Joe Rotino07.26.10
New Manufacturing Supplier Controls: How Do You Stack Up?
As the regulatory landscape continues to evolve, both the U.S. Food and Drug Administration (FDA) and international regulatory bodies have moved their focus to supplier controls as a key element of medical device compliance. Medical device companies and their suppliers must ensure their quality management system is being enhanced regarding supplier programs. Companies must establish a risk-based structure that practices accountability and ensures product safety, effectiveness and regulatory compliance. During the last several years, the FDA has reported up to a 40 percent increase in warning letters regarding purchasing control deficiencies.
Historically, companies have relied on their suppliers for consistent product quality and have relaxed their controls and monitoring of incoming product. In addition, there has been a lack of focus on more rigorous supplier selection processes when adding new suppliers.
The FDA is looking to medical device firms and their suppliers to further address the following:
• Increased accountability to prevent harm by recognizing the supplier’s impact on device safety and quality
• Responsibility to address hazards introduced by supplier processes
• Extension of process validation down the supply chain.
Notified Bodies also are concentrating on supplier controls during ISO certification assessments. This is now backed up by changes to the Medical Device Directive in the European Union with the issuance of MDD 2007/47/EEC. These changes focus on demonstrating appropriate supplier controls based on the risk of the supplied product or service.
“With our national and international partners, we must find ways to ensure that global and domestic companies secure their increasingly complex and ever-growing supply chains,” FDA Commissioner Margaret Hamburg, M.D., said during a speech last August.
If your company hasn’t felt the impact of these industry changes already, then you are behind the industry curve. First-tier suppliers already should be reviewing quality systems and making immediate changes to strengthen these processes. Here are the primary areas of focus and some details on how to implement these changes.
Based upon information from Global Harmonization Task Force guidance document GHTF/SG3/N17:2008, the key phases of supplier controls are as follows:
• Planning
• Selection of potential suppliers
• Supplier evaluation and acceptance
• Finalization of controls
• Delivery, measurement and monitoring
• Feedback and communication
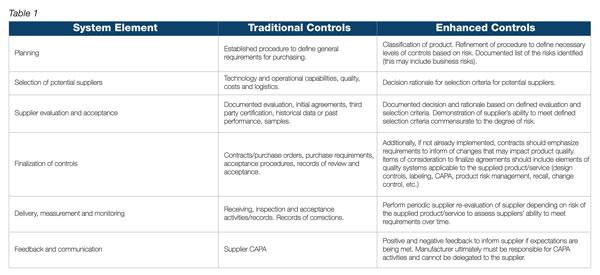
Table 1 shows how a changing regulatory profile will require enhanced controls compared to an existing system’s more traditional controls.
To help in better understanding the six phases, here are some definitions:
1. Planning: During the planning and execution of product realization for a new or existing medical device, the manufacturer identifies products or services to be obtained from a supplier. Planning provides the direction for establishing the extent of controls required for product and services to be obtained from suppliers.
2. Selection of potential suppliers: When selecting potential suppliers, manufacturers should investigate business and operational capabilites, which may include technological capabilities, to ensure that the supplier can provide the necessary quality, safety, performance and reliability of the products and services.
3. Supplier evaluation and acceptance: Define the process by which the manufacturer evaluates that the selected potential supplier is actually capable of supplying the product or service in accordance with the manufacturer’s requirements. The extent of the evaluation and acceptance activity performed should be in proportion to the identified risk of the procured product, and/or services, on the safety and effectiveness/performance of the final product.
4. Finalization of controls: The finalization of the controls is mutually agreed upon by the manufacturer and the supplier. Determining the extent and degree of controls, as well as defining clear lines of responsibilities, should be defined by the manufacturer. As a result of the supplier evaluation and acceptance, the controls need to be finalized as previously defined in the planning process.
5. Delivery, measurement and monitoring: In this phase, the accepted supplier will deliver products/service according to the agreed arrangements and these products will be used by the manufacturer in the product realization process. Within the product realization process, the manufacturer will establish checkpoints to monitor the supplier’s performance to ensure that customer and regulatory requirements continue to be met.
6. Feedback and communication: Provisions should be in place for the manufacturer to inform the supplier of whether the manufacturer’s expectations are being met. Feedback should be both positive and negative. The manufacturer should ensure there are effective lines of communication open to both parties to discuss problems/complaints or other matters. It is important that trust be developed between parties so that any problems can be resolved quickly in a cooperative way.
The Result
Medical device manufacturers following these six phases will be looking closely at suppliers to address the changes and implement stricter controls. Suppliers working with their manufacturing partners should review their own internal processes to ensure that they meet these new, tighter controls. Suppliers are assigning internal RA/QA (regulatory affairs and quality assurance) and materials teams to review how the guidelines impact their existing processes. These teams are taking a proactive approach to determine the appropriate method for responding to the current changes as well as planning for future requirements.
Device manufacturers also will look closely at supplier certifications. Some only select and approve ISO 13485-certified suppliers as part of this shift in responsibility for the final product. If a supplier is not ISO 13485-certified, but manufactures components that are critical in the performance of the final product, the supplier must meet these new control requirements. If their quality system and internal processes cannot meet these specific supplier controls, thedevice company would begin to source a new qualified supplier. Companies such as Zimmer and Medtronic have responded by developing clear supplier control guidelines.
It is the obligation of the supplier as part of the medical device supply chain to identify risks as early in the process as possible, select the best subcontractors with well-structured quality systems and excellent design and process controls, to support their partner, the medical device company and ultimately the goal of product and
patient safety.
The first step is to understand how these changes will affect medical device suppliers in the near and future term. The FDA offers support for smaller companies to make the transition easier. Companies also may get advice from notified bodies for additional guidance and information. Medical device firms and their suppliers should align their systems to ensure they stay ahead of the curve regarding their supplier controls. As the FDA is focusing more on these controls regarding product safety throughout the supply chain, so should all medical device companies.
Joe Rotino is vice president of RA/QA & Engineering for Pro-Dex, Inc., based in Irvine, Calif. Pro-Dex designs, develops and manufacturers powered medical products, components, and subassemblies for some of the world’s leading medical device OEMs.
As the regulatory landscape continues to evolve, both the U.S. Food and Drug Administration (FDA) and international regulatory bodies have moved their focus to supplier controls as a key element of medical device compliance. Medical device companies and their suppliers must ensure their quality management system is being enhanced regarding supplier programs. Companies must establish a risk-based structure that practices accountability and ensures product safety, effectiveness and regulatory compliance. During the last several years, the FDA has reported up to a 40 percent increase in warning letters regarding purchasing control deficiencies.
Historically, companies have relied on their suppliers for consistent product quality and have relaxed their controls and monitoring of incoming product. In addition, there has been a lack of focus on more rigorous supplier selection processes when adding new suppliers.
The FDA is looking to medical device firms and their suppliers to further address the following:
• Increased accountability to prevent harm by recognizing the supplier’s impact on device safety and quality
• Responsibility to address hazards introduced by supplier processes
• Extension of process validation down the supply chain.
Notified Bodies also are concentrating on supplier controls during ISO certification assessments. This is now backed up by changes to the Medical Device Directive in the European Union with the issuance of MDD 2007/47/EEC. These changes focus on demonstrating appropriate supplier controls based on the risk of the supplied product or service.
“With our national and international partners, we must find ways to ensure that global and domestic companies secure their increasingly complex and ever-growing supply chains,” FDA Commissioner Margaret Hamburg, M.D., said during a speech last August.
If your company hasn’t felt the impact of these industry changes already, then you are behind the industry curve. First-tier suppliers already should be reviewing quality systems and making immediate changes to strengthen these processes. Here are the primary areas of focus and some details on how to implement these changes.
Based upon information from Global Harmonization Task Force guidance document GHTF/SG3/N17:2008, the key phases of supplier controls are as follows:
• Planning
• Selection of potential suppliers
• Supplier evaluation and acceptance
• Finalization of controls
• Delivery, measurement and monitoring
• Feedback and communication
Table 1 shows how a changing regulatory profile will require enhanced controls compared to an existing system’s more traditional controls.
To help in better understanding the six phases, here are some definitions:
1. Planning: During the planning and execution of product realization for a new or existing medical device, the manufacturer identifies products or services to be obtained from a supplier. Planning provides the direction for establishing the extent of controls required for product and services to be obtained from suppliers.
2. Selection of potential suppliers: When selecting potential suppliers, manufacturers should investigate business and operational capabilites, which may include technological capabilities, to ensure that the supplier can provide the necessary quality, safety, performance and reliability of the products and services.
3. Supplier evaluation and acceptance: Define the process by which the manufacturer evaluates that the selected potential supplier is actually capable of supplying the product or service in accordance with the manufacturer’s requirements. The extent of the evaluation and acceptance activity performed should be in proportion to the identified risk of the procured product, and/or services, on the safety and effectiveness/performance of the final product.
4. Finalization of controls: The finalization of the controls is mutually agreed upon by the manufacturer and the supplier. Determining the extent and degree of controls, as well as defining clear lines of responsibilities, should be defined by the manufacturer. As a result of the supplier evaluation and acceptance, the controls need to be finalized as previously defined in the planning process.
5. Delivery, measurement and monitoring: In this phase, the accepted supplier will deliver products/service according to the agreed arrangements and these products will be used by the manufacturer in the product realization process. Within the product realization process, the manufacturer will establish checkpoints to monitor the supplier’s performance to ensure that customer and regulatory requirements continue to be met.
6. Feedback and communication: Provisions should be in place for the manufacturer to inform the supplier of whether the manufacturer’s expectations are being met. Feedback should be both positive and negative. The manufacturer should ensure there are effective lines of communication open to both parties to discuss problems/complaints or other matters. It is important that trust be developed between parties so that any problems can be resolved quickly in a cooperative way.
The Result
Medical device manufacturers following these six phases will be looking closely at suppliers to address the changes and implement stricter controls. Suppliers working with their manufacturing partners should review their own internal processes to ensure that they meet these new, tighter controls. Suppliers are assigning internal RA/QA (regulatory affairs and quality assurance) and materials teams to review how the guidelines impact their existing processes. These teams are taking a proactive approach to determine the appropriate method for responding to the current changes as well as planning for future requirements.
Device manufacturers also will look closely at supplier certifications. Some only select and approve ISO 13485-certified suppliers as part of this shift in responsibility for the final product. If a supplier is not ISO 13485-certified, but manufactures components that are critical in the performance of the final product, the supplier must meet these new control requirements. If their quality system and internal processes cannot meet these specific supplier controls, thedevice company would begin to source a new qualified supplier. Companies such as Zimmer and Medtronic have responded by developing clear supplier control guidelines.
It is the obligation of the supplier as part of the medical device supply chain to identify risks as early in the process as possible, select the best subcontractors with well-structured quality systems and excellent design and process controls, to support their partner, the medical device company and ultimately the goal of product and
patient safety.
The first step is to understand how these changes will affect medical device suppliers in the near and future term. The FDA offers support for smaller companies to make the transition easier. Companies also may get advice from notified bodies for additional guidance and information. Medical device firms and their suppliers should align their systems to ensure they stay ahead of the curve regarding their supplier controls. As the FDA is focusing more on these controls regarding product safety throughout the supply chain, so should all medical device companies.
Joe Rotino is vice president of RA/QA & Engineering for Pro-Dex, Inc., based in Irvine, Calif. Pro-Dex designs, develops and manufacturers powered medical products, components, and subassemblies for some of the world’s leading medical device OEMs.



























