06.02.15
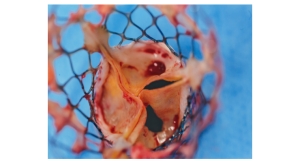
Edwards Lifesciences Corp. has halted enrollment in a clinical trial evaluating the Fortis heart valve. Subjects were
developing blood clots, and company officials felt the occurrences warranted further investigation.
To date, the trial treated more than 20 patients worldwide, all of whom had symptomatic mitral regurgitation and who were either compassionate cases or fell within in one of Edwards Lifesciences’ high-risk registries. The Fortis valve is not approved for sale in any country.
The company now is working with trial investigators and Heart Teams to gather additional data in this early study of transcatheter mitral valve replacement therapy.
Edwards Lifesciences develops technology for heart valves and hemodynamic monitoring. The Irvine, Calif.-based firm employs more than 9,000 people.
developing blood clots, and company officials felt the occurrences warranted further investigation.
To date, the trial treated more than 20 patients worldwide, all of whom had symptomatic mitral regurgitation and who were either compassionate cases or fell within in one of Edwards Lifesciences’ high-risk registries. The Fortis valve is not approved for sale in any country.
The company now is working with trial investigators and Heart Teams to gather additional data in this early study of transcatheter mitral valve replacement therapy.
Edwards Lifesciences develops technology for heart valves and hemodynamic monitoring. The Irvine, Calif.-based firm employs more than 9,000 people.