05.06.15
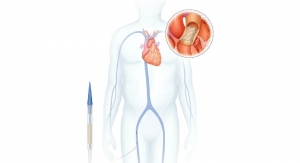
Dublin, Ireland-based Medtronic plc has earned Japanese regulatory approval for the self-expanding transcatheter CoreValve system for patients with severe aortic stenosis (AS) unable to undergo surgery, and for whom treatment with the CoreValve is determined to be the best option. Japanese regulatory authorities granted approval of the Corevalve system for transcatheter aortic valve implantation (TAVI) based on data from CoreValve U.S. pivotal trials and the Medtronic Corevalve Japan trial, which is the first study to evaluate a self-expandable transcatheter valve in the Japanese patient population.
“We are excited to be able to offer this new option for our patients, as the Core Valve system has demonstrated exceptional clinical results in studies globally as well as in Japan,” said Yoshiki Sawa, M.D., professor in the department of cardiovascular surgery at Osaka University Graduate School of Medicine in Osaka, Japan.
“Physicians in Japan should have confidence in making TAVI treatment decisions for patients who may benefit from a less invasive valve replacement procedure with this self-expandable valve.”
Data from extreme- and high-risk studies in the CoreValve U.S. pivotal trial demonstrated the CoreValve system is safe and effective with high rates of survival and some of the lowest rates of stroke and valve leakage reported, Medtronic claims. The system, with a supra-annular valve design, also has achieved hemodynamics, or blood flow, post-implant with results similar to the gold standard, surgical valves.
The Medtronic CoreValve Japan trial confirmed the performance of CoreValve shown in the U.S. trial. At six months, 91.7 percent of the transfemoral (TAVI procedure through the femoral artery) patients in the Japan trial met the primary endpoint of improvement of at least one New York Heart Association class (measures severity of heart failure symptoms) and aortic valve area (measures the severity of aortic stenosis) greater than 1.2 centimeters squared. For all patients in the trial, freedom from all-cause mortality at six months was 90.8 percent. The trial also demonstrated functional and anatomical effectiveness of CoreValve.
The CoreValve self-expanding nitinol frame enables physicians to deliver the device to the diseased valve in a controlled manner, allowing for accurate placement. Valves are delivered via the smallest (18 Fr, or approximately 1/4 inch) TAVR delivery system available, making it possible to treat patients with difficult or small vasculature.
“The feedback from heart teams around the world using CoreValve has been overwhelmingly positive and we are confident that Japanese physicians will embrace the therapy,” said Rhonda Robb, vice president and general manager of the Heart Valve Therapies business, which is part of the Cardiac and Vascular Group at Medtronic. “We look forward to working with Japan’s Ministry of Health, Labor and Welfare to gain reimbursement for CoreValve, and to initiating comprehensive training and
education programs to support heart teams so that more patients may benefit from this breakthrough technology.”
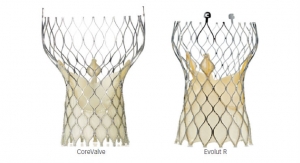
The CoreValve system received CE mark in 2007, was approved by the U.S. Food and Drug Administration in January 2014 for patients at extreme risk, and approved for high-risk patients in June 2014. The next-generation recapturable CoreValve Evolut R transcatheter valve and the CoreValve EnVeo R delivery catheter system are available in Europe and other countries that recognize the CE mark.
“We are excited to be able to offer this new option for our patients, as the Core Valve system has demonstrated exceptional clinical results in studies globally as well as in Japan,” said Yoshiki Sawa, M.D., professor in the department of cardiovascular surgery at Osaka University Graduate School of Medicine in Osaka, Japan.
“Physicians in Japan should have confidence in making TAVI treatment decisions for patients who may benefit from a less invasive valve replacement procedure with this self-expandable valve.”
Data from extreme- and high-risk studies in the CoreValve U.S. pivotal trial demonstrated the CoreValve system is safe and effective with high rates of survival and some of the lowest rates of stroke and valve leakage reported, Medtronic claims. The system, with a supra-annular valve design, also has achieved hemodynamics, or blood flow, post-implant with results similar to the gold standard, surgical valves.
The Medtronic CoreValve Japan trial confirmed the performance of CoreValve shown in the U.S. trial. At six months, 91.7 percent of the transfemoral (TAVI procedure through the femoral artery) patients in the Japan trial met the primary endpoint of improvement of at least one New York Heart Association class (measures severity of heart failure symptoms) and aortic valve area (measures the severity of aortic stenosis) greater than 1.2 centimeters squared. For all patients in the trial, freedom from all-cause mortality at six months was 90.8 percent. The trial also demonstrated functional and anatomical effectiveness of CoreValve.
The CoreValve self-expanding nitinol frame enables physicians to deliver the device to the diseased valve in a controlled manner, allowing for accurate placement. Valves are delivered via the smallest (18 Fr, or approximately 1/4 inch) TAVR delivery system available, making it possible to treat patients with difficult or small vasculature.
“The feedback from heart teams around the world using CoreValve has been overwhelmingly positive and we are confident that Japanese physicians will embrace the therapy,” said Rhonda Robb, vice president and general manager of the Heart Valve Therapies business, which is part of the Cardiac and Vascular Group at Medtronic. “We look forward to working with Japan’s Ministry of Health, Labor and Welfare to gain reimbursement for CoreValve, and to initiating comprehensive training and
education programs to support heart teams so that more patients may benefit from this breakthrough technology.”
The CoreValve system received CE mark in 2007, was approved by the U.S. Food and Drug Administration in January 2014 for patients at extreme risk, and approved for high-risk patients in June 2014. The next-generation recapturable CoreValve Evolut R transcatheter valve and the CoreValve EnVeo R delivery catheter system are available in Europe and other countries that recognize the CE mark.