Michael Barbella , Managing Editor03.13.14
The End is coming. Ready or not, like it or not—it’s coming.
And coming quickly.
Renowned futurist Raymond Kurzweil predicts humanity has only 31 years of cerebral dominance left before it succumbs to ultraintelligent machines. By 2045, he reckons, the computers we’ve spent decades building and programming not only will be infinitely smarter than us, they’ll ironically also have the ability to ration, reason and feel like the frontal, temporal and parietal lobes of an organic brain.
At that point—in the Year of Singularity—computer intelligence will be roughly 1 billion times as powerful as the sum of all human intellect. Mankind’s reign on Planet Earth will be over, prompting a societal transformation that Kurzweil calls “a singular change in human history.”
This is the way the world ends
This is the way the world ends
This is the way the world ends
Not with a bang but a whimper.
— T.S. Eliot, “The Hollow Men,” 1925.
Or, the press of the “delete” key.
Or, maybe it doesn’t end at all. Kurzweil is confident that artificial intelligence dramatically will improve and eventually help extend the human experience. Expert systems will eradicate viruses through rapid DNA sequencing; they’ll create new body parts (already occurring through 3-D printing); and perhaps most impressively, they’ll reprogram the body’s “stone-age software” to halt the aging process, allowing man to achieve his ultimate dream of immortality. In Kurzweil’s future world, humans will be able to sprint for 15 minutes without taking a breath and go scuba diving with no gear. He also expects nanotechnology to significantly enhance our mental capabilities, and virtual reality to become a ubiquitous daily experience.
“We’re going to become increasingly non-biological to the point where the non-biological part dominates and the biological part is not important anymore,” Kurzweil said during last year’s Global Future 2045 International Congress. “In fact, the non-biological part—the machine part—will be so powerful it can completely model and understand the biological part. So even if that biological part went away it wouldn’t make any difference. We’ll also have non-biological bodies—we can create bodies with nanotechnology, we can create virtual bodies and virtual reality in which the virtual reality will be as realistic as the actual reality. I think we’ll have a choice of bodies…we’ll have different ways we can create bodies.”
Full-body construction is still a bit too advanced for the human brain’s 100 billion neurons (though exoskeletons and the walking, talking 6-foot-tall bionic robot currently housed at the Smithsonian Museum arguably measure up—in a rudimentary, pre-Singularity kind of way). Nevertheless, the cerebral cortex has become quite adept through the centuries at creating body parts: Ancient Egyptians crafted jawbone implants from ivory while the Maya preferred shells (their calcium carbonate purportedly integrated well with bone). The Romans’ proclivity for gold cranium implants was mimicked several millennia later by the Incas of Peru, although the doomed society also liked silver for skull repairs.
Twentieth-century advances in biomaterial science have created more superior body parts and biologically worthy materials. Most joint replacements, for example, are made of a stainless steel, zirconium, polyethylene and in certain cases, ceramic mix; prosthetic limbs are composed of a carbon-fiber composite/polypropylene or polyethylene blend; artificial ears generally are manufactured from silicone or spider silk, and the latest retinal implant designs—otherwise known as “bionic eyes”—contain diamond electrodes.
“The number of engineering materials available across all industries has expanded dramatically in the last century from a few hundred to more than 160,000,” noted Sarah Egan, Ph.D., product manager for medical devices at Granta Design Ltd., a Cambridge, United Kingdom-based company providing materials data and selection software. “The same is true for medical, albeit with a smaller subset of materials available to the industry.”
The ringleaders of that subset have long been stainless steel, titanium and cobalt chromium, but newer materials such as magnesium, microstructured glass, polycarbonates (LNP Libricomp DCI06APH) and polyoxymethylene plastics increasingly are infiltrating the group with innovations like transparent ceramics and biodegradable alloys.
Advanced ceramics supplier CeramTec recently gained admission to the group by developing a magnesium aluminum spinel called Perlucor that features a high refractive index and resistance to both scratches and harsh chemicals (i.e., acids, lyes). The isotropic, polycrystalline substance—ideal for endoscopy devices—allows infrared rays to pass through a thickness of up to 6 millimeters, nearly triple the 2.5 millimeters found in most alternative materials.
South Korean researchers, meanwhile, earned a place in the bunch with their experimental biodegradable/bioabsorbable magnesium alloy implant that decomposes within six to 24 months of transplantation in the body. Scientists melded the metal with calcium and zinc to increase its corrosion resistance and prevent the formation of hydrogen gas bubbles that can accumulate around the implant and delay healing.
“The most important material additions for medical [devices] have been new metallic alloys (stainless steels, chromium and titanium alloys, nitinol and zirconium alloys) and a wide variety of polymers,” Egan told Medical Product Outsourcing. “The range of properties polymers have to offer are helping to push the boundaries of medical device technology. Polyurethanes, plus the use of glass and carbon-filled polymers, are good examples: The materials can be designed or tailored to meet certain requirements.”
Such customization is masterly epitomized in two carbon fiber-reinforced thermoplastics from Middle Eastern petrochemical manufacturer SABIC (Saudi Basic Industries Corporation). The company’s new high-modulus compounds, LNP Thermocomp EC006AQH and LNP Lubricomp DCI06APH, are reinforced with 30 percent carbon fiber, making the pair more lightweight and durable than traditional metals.
The strong but ductile Lubricomp material is designed specifically for components of drug delivery systems, disposable surgical instruments and medical device housings, according to the Riyadh, Saudi Arabia-based company. Its equally strong sister substance, conversely, was built to withstand multiple sterilization processes as well as chemically harsh hospital disinfectants (its defenses include a high tensile modulus, extreme heat resistance, chemical compatibility and mechanical stability).
“Hospitals have become incubators for some pretty strong bacteria and germs. As a consequence, the cleaning agents they use on equipment is getting stronger and that is causing a big issue with reusable devices,” said Ryan Case, sales and business development director for Orchard Park, N.Y.-headquartered Polymer Conversions, a full-service contract manufacturer specializing in medical clean-room plastic injection molding, assembly and packaging. “The plastics used in some of this equipment is breaking down because the disinfectants have become so potent. As a result, there’s been a trend among materials developers to find more chemically resistant substances for things like monitors and MRI machines.”
Material Design Drivers
Methicillin-resistant staphylococcus aureus (MRSA) is a stealthy warrior. It can live undetected among its foes, easily blending into the natural environment as it waits for the perfect opportunity to launch an attack. Wise to the body’s formidable defense strategy, the indomitable bacteria storms past the immune system and fires toxins into muscle and tissue, detonating cells.
MRSA’s war casualties as well as the conflict’s escalating cost in recent years ($63 million, by some estimates) is prompting many materials companies to design compounds with antimicrobial properties. Specialty Coating Systems Inc. (SCS) of Indianapolis, Ind., has developed thin Parylene coatings that protect medical devices against bodily fluids, moisture and chemicals. The Parylene—a trade name for chemical vapor deposited poly (p-xylylene) polymers—can be applied much thinner than alternative coatings but with superior barrier properties.
“Parylene is normally used as a barrier…it protects the body from the leaching of metals or plastics and protects the device from the fluids of the human body attacking the device. So it works both ways,” SCS Medical Market Manager Juan Gudino noted.
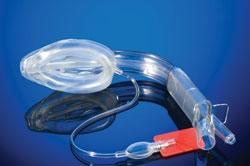
Medical-grade PVC (DEHP-free) was used for the blown cuff and inflation tubing of this laryngeal mask manufactured by Forefront Medical Technology. Image courtesy of Forefront Medical Technology.
SCS creates its microResist coating from ISO 10993-compliant medical Parylenes N, C and HT. The polymer is considered one of the most biocompatible barrier coatings for permanent medical implants such as stents, defibrillators and pacemakers. SCS tests on the material showed greater than 5 Log reduction after 24 hours of incubation with 14 common pathogens, including a 99.99997 percent efficacy protecting against MRSA. The microResist coating also offers high lubricity, high temperature abrasion and can withstand sterilization processes from ethylene oxide to gamma, according to the company.
BASF’s new weapons in the war against MRSA and other hospital-associated infections include HyGentic SBC and HyGentic PA, two polymers with “inbuilt antimicrobial functions,” according to the German chemical behemoth. The SBC grade is a transparent injection-moldable styrene butadiene block co-polymer granulate material with silver ions that can be used directly to manufacture devices like inhalers or ventilation filters.
The PA series is an injection-molded polyamide granulate as well but it is reinforced with glass fiber, making it ideal for medical device components.
“Fighting infection is a big driver [of material design], with many more ‘antimicrobial’ polymers appearing on the market in the last couple of years,” Granta’s Egan said. “These are typically polymers impregnated with a small amount of silver, though there are some polymers that exhibit antimicrobial properties in their own right. They are finding applications in devices such as inhalers as well as instrument handles and hospital equipment (e.g., bed rails).”
In the operating room, hospitals are employing another soldier in their fight against MRSA—disposable instruments. Global demand for disposable medical devices is projected to grow 6.4 percent annually to nearly $200 billion by 2016 as the battle against clinical infections intensifies; patient room equipment sales could reach $7.5 billion, while surgical instruments and suppliers may possibly top $32.2 billion, according to industry data.
Disposable instruments and supplies generally are crafted from soft-touch materials like thermoplastic elastomers or thermoplastic olefins. These materials are ideal for disposable products due to their low levels of extractables, cost-effectiveness, outstanding sealing performance and lack of polyvinyl chloride, a widely used but environmentally hazardous plastic.
“We are seeing a trend toward disposable tools used in operating room environments or in various therapies,” said David L. Smith, vice president of Business Development at Boston Centerless, a Woburn, Mass., supplier of precision ground bar materials and grinding services. “Hospitals used to go to a great deal of trouble to sterilize instruments that were used over and over again—things like scalpels, scissors, clamps and wrenches. We’ve noticed a move toward disposable instrumentation rather than reusable instrumentation. We have one customer that makes a line of surgical wrenches. In the past, those wrenches would have a steel shank on it where the actual work happens and a metallic handle. Today, that customer is making those [wrench] handles out of plastic. The work portion of the tool is still the same type of material but they can make it in such a way that the hospital saves money by using the tool once and disposing of it. So companies are doing things like substituting more permanent type handles with plastics in order to make [surgical] instruments cheap enough to use once and throw away rather than reusing it through autoclaving or cleaning.”
Infection control, however, is not the only driver of material design. Factors such as risk minimization, performance and regulatory requirements affect innovation as well. The U.S. Food and Drug Administration’s (FDA) newest draft guidance on animal-derived materials, for instance, could force companies to reassess the use of bovine, ovine, porcine and avian substances in finished products. The January guidance, meant to replace the FDA’s 1998 rules on the transmission risks of bovine spongiform encephalopathy (Mad Cow Disease), also warns of risks from such other transmissible spongiform encephalopathies as scrapie (found in sheep), chronic waste disease (in deer), and the human degenerative neurological disorder Creutzfeldt-Jakob Disease.
Similarly, the European Union ban on phthalate is likely to inspire material designs with higher biocompatibility ratings, experts claim.
“Greater understanding of the substances that can leach in drugs and cause biocompatibility issues has led to greater regulation,” noted Daniel Benze, vice president, Operations and Technical, at Forefront Medical Technology, a specialty contract manufacturer with a focus in disposable diagnostic, drug infusion and medical device systems. “For example, Di(2-ethylhexyl) phthalate (DEHP) will be banned in the EU after Feb. 21, 2015. This is driving the use of materials with higher biocompatibility ratings which still provide the required levels of strength and flexibility such as DEHP-free PVC, thermoplastic polyurethane, PEEK, Kevlar and polybutadiene.”
Even the best biocompatible materials, though, are useless without an economically viable end market. Pricing pressures, heightened regulatory compliance and perhaps most importantly, shrinking reimbursement rates, increasingly are influencing medtech material design and selection.
“A common challenge today is that companies are able to make different materials and Vesta has the capabilities to fabricate these into innovative devices for customers, but this alone is not enough. The innovation has to show economic benefit as well,” explained Julie Cameron, marketing vice president for Vesta Inc., a Franklin, Wis., medical device outsourcing provider specializing in molding, extrusion and assembly. “At a recent supplier event, the message from an R&D executive was ‘a good idea isn’t good enough’ and he went on to explain how innovation has to deliver economic benefits to the customer well. The customer is not only the patient or the physician anymore. The customer includes who is paying, meaning hospital buying groups and insurance provider. These are the economic engine and we are all being challenged to deliver patient outcomes more efficiently.”
* * *
T-minus 31 years and counting.
Humans have roughly three decades left to experiment with body part materials before artificial intelligence perfects the process. While trends such as miniaturization, infection control, environmental responsibility and regulation are driving material designs, true innovation often rests with tried-and-true favorites—the metals, plastics, glass, ceramics, silicone and diamonds responsible for the replacement eyes, ears, hands, limbs and organs that will likely sustain us until the moment of Singularity arrives. As Egan notes, “the innovation is not necessarily in the development of a new material, but in better understanding of what materials are already available, and applying existing materials more effectively. Achieving this requires access to right materials data and tools.”
With the help of machines, of course.
And coming quickly.
Renowned futurist Raymond Kurzweil predicts humanity has only 31 years of cerebral dominance left before it succumbs to ultraintelligent machines. By 2045, he reckons, the computers we’ve spent decades building and programming not only will be infinitely smarter than us, they’ll ironically also have the ability to ration, reason and feel like the frontal, temporal and parietal lobes of an organic brain.
At that point—in the Year of Singularity—computer intelligence will be roughly 1 billion times as powerful as the sum of all human intellect. Mankind’s reign on Planet Earth will be over, prompting a societal transformation that Kurzweil calls “a singular change in human history.”
This is the way the world ends
This is the way the world ends
This is the way the world ends
Not with a bang but a whimper.
— T.S. Eliot, “The Hollow Men,” 1925.
Or, the press of the “delete” key.
Or, maybe it doesn’t end at all. Kurzweil is confident that artificial intelligence dramatically will improve and eventually help extend the human experience. Expert systems will eradicate viruses through rapid DNA sequencing; they’ll create new body parts (already occurring through 3-D printing); and perhaps most impressively, they’ll reprogram the body’s “stone-age software” to halt the aging process, allowing man to achieve his ultimate dream of immortality. In Kurzweil’s future world, humans will be able to sprint for 15 minutes without taking a breath and go scuba diving with no gear. He also expects nanotechnology to significantly enhance our mental capabilities, and virtual reality to become a ubiquitous daily experience.
“We’re going to become increasingly non-biological to the point where the non-biological part dominates and the biological part is not important anymore,” Kurzweil said during last year’s Global Future 2045 International Congress. “In fact, the non-biological part—the machine part—will be so powerful it can completely model and understand the biological part. So even if that biological part went away it wouldn’t make any difference. We’ll also have non-biological bodies—we can create bodies with nanotechnology, we can create virtual bodies and virtual reality in which the virtual reality will be as realistic as the actual reality. I think we’ll have a choice of bodies…we’ll have different ways we can create bodies.”
Full-body construction is still a bit too advanced for the human brain’s 100 billion neurons (though exoskeletons and the walking, talking 6-foot-tall bionic robot currently housed at the Smithsonian Museum arguably measure up—in a rudimentary, pre-Singularity kind of way). Nevertheless, the cerebral cortex has become quite adept through the centuries at creating body parts: Ancient Egyptians crafted jawbone implants from ivory while the Maya preferred shells (their calcium carbonate purportedly integrated well with bone). The Romans’ proclivity for gold cranium implants was mimicked several millennia later by the Incas of Peru, although the doomed society also liked silver for skull repairs.
Twentieth-century advances in biomaterial science have created more superior body parts and biologically worthy materials. Most joint replacements, for example, are made of a stainless steel, zirconium, polyethylene and in certain cases, ceramic mix; prosthetic limbs are composed of a carbon-fiber composite/polypropylene or polyethylene blend; artificial ears generally are manufactured from silicone or spider silk, and the latest retinal implant designs—otherwise known as “bionic eyes”—contain diamond electrodes.
“The number of engineering materials available across all industries has expanded dramatically in the last century from a few hundred to more than 160,000,” noted Sarah Egan, Ph.D., product manager for medical devices at Granta Design Ltd., a Cambridge, United Kingdom-based company providing materials data and selection software. “The same is true for medical, albeit with a smaller subset of materials available to the industry.”
The ringleaders of that subset have long been stainless steel, titanium and cobalt chromium, but newer materials such as magnesium, microstructured glass, polycarbonates (LNP Libricomp DCI06APH) and polyoxymethylene plastics increasingly are infiltrating the group with innovations like transparent ceramics and biodegradable alloys.
Advanced ceramics supplier CeramTec recently gained admission to the group by developing a magnesium aluminum spinel called Perlucor that features a high refractive index and resistance to both scratches and harsh chemicals (i.e., acids, lyes). The isotropic, polycrystalline substance—ideal for endoscopy devices—allows infrared rays to pass through a thickness of up to 6 millimeters, nearly triple the 2.5 millimeters found in most alternative materials.
South Korean researchers, meanwhile, earned a place in the bunch with their experimental biodegradable/bioabsorbable magnesium alloy implant that decomposes within six to 24 months of transplantation in the body. Scientists melded the metal with calcium and zinc to increase its corrosion resistance and prevent the formation of hydrogen gas bubbles that can accumulate around the implant and delay healing.
“The most important material additions for medical [devices] have been new metallic alloys (stainless steels, chromium and titanium alloys, nitinol and zirconium alloys) and a wide variety of polymers,” Egan told Medical Product Outsourcing. “The range of properties polymers have to offer are helping to push the boundaries of medical device technology. Polyurethanes, plus the use of glass and carbon-filled polymers, are good examples: The materials can be designed or tailored to meet certain requirements.”
Such customization is masterly epitomized in two carbon fiber-reinforced thermoplastics from Middle Eastern petrochemical manufacturer SABIC (Saudi Basic Industries Corporation). The company’s new high-modulus compounds, LNP Thermocomp EC006AQH and LNP Lubricomp DCI06APH, are reinforced with 30 percent carbon fiber, making the pair more lightweight and durable than traditional metals.
The strong but ductile Lubricomp material is designed specifically for components of drug delivery systems, disposable surgical instruments and medical device housings, according to the Riyadh, Saudi Arabia-based company. Its equally strong sister substance, conversely, was built to withstand multiple sterilization processes as well as chemically harsh hospital disinfectants (its defenses include a high tensile modulus, extreme heat resistance, chemical compatibility and mechanical stability).
“Hospitals have become incubators for some pretty strong bacteria and germs. As a consequence, the cleaning agents they use on equipment is getting stronger and that is causing a big issue with reusable devices,” said Ryan Case, sales and business development director for Orchard Park, N.Y.-headquartered Polymer Conversions, a full-service contract manufacturer specializing in medical clean-room plastic injection molding, assembly and packaging. “The plastics used in some of this equipment is breaking down because the disinfectants have become so potent. As a result, there’s been a trend among materials developers to find more chemically resistant substances for things like monitors and MRI machines.”
Material Design Drivers
Methicillin-resistant staphylococcus aureus (MRSA) is a stealthy warrior. It can live undetected among its foes, easily blending into the natural environment as it waits for the perfect opportunity to launch an attack. Wise to the body’s formidable defense strategy, the indomitable bacteria storms past the immune system and fires toxins into muscle and tissue, detonating cells.
MRSA’s war casualties as well as the conflict’s escalating cost in recent years ($63 million, by some estimates) is prompting many materials companies to design compounds with antimicrobial properties. Specialty Coating Systems Inc. (SCS) of Indianapolis, Ind., has developed thin Parylene coatings that protect medical devices against bodily fluids, moisture and chemicals. The Parylene—a trade name for chemical vapor deposited poly (p-xylylene) polymers—can be applied much thinner than alternative coatings but with superior barrier properties.
“Parylene is normally used as a barrier…it protects the body from the leaching of metals or plastics and protects the device from the fluids of the human body attacking the device. So it works both ways,” SCS Medical Market Manager Juan Gudino noted.
Medical-grade PVC (DEHP-free) was used for the blown cuff and inflation tubing of this laryngeal mask manufactured by Forefront Medical Technology. Image courtesy of Forefront Medical Technology.
BASF’s new weapons in the war against MRSA and other hospital-associated infections include HyGentic SBC and HyGentic PA, two polymers with “inbuilt antimicrobial functions,” according to the German chemical behemoth. The SBC grade is a transparent injection-moldable styrene butadiene block co-polymer granulate material with silver ions that can be used directly to manufacture devices like inhalers or ventilation filters.
The PA series is an injection-molded polyamide granulate as well but it is reinforced with glass fiber, making it ideal for medical device components.
“Fighting infection is a big driver [of material design], with many more ‘antimicrobial’ polymers appearing on the market in the last couple of years,” Granta’s Egan said. “These are typically polymers impregnated with a small amount of silver, though there are some polymers that exhibit antimicrobial properties in their own right. They are finding applications in devices such as inhalers as well as instrument handles and hospital equipment (e.g., bed rails).”
In the operating room, hospitals are employing another soldier in their fight against MRSA—disposable instruments. Global demand for disposable medical devices is projected to grow 6.4 percent annually to nearly $200 billion by 2016 as the battle against clinical infections intensifies; patient room equipment sales could reach $7.5 billion, while surgical instruments and suppliers may possibly top $32.2 billion, according to industry data.
Disposable instruments and supplies generally are crafted from soft-touch materials like thermoplastic elastomers or thermoplastic olefins. These materials are ideal for disposable products due to their low levels of extractables, cost-effectiveness, outstanding sealing performance and lack of polyvinyl chloride, a widely used but environmentally hazardous plastic.
“We are seeing a trend toward disposable tools used in operating room environments or in various therapies,” said David L. Smith, vice president of Business Development at Boston Centerless, a Woburn, Mass., supplier of precision ground bar materials and grinding services. “Hospitals used to go to a great deal of trouble to sterilize instruments that were used over and over again—things like scalpels, scissors, clamps and wrenches. We’ve noticed a move toward disposable instrumentation rather than reusable instrumentation. We have one customer that makes a line of surgical wrenches. In the past, those wrenches would have a steel shank on it where the actual work happens and a metallic handle. Today, that customer is making those [wrench] handles out of plastic. The work portion of the tool is still the same type of material but they can make it in such a way that the hospital saves money by using the tool once and disposing of it. So companies are doing things like substituting more permanent type handles with plastics in order to make [surgical] instruments cheap enough to use once and throw away rather than reusing it through autoclaving or cleaning.”
Infection control, however, is not the only driver of material design. Factors such as risk minimization, performance and regulatory requirements affect innovation as well. The U.S. Food and Drug Administration’s (FDA) newest draft guidance on animal-derived materials, for instance, could force companies to reassess the use of bovine, ovine, porcine and avian substances in finished products. The January guidance, meant to replace the FDA’s 1998 rules on the transmission risks of bovine spongiform encephalopathy (Mad Cow Disease), also warns of risks from such other transmissible spongiform encephalopathies as scrapie (found in sheep), chronic waste disease (in deer), and the human degenerative neurological disorder Creutzfeldt-Jakob Disease.
Similarly, the European Union ban on phthalate is likely to inspire material designs with higher biocompatibility ratings, experts claim.
“Greater understanding of the substances that can leach in drugs and cause biocompatibility issues has led to greater regulation,” noted Daniel Benze, vice president, Operations and Technical, at Forefront Medical Technology, a specialty contract manufacturer with a focus in disposable diagnostic, drug infusion and medical device systems. “For example, Di(2-ethylhexyl) phthalate (DEHP) will be banned in the EU after Feb. 21, 2015. This is driving the use of materials with higher biocompatibility ratings which still provide the required levels of strength and flexibility such as DEHP-free PVC, thermoplastic polyurethane, PEEK, Kevlar and polybutadiene.”
Even the best biocompatible materials, though, are useless without an economically viable end market. Pricing pressures, heightened regulatory compliance and perhaps most importantly, shrinking reimbursement rates, increasingly are influencing medtech material design and selection.
“A common challenge today is that companies are able to make different materials and Vesta has the capabilities to fabricate these into innovative devices for customers, but this alone is not enough. The innovation has to show economic benefit as well,” explained Julie Cameron, marketing vice president for Vesta Inc., a Franklin, Wis., medical device outsourcing provider specializing in molding, extrusion and assembly. “At a recent supplier event, the message from an R&D executive was ‘a good idea isn’t good enough’ and he went on to explain how innovation has to deliver economic benefits to the customer well. The customer is not only the patient or the physician anymore. The customer includes who is paying, meaning hospital buying groups and insurance provider. These are the economic engine and we are all being challenged to deliver patient outcomes more efficiently.”
* * *
T-minus 31 years and counting.
Humans have roughly three decades left to experiment with body part materials before artificial intelligence perfects the process. While trends such as miniaturization, infection control, environmental responsibility and regulation are driving material designs, true innovation often rests with tried-and-true favorites—the metals, plastics, glass, ceramics, silicone and diamonds responsible for the replacement eyes, ears, hands, limbs and organs that will likely sustain us until the moment of Singularity arrives. As Egan notes, “the innovation is not necessarily in the development of a new material, but in better understanding of what materials are already available, and applying existing materials more effectively. Achieving this requires access to right materials data and tools.”
With the help of machines, of course.



























