Maria Shepherd, Data Decision Group03.13.14
More than 70 million Americans suffer from sleep disorders and of those, more than 18 million have obstructive sleep apnea (OSA).1 OSA is caused by a blockage in the throat, generally associated with a collapse of the soft tissue. With each episode of apnea, the brain partially or fully wakes the sleeping patient, as an alert to continue breathing. The interval between breaths can last from seconds to minutes, and these intervals can happen more than 30 times per hour.2
Why It’s Important
If left untreated, this condition can trigger high blood pressure and other cardiovascular co-morbidities that touch almost every disease state addressed by the medical device industry. OSA is a complex disease and patients, despite their growing numbers, remain an underserved market. This is a significant opportunity for innovators in the medical device industry.
Heart Failure and OSA
According an article in Circulation: Journal of the American Heart Association, OSA raised the risk of heart failure for middle-aged and older men, and significantly raised the risk of coronary heart disease (CHD) in men up to age 70.4
The study followed 1,927 men and 2,495 women (40 years of age or older) that were free of heart problems when the study began. A total of 24 percent of the men and 11 percent of the women had at least moderately severe OSA. Researchers assessed participants’ health for a median follow-up of 8.7 years. The study found that men with severe OSA faced a 58 percent higher risk of developing heart failure than those without OSA. Men between 40-70 years old, with severe OSA, had a 68 percent higher risk of developing CHD than those without OSA.
OSA and Quality of Life
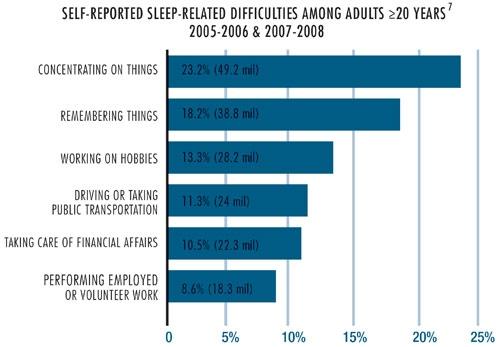
OSA isn’t just a health hazard, it also is a disease state that affects quality of life. According to the Centers for Disease Control and Prevention, insufficient sleep is a public health epidemic, with “sleep insufficiency linked to motor vehicle crashes, industrial disasters, and medical and other occupational errors.”5,6 When OSA is not the root cause of a disaster, it is an unpleasant disease state, as seen in the data in Chart 2.
The Opportunity in Sleep Apnea
OSA treatment options haven’t changed much in recent years and there still is great opportunity to treat this disease state. Chart 3 shows treatment options for OSA.
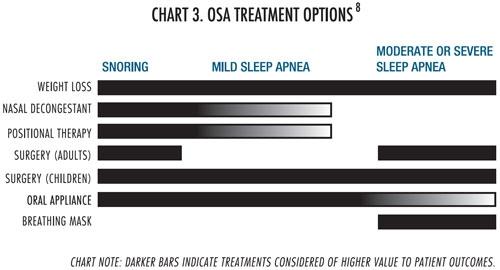
The most common form of surgery for OSA is uvulopalatopharyngoplasty (UPPP) and the success rate for UPPP is estimated to be approximately 50 percent.9,10,11 It is a complicated surgery because the surgeon must identify the section of the upper airway causing the airflow obstruction. There are multiple sites for possible treatment, and no diagnostic test exists today to identify the specific area the surgeon should treat. If the surgeon does not identify the appropriate surgical site in the airway, or if many obstruction sites exist, sleep apnea will persist and another surgery may be required.12
Overall Impact on the Healthcare System
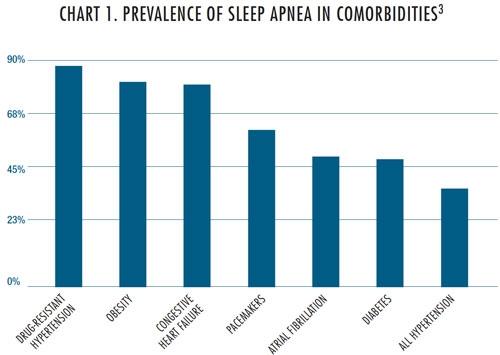
As is the case with diabetes, an effective treatment for OSA could have a significant ripple effect across the healthcare system, addressing all the co-morbidities defined in Chart 1. Medical device companies have a great opportunity to find treatments to stop or mitigate this complex disease state.
References:
Maria Shepherd has 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career—including her role as vice president of marketing for Oridion Medical, where she helped boost the company valuation prior to its acquisition by Covidien; director of marketing for Philips Medical; and senior management roles at Boston Scientific Inc.—she founded Data Decision Group. Data Decision Group (www.ddecisiongroup.com) provides whitespace research and critical data to support medical device product development. The firm performs market research, evaluates new technologies and provides marketing services and human factors usability testing. Shepherd has taught marketing and product development courses and recently was appointed to the board of the MSBiV Medtech Investment Committee. She can be reached at (617) 548-9892 or at mshepherd@ddecisiongroup.com.
Why It’s Important
If left untreated, this condition can trigger high blood pressure and other cardiovascular co-morbidities that touch almost every disease state addressed by the medical device industry. OSA is a complex disease and patients, despite their growing numbers, remain an underserved market. This is a significant opportunity for innovators in the medical device industry.
Heart Failure and OSA
According an article in Circulation: Journal of the American Heart Association, OSA raised the risk of heart failure for middle-aged and older men, and significantly raised the risk of coronary heart disease (CHD) in men up to age 70.4
OSA and Quality of Life
OSA isn’t just a health hazard, it also is a disease state that affects quality of life. According to the Centers for Disease Control and Prevention, insufficient sleep is a public health epidemic, with “sleep insufficiency linked to motor vehicle crashes, industrial disasters, and medical and other occupational errors.”5,6 When OSA is not the root cause of a disaster, it is an unpleasant disease state, as seen in the data in Chart 2.
OSA treatment options haven’t changed much in recent years and there still is great opportunity to treat this disease state. Chart 3 shows treatment options for OSA.
Overall Impact on the Healthcare System
As is the case with diabetes, an effective treatment for OSA could have a significant ripple effect across the healthcare system, addressing all the co-morbidities defined in Chart 1. Medical device companies have a great opportunity to find treatments to stop or mitigate this complex disease state.
References:
- www.sleepapnea.org/learn/sleep-apnea/
- www.nlm.nih.gov/medlineplus/sleepapnea.html
- www.resmed.com/us/clinicians/about_sleep_and_breathing/comorbidities/associated-risks.html?nc=clinicians
- Circulation, 2008; 118: 1080-111
- www.cdc.gov/features/dssleep/
- Institute of Medicine. Sleep Disorders and Sleep Deprivation: An Unmet Public Health Problem. Washington, D.C.: The National Academies Press; 2006.
- www.cdc.gov/features/dssleep/
- www.sleepapnea.org/treat/sleep-apnea-treatment-options/
- Ibid
- Friedman M, Ibrahim H, Bass L. Clinical staging for sleep-disordered breathing. Otolaryngol Head Neck Surg. Jul 2002;127(1):1321.
- Friedman M, Ibrahim H, Joseph NJ. Staging of obstructive sleep apnea/hypopnea syndrome: a guide to appropriate treatment. Laryngoscope. Mar 2004;114(3):454-9.
- www.sleepapnea.org/treat/sleep-apnea-treatment-options/
Maria Shepherd has 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career—including her role as vice president of marketing for Oridion Medical, where she helped boost the company valuation prior to its acquisition by Covidien; director of marketing for Philips Medical; and senior management roles at Boston Scientific Inc.—she founded Data Decision Group. Data Decision Group (www.ddecisiongroup.com) provides whitespace research and critical data to support medical device product development. The firm performs market research, evaluates new technologies and provides marketing services and human factors usability testing. Shepherd has taught marketing and product development courses and recently was appointed to the board of the MSBiV Medtech Investment Committee. She can be reached at (617) 548-9892 or at mshepherd@ddecisiongroup.com.