentration of the immersion solution. Finally, the lines representing the three data sets have similar slopes, indicating that the ratio of the two drugs impregnated within the silicone is dependent on the ratio of the drugs in solution and largely independent of the overall solution concentration.
Impact of Silicone on Drug Impregnation
A second experiment investigated the impact of elastomer formulation on both the mass transfer of the drugs from solution as well as the rate of impregnation. CLIN and RIF were added to chloroform to produce a 2x solution identical to that discussed in the previous test. Short lengths of 20 and 80 durometer tubing were immersed and then removed at the same five-time intervals as the prior experiment. Tubing segments were again assayed for total drug content.
Durometer is a measure of hardness. In general, in silica-reinforced systems such as the materials used in this study, an increase in durometer is mirrored by an increase in the silica content of the rubber. Approximate densities of silica and PDMS polymers are 2.2 and 1.0 g/cc, respectively. The specific gravity values for silicone elastomers provides a convenient means for quantifying and comparing the silica-polymer ratio between different rubbers. Specific gravities of the 20, 50, and 80 durometer tubes investigated here were 1.10, 1.16, and 1.21, respectively. In this regard, durometer was used as a proxy for polymer concentration within the rubber.
SSF study investigators hypothesized that the quasi-organic PDMS polymer, relative to the inorganic silica, would be more readily swelled by halogenated-organic solvents and the mass and rate of drug transfer would therefore increase as durometer decreased.
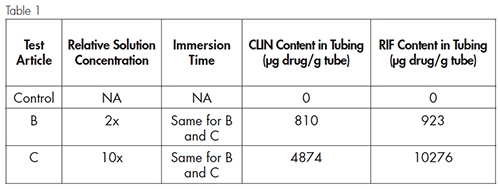
Results are summarized in Figure 2. Twenty durometer tubes, with high polymer content, absorbed more Clindamycin and at a faster rate than the 50 durometer tubes containing less polymer. Somewhat surprisingly, the mass and impregnation rate of rifampicin in the 20 durometer tubes was lower than that observed in the 50 durometer samples. Both API were below test detection limits in the 80 durometer tubes.
Varying API Ratios, Impact on Impregnation
A third experiment investigated how changes in the ratio of the two drugs in solution impacted drug concentrations in the silicone elastomer.
It is known that some types of ventricular catheters, a key component of life-saving hydrocephalus shunts, contain CLIN and RIF, and that studies have confirmed the antimicrobial efficacy of these devices. The literature does not address the manufacturing method used to produce these products but it does provide detailed information about the target concentrations of both drugs as well as specification ranges. Labeling for the devices notes CLIN and RIF concentrations of 0.15 percent (wt/wt) and 0.054 percent, respectively. Supporting literature discusses a specification ranges of ±45 percent for CLIN and 60 percent for RIF. These drug concentrations and ranges were used as targets in this experiment.
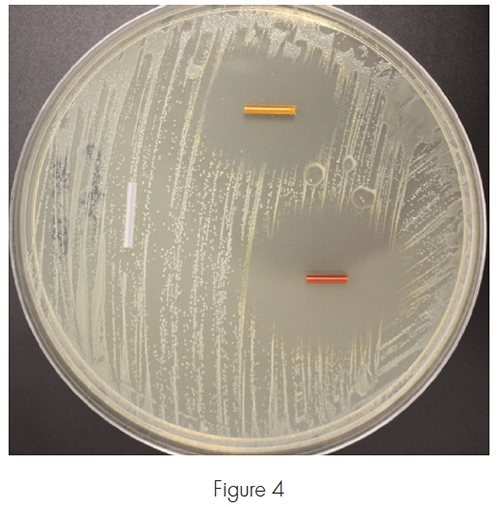
Several solutions were prepared in which the concentrations of CLIN and RIF were adjusted from the 1:1 ratio used in the previous experiments. As in the earlier tests, a number of immersion times were investigated for each of the solutions. Results are shown in Figure 3. The target concentration for both API is shown here as the white circle while the specification ranges for the drugs (RIF = 216-864, CLIN = 825-2,195) are identified as the area within the white box. Experimental results showed that several immersion times in two different solutions resulted in tubing drug concentrations that were within the specification range and that two data points (circled green) were very close to the published API target of the predicate devices.
Bacterial Challenge—Antimicrobial Efficacy Confirmed
The immersion experiments discussed previously demonstrated conclusively the mass transfer of both drugs from solution to the silicone tubing. Additionally, it was shown that the masses of drugs impregnated within the tubing were dependent upon controllable factors including solution concentration, immersion duration, and silicone type.
Importantly, though, the correlation between drug contents within the silicone and antimicrobial efficacy remained undetermined. SSF study investigators commissioned a Kirby-Bauer Zone of Inhibition (ZOI) test to assess the impact of drug content on the gram-positive coccal bacterium, Staphylococcus aureus. Staph aureus is responsible for particularly high incidence rates of HAI and DRI. In a technical brief,4 the National Institute of Allergy and Infectious Disease (NIAID) notes that each year an estimated 500,000 patients contract staph infections in American hospitals and that “S. aureus is the most common cause of HAI.” Other researchers note that “infections associated with indwelling medical devices ranging from simple intravenous catheters to prosthetic joints and replacement heart valves can be caused by S. aureus.”5
Test articles were chosen so that antimicrobial efficacy could be evaluated across a range of samples produced under varying conditions and containing different drug concentrations. Immersion conditions and drug content are summarized in Table 1.
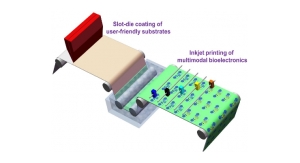
The three test articles were placed on an agar plate that had been inoculated with a suspension containing Staphylococcus aureas. The plate was photographed after a 24 hour incubation period and is depicted in Figure 4.
The drug-impregnated tubing had a powerful inhibitory effect on the growth of S. aureus and clear zones of inhibition are seen surrounding test articles B and C. As expected, the higher drug concentrations in sample C produced a larger ZOI (18 mm) than sample B (14 mm), which contained lower concentrations of both drugs. No ZOI was observed around the control sample.
Discussion
Attractive forces between silicone polymer chains are quite weak. This contributes to the characteristic high free volume of silicone elastomers and their exceptional permeability. In turn, the high permeability of silicone rubber makes this biomaterial especially attractive as a matrix for drug-device combination products. As noted, the process for manufacturing most silicone drug-device components involves adding a drug powder to a silicone raw material, forming the mixture into a desired shape, then vulcanizing at elevated temperatures. But as discussed, there are several inherent concerns with this process that add complexity and costs.
Advantages of Immersion/Impregnation
The immersion/impregnation processes presented in these experiments offer several important advantages over current silicone drug-device manufacturing methods. These advantages include the following:
Healthcare costs associated with fighting device related infections are significant. The use of medical devices, including many life-saving devices made from silicone, will only accelerate as the world’s population ages. Additionally, recent changes to hospital reimbursement policy for some types of device-related infections have prompted medical device manufacturers to differentiate their products by including additives known to resist bacterial colonization. While it is doubtful that device-related infection can be completely eliminated, various antimicrobial strategies can effectively reduce the incidence of infection.
The experiments described in this analysis presented a cost-effective process for the impregnation of drugs within prefabricated silicone components. It was demonstrated that variables such as the concentration of drugs in solution, immersion duration and the type of silicone will impact the mass transfer of drugs. It is important to note, however, that while this study investigated chloroform solutions containing clindamycin hydrochloride and rifampicin, the process as described provides a broad and important platform suitable for numerous other antibiotic drugs and active pharmaceutical ingredients.
References:
Mark Paulsen is president and founder of Silicone Consulting, LLC. He has held key positions within various sectors of the silicone medical device industry including raw material manufacturing (NuSil Technology, director of Healthcare Materials, 1992-2007) and component fabrication (SSF, director of Business Development, 2008-2013). Paulsen consults widely within the medical device industry on topics such as silicone chemistry, material selection, process optimization, and drug-device design and testing. Paulsen holds three patents for the development of a novel silicone elastomer used as insulation on cardiac and neurological stimulation devices. In 2004, he was recognized as one of the top 100 Notable People in the medical device industry. Paulsen studied chemistry at Harvard University and currently lives in Santa Barbara, Calif.
For more than 30 years Specialty Silicone Fabricators (SSF) has provided customized manufacturing solutions to the medical device industry. The company maintains three manufacturing sites and employs nearly 250 people. SSF has been awarded six patents for its developments in silicone extrusion and thin film processing. Additional services include micromolding, coating expertise, device assembly, and final package design and validation. In 2009, SSF received federal registration and state licensing to begin working with active pharmaceutical ingredients.
Silicone elastomers are in many ways an ideal biomaterial. These elastomers can be fabricated using standardized processes to produce tubing, coatings, thin membranes, and various other types of molded components. Silicones enjoy a 60-year history as an implant material and represent the gold standard for biocompatibility and biostability.
Silicones are not, however, immune to bacterial colonization. Like other hydrophobic materials, the surface of silicone medical devices allow attachment and adhesion of various micoorganisms.3 Various techniques have been investigated to minimize the attachment of bacteria to the surface of silicone medical devices and hence to reduce the incidence of surgical site (SSI) and bloodstream infection (BSI). Coatings, often containing silver-based compounds, have shown to be effective at reducing the population of certain types of bacteria. Coating processes have several drawbacks, though. The process often involves costly automated spraying or dipping equipment; analytical techniques must be developed to measure coat weight uniformity; the durability of the coating and adhesion to the silicone substrate must be proven under different conditions; and the antimicrobial efficacy of the thin coating is often of limited duration. Additionally, in the case of catheters, most coating processes cannot treat the surface of the inner lumen.
A second approach involves the addition of antibiotic active pharmaceutical ingredients (API) to silicone raw materials. Specialty Silicone Fabricators’ (SSF) lab has previously worked with several antibiotic drugs including chlorhexidine, gentamycin, xifaxin and doxycycline. In all cases, the drugs, in powder form, were combined with different silicones using numerous types of mixing equipment. After homogenization, these silicone-drug mixtures can be formed into desired shapes and vulcanized using various fabrication processes.
But this process also suffers limitations. It is known that some API will inhibit or poison the cure system of certain silicones. Also, many drugs are unstable at the elevated temperatures required for the cost-effective vulcanization of silicone components. The development of robust mixing processes often involve numerous trial iterations using highly specialized equipment. Analytical test methods must be developed to confirm the homogeneity of the drug within a mix-batch. The qualification and validation of mixing processes is performed using production-ready equipment as batch sizes are typically large and drug costs can be considerable. The particle size of the drug powder is an important variable as it affects release kinetics from the finished component. Particle size, therefore, must be maintained within a narrow window despite the tendency of many drug powders to agglomerate during storage.
Finally, post-manufacturing cleaning processes must be developed and validated to ensure that drug-contacting surfaces of both mixing and fabrication equipment are within API carry-over limits.
The work described below provides an alternative approach to manufacturing antimicrobial silicone device components that offers considerable advantages over traditional processes.
Study Methodology
The vast majority of silicone medical components are manufactured from raw material formulations containing polydimethylsiloxane (PDMS) polymers reinforced with amorphous non-crystalline silica. It is known that vulcanized PDMS elastomers easily swell when immersed in various organic solvents.
SSF’s study investigated the simultaneous transfer of two drugs from solution into pre-vulcanized silicone rubber components. The two antibiotic API investigated were Clindamycin Hydrochloride (CLIN)—mol weight = 461.45 g/mol—and Rifampicin (RIF)—mol weight = 822.94 g/mol. Study variables included 1) the total concentration of the drugs in solution; 2) the ratio of the drugs in solution; 3) immersion time of the silicone component; and 4) the durometer of the silicone test article.
Effect of Solution Concentration on Drug Impregnation
CLIN and RIF were added to chloroform to produce three unique solutions. The CLIN/RIF ratio was held constant but the overall concentration of the two drugs in solution was varied from x to 2x to 10x. Six-inch lengths of silicone tubing (50 durometer Shore A) were immersed in the solutions. Tubes were removed from solution at predetermined intervals; immersion times ranged from a few minutes to several hours. The impregnated tubing was then blotted dry and the inner lumen flushed with IPA. The test articles were then allowed to devolatilize for 24 hours.
SSF’s analytical lab developed methodology to extract both drugs from the silicone test articles as well as HPLC test methods to quantify drug concentrations. Small sections of tubes were weighed and extracted. The extract was then evaluated via HPLC to determine the masses of both drugs within a known mass of tubing. Results for the drug content testing of both CLIN and RIF are expressed in the units of micrograms of drug per gram of tube. Data for tubing immersed in the three different solutions is summarized in Figure 1.
Five data points, representing different immersion times, are plotted for each of the three solutions. In general, as the concentration of API in solution increased, so too did the resultant concentration within the silicone test article. It’s also evident from the plotted data that the rate of impregnation is strongly influenced by the concentration of the immersion solution. Finally, the lines representing the three data sets have similar slopes, indicating that the ratio of the two drugs impregnated within the silicone is dependent on the ratio of the drugs in solution and largely independent of the overall solution concentration.
Impact of Silicone on Drug Impregnation
A second experiment investigated the impact of elastomer formulation on both the mass transfer of the drugs from solution as well as the rate of impregnation. CLIN and RIF were added to chloroform to produce a 2x solution identical to that discussed in the previous test. Short lengths of 20 and 80 durometer tubing were immersed and then removed at the same five-time intervals as the prior experiment. Tubing segments were again assayed for total drug content.
Durometer is a measure of hardness. In general, in silica-reinforced systems such as the materials used in this study, an increase in durometer is mirrored by an increase in the silica content of the rubber. Approximate densities of silica and PDMS polymers are 2.2 and 1.0 g/cc, respectively. The specific gravity values for silicone elastomers provides a convenient means for quantifying and comparing the silica-polymer ratio between different rubbers. Specific gravities of the 20, 50, and 80 durometer tubes investigated here were 1.10, 1.16, and 1.21, respectively. In this regard, durometer was used as a proxy for polymer concentration within the rubber.
SSF study investigators hypothesized that the quasi-organic PDMS polymer, relative to the inorganic silica, would be more readily swelled by halogenated-organic solvents and the mass and rate of drug transfer would therefore increase as durometer decreased.
Results are summarized in Figure 2. Twenty durometer tubes, with high polymer content, absorbed more Clindamycin and at a faster rate than the 50 durometer tubes containing less polymer. Somewhat surprisingly, the mass and impregnation rate of rifampicin in the 20 durometer tubes was lower than that observed in the 50 durometer samples. Both API were below test detection limits in the 80 durometer tubes.
Varying API Ratios, Impact on Impregnation
A third experiment investigated how changes in the ratio of the two drugs in solution impacted drug concentrations in the silicone elastomer.
It is known that some types of ventricular catheters, a key component of life-saving hydrocephalus shunts, contain CLIN and RIF, and that studies have confirmed the antimicrobial efficacy of these devices. The literature does not address the manufacturing method used to produce these products but it does provide detailed information about the target concentrations of both drugs as well as specification ranges. Labeling for the devices notes CLIN and RIF concentrations of 0.15 percent (wt/wt) and 0.054 percent, respectively. Supporting literature discusses a specification ranges of ±45 percent for CLIN and 60 percent for RIF. These drug concentrations and ranges were used as targets in this experiment.
Several solutions were prepared in which the concentrations of CLIN and RIF were adjusted from the 1:1 ratio used in the previous experiments. As in the earlier tests, a number of immersion times were investigated for each of the solutions. Results are shown in Figure 3. The target concentration for both API is shown here as the white circle while the specification ranges for the drugs (RIF = 216-864, CLIN = 825-2,195) are identified as the area within the white box. Experimental results showed that several immersion times in two different solutions resulted in tubing drug concentrations that were within the specification range and that two data points (circled green) were very close to the published API target of the predicate devices.
Bacterial Challenge—Antimicrobial Efficacy Confirmed
The immersion experiments discussed previously demonstrated conclusively the mass transfer of both drugs from solution to the silicone tubing. Additionally, it was shown that the masses of drugs impregnated within the tubing were dependent upon controllable factors including solution concentration, immersion duration, and silicone type.
Importantly, though, the correlation between drug contents within the silicone and antimicrobial efficacy remained undetermined. SSF study investigators commissioned a Kirby-Bauer Zone of Inhibition (ZOI) test to assess the impact of drug content on the gram-positive coccal bacterium, Staphylococcus aureus. Staph aureus is responsible for particularly high incidence rates of HAI and DRI. In a technical brief,4 the National Institute of Allergy and Infectious Disease (NIAID) notes that each year an estimated 500,000 patients contract staph infections in American hospitals and that “S. aureus is the most common cause of HAI.” Other researchers note that “infections associated with indwelling medical devices ranging from simple intravenous catheters to prosthetic joints and replacement heart valves can be caused by S. aureus.”5
Test articles were chosen so that antimicrobial efficacy could be evaluated across a range of samples produced under varying conditions and containing different drug concentrations. Immersion conditions and drug content are summarized in Table 1.
The three test articles were placed on an agar plate that had been inoculated with a suspension containing Staphylococcus aureas. The plate was photographed after a 24 hour incubation period and is depicted in Figure 4.
The drug-impregnated tubing had a powerful inhibitory effect on the growth of S. aureus and clear zones of inhibition are seen surrounding test articles B and C. As expected, the higher drug concentrations in sample C produced a larger ZOI (18 mm) than sample B (14 mm), which contained lower concentrations of both drugs. No ZOI was observed around the control sample.
Discussion
Attractive forces between silicone polymer chains are quite weak. This contributes to the characteristic high free volume of silicone elastomers and their exceptional permeability. In turn, the high permeability of silicone rubber makes this biomaterial especially attractive as a matrix for drug-device combination products. As noted, the process for manufacturing most silicone drug-device components involves adding a drug powder to a silicone raw material, forming the mixture into a desired shape, then vulcanizing at elevated temperatures. But as discussed, there are several inherent concerns with this process that add complexity and costs.
Advantages of Immersion/Impregnation
The immersion/impregnation processes presented in these experiments offer several important advantages over current silicone drug-device manufacturing methods. These advantages include the following:
- Because the process involves immersion of pre-vulcanized components, the API cannot interfere with the cure chemistry of the silicone.
- The process can be easily tailored to accommodate different types of components including tubing, molded parts, and thin films.
- The immersion process may be conducted at room temperature, thereby eliminating concerns regarding the thermal degradation of the API.
- Solutions are, by definition, homogenous mixtures of solute and solvent. Silicone components immersed in these drug solutions are exposed to a uniform environment. Concerns regarding the mixing homogeneity of silicone and drugs are eliminated.
- Dissolved drugs are impregnated within the silicone elastomer as discrete molecules. Rifampicin is an intensely red powder and solutions containing the API exhibit a similar color. As shown in Figure 5, the impregnation of RIF, depending on solution concentration and immersion times, will cause a uniformly transparent tint throughout the catheter; API particles are not observed. Concerns and costs associated with specifying and maintaining a particular size and distribution of particles are eliminated.
- Equipment requirements for the immersion/impregnation process are minimal. A single containment vessel can be used for combining the drug powder with solvent as well as immersing the silicone components. Costs associated with cleaning method development and validation are minimal relative to those required for other processes where unvulcanized silicone-drug mixtures contact numerous surfaces of both mixing and fabrication equipment.
Healthcare costs associated with fighting device related infections are significant. The use of medical devices, including many life-saving devices made from silicone, will only accelerate as the world’s population ages. Additionally, recent changes to hospital reimbursement policy for some types of device-related infections have prompted medical device manufacturers to differentiate their products by including additives known to resist bacterial colonization. While it is doubtful that device-related infection can be completely eliminated, various antimicrobial strategies can effectively reduce the incidence of infection.
The experiments described in this analysis presented a cost-effective process for the impregnation of drugs within prefabricated silicone components. It was demonstrated that variables such as the concentration of drugs in solution, immersion duration and the type of silicone will impact the mass transfer of drugs. It is important to note, however, that while this study investigated chloroform solutions containing clindamycin hydrochloride and rifampicin, the process as described provides a broad and important platform suitable for numerous other antibiotic drugs and active pharmaceutical ingredients.
References:
- Scott, RD. The Direct Medical Costs of Healthcare-Associated Infections in U.S. Hospitals and the Benefits of Prevention, 2010, http://www.cdc.gov/HAI/pdfs/hai/Scott_CostPaper.pdf
- Darouiche, RO. The Road to Eliminating Device-Related Infections. Infection Control, 2008,
- http://www.patientsafetyfocus.com/2008/09/the-road-to-eli.html
- Knetch, MLW, Koole, LH. New Strategies in the Development of Antimicrobial Coatings: The Example of Increasing Usage of Silver and Silver Nanoparticles. Polymers, 2011, 3, 340-366
- http://web.archive.org/web/20070505050641/http:/www3.niaid.nih.gov/news/newsreleases/1999/staph
- Foster, T. Medical Microbiology, University of Texas Medical Branch at Galveston, 1996, http://www.ncbi.nlm.nih.gov/books/NBK8448/#A785
Mark Paulsen is president and founder of Silicone Consulting, LLC. He has held key positions within various sectors of the silicone medical device industry including raw material manufacturing (NuSil Technology, director of Healthcare Materials, 1992-2007) and component fabrication (SSF, director of Business Development, 2008-2013). Paulsen consults widely within the medical device industry on topics such as silicone chemistry, material selection, process optimization, and drug-device design and testing. Paulsen holds three patents for the development of a novel silicone elastomer used as insulation on cardiac and neurological stimulation devices. In 2004, he was recognized as one of the top 100 Notable People in the medical device industry. Paulsen studied chemistry at Harvard University and currently lives in Santa Barbara, Calif.
For more than 30 years Specialty Silicone Fabricators (SSF) has provided customized manufacturing solutions to the medical device industry. The company maintains three manufacturing sites and employs nearly 250 people. SSF has been awarded six patents for its developments in silicone extrusion and thin film processing. Additional services include micromolding, coating expertise, device assembly, and final package design and validation. In 2009, SSF received federal registration and state licensing to begin working with active pharmaceutical ingredients.