07.31.13
24. Toshiba
$3.97 Billion ($61.5B total)
KEY EXECUTIVES:
Hisao Tanaka, President & CEO, Toshiba Corp.
Satoshi Tsunakawa, President & CEO, Toshiba Medical Systems Corp.
Donald Fowler, President & CEO, Toshiba America Medical Systems
Calum G. Cunningham, VP, Business Operations, Toshiba America Medical Systems
Ted Nemetz, VP, Technology Services Business, Toshiba America Medical Systems
Scott Goodwin, VP of Sales, Toshiba America Medical Systems
Stuart Clarkson, director, MR Business Unit, Toshiba
NO. OF EMPLOYEES: 210,000 (total)
GLOBAL HEADQUARTERS: Tokyo, Japan
Toshiba Medical Systems Corp. and its U.S. subsidiary Toshiba America Medical Systems (in Tustin, Calif.) make medical diagnostic imaging systems, such as CT, X-ray and vascular, ultrasound, nuclear medicine and MRI systems, as well as information systems for medical facilities. A division of Japanese electronics behemoth Toshiba, the medical systems group is no newcomer to the medtech space. It has been in business for more than 80 years. In fact, the company—before the healthcare division was formed—was the first in Japan to begin research on X-ray tubes in 1914. The company also developed one of the world’s first X-ray machines in 1932 to help physicians manage a tuberculosis outbreak in Japan. A lot has happened in the last 100 years. Toshiba’s medical group claims its mission is to improve lives globally with cutting-edge imaging technology and solutions. Well, during fiscal 2012 (ended March 31, 2013), it seemed as if the company’s mission seemed to be stockpiling approvals and clearances from the U.S. Food and Drug Administration (FDA).
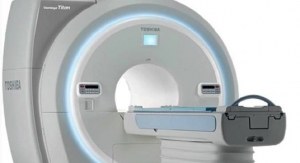
The company received FDA 510(k) clearance for the new Vantage Titan 1.5T series, which features the 8-, 16- and 32-channel magnetic resonance (MR) systems. The series offers a scalable solution with a full upgrade path from the 8-channel to the 32-channel system. In addition to these upgrades, the entire Titan 1.5T MR line was enhanced with a new modern, sleek exterior, including new covers and soft, patient-soothing lighting around the bore, according to the company. The company also claims the Titan 8-channel is a “workhorse” for most clinical applications.
In August last year, Toshiba received FDA clearance for its high-density 16-element flexible coil system, developed in partnership with Pewaukee, Wis.-based Neocoil, a medical imaging device company that specializes in magnetic resonance imaging radiofrequency (RF) coils. The new coil system makes it easier for clinicians to complete high-quality exams and improve diagnostic efficiency, the company claims.
“The 16-element MR coils feature a flexible design, enabling clinicians to use the coils for various exam types,” said Steve Nichols, chief operating officer, NeoCoil. “The lightweight coils come in two sizes and wrap around a patient’s anatomy, accommodating patients of various sizes.”
Available for the Vantage Titan 1.5T, the 16-element flexible coils conform more closely to the anatomy, greatly improving signal-to-noise ratio. The coils are available in medium and large sizes and ideally are suited for general orthopedic and body imaging of large and small patients. Where standard coils are not optimal, the flexible coils can be used for general-purpose as well as head, neck and spine imaging. Large patients are not forced into the hard plastic of traditional rigid coils. Adding to its ultrasound product line, following FDA clearance, the company rolled out the Aplio 500 and 300 ultrasound systems. The Aplio 500 and Aplio 300 combine advanced visualization capabilities, workflow automation tools and improved ergonomics for improved departmental efficiency and greater patient access, according to the company.
The Aplio 500 offers advanced visualization features, including Fly Thru and Smart Fusion. Fly Thru, which the company says is an industry first, uses four-dimensional ultrasound to “fly through” interiors of fluid-filled ducts and vessels for better exploration of lesions and masses and to assist in planning interventional procedures. Smart Fusion combines the best in imaging and synchronizes computed tomography (CT) or magnetic resonance with ultrasound, helping to locate hard-to-find lesions and aiding in ultrasound-guided biopsies. The Aplio 300 offers exceptional image quality in a smaller, more versatile platform designed to be a clinical workhorse for all routine ultrasound exams.
“Toshiba’s new ultrasound systems are designed to meet the clinical needs of hospitals everywhere,” said Tomohiro Hasegawa, director, Ultrasound Business Unit, Toshiba. “Aplio 500 has advanced features to help improve ultrasound diagnoses, while the Aplio 300 is a versatile system designed for many routine exams.”
Toshiba received FDA clearance of the Aquilion Prime 80 series CT system, the latest addition to the Aquilion CT product line. Producing higher-quality clinical images and reducing radiation exposure, the system can generate 80 unique slices per rotation, according to the company. The system is designed with in-field upgradeability to 160 slices, giving healthcare facilities the ability to perform a wide variety of advanced clinical procedures today and to grow as clinical needs expand. The combination of a 660-pound patient-weight-capacity couch with a large gantry bore makes the Aquilion Prime 80 series a good system for routine clinical scanning or bariatric patient studies, the company claims.
“The PRIME 80 series is an example of how Toshiba constantly develops new technologies to improve safety in imaging and patient care,” said Tim Nicholson, senior manager, Market Development, CT Business Unit, Toshiba. “Giving hospitals the opportunity to conduct advanced exams in mere seconds and grow clinical abilities based on patient needs results in improved patient care and overall departmental efficiency.”
Also in fiscal 2012, the FDA cleared the Adaptive Iterative Dose Reduction (AIDR) 3D (AIDR 3D), the company’s newest dose-reduction technology. AIDR 3D is Toshiba’s third-generation iterative dose reconstruction software, which has been enhanced to reduce radiation dose compared to conventional scanning. It is a sophisticated algorithm designed to work in both the raw data and image data space, reducing noise while attempting to maintain image quality. AIDR 3D can be integrated with SureExposure 3D, a software that calculates the minimum radiation exposure required for every exam. It adjusts the dose for each patient based on a pre-set, targeted level of image quality.
On the management side, Toshiba America Medical Systems named Satrajit Misra senior director of the CT Business Unit, where he will implement tactical and strategic marketing plans for Toshiba’s CT product line. Prior to joining Toshiba, Misra served as senior director and head of product marketing for Nuclear Medicine at Philips Healthcare and was a medical systems director of business management at Siemens. Toshiba America Medical Systems also named Calum Cunningham as vice president of
business operations. Cunningham will oversee a range of activities, from national quotes and bids, to market data submissions and reporting. He also is responsible for sales logistics management, including warehouse logistics, customer site planning, installation support, clinical applications and service training. Cunningham joined Toshiba in 2009 as senior vice president and general manager of Toshiba Medical Visualization Systems. Upon Toshiba’s merger with Minnetonka, Minn.-based Vital Images Inc. in 2011, he was named executive vice president of corporate and business development at Vital Images.
By the numbers, Toshiba’s healthcare unit had a good year, increasing sales 6.6 percent (in yen) to approximately $3.97 billion. The division reported operating income of approximately $251.4 million, up 37.8 percent.
$3.97 Billion ($61.5B total)
KEY EXECUTIVES:
Hisao Tanaka, President & CEO, Toshiba Corp.
Satoshi Tsunakawa, President & CEO, Toshiba Medical Systems Corp.
Donald Fowler, President & CEO, Toshiba America Medical Systems
Calum G. Cunningham, VP, Business Operations, Toshiba America Medical Systems
Ted Nemetz, VP, Technology Services Business, Toshiba America Medical Systems
Scott Goodwin, VP of Sales, Toshiba America Medical Systems
Stuart Clarkson, director, MR Business Unit, Toshiba
NO. OF EMPLOYEES: 210,000 (total)
GLOBAL HEADQUARTERS: Tokyo, Japan
Toshiba Medical Systems Corp. and its U.S. subsidiary Toshiba America Medical Systems (in Tustin, Calif.) make medical diagnostic imaging systems, such as CT, X-ray and vascular, ultrasound, nuclear medicine and MRI systems, as well as information systems for medical facilities. A division of Japanese electronics behemoth Toshiba, the medical systems group is no newcomer to the medtech space. It has been in business for more than 80 years. In fact, the company—before the healthcare division was formed—was the first in Japan to begin research on X-ray tubes in 1914. The company also developed one of the world’s first X-ray machines in 1932 to help physicians manage a tuberculosis outbreak in Japan. A lot has happened in the last 100 years. Toshiba’s medical group claims its mission is to improve lives globally with cutting-edge imaging technology and solutions. Well, during fiscal 2012 (ended March 31, 2013), it seemed as if the company’s mission seemed to be stockpiling approvals and clearances from the U.S. Food and Drug Administration (FDA).
The company received FDA 510(k) clearance for the new Vantage Titan 1.5T series, which features the 8-, 16- and 32-channel magnetic resonance (MR) systems. The series offers a scalable solution with a full upgrade path from the 8-channel to the 32-channel system. In addition to these upgrades, the entire Titan 1.5T MR line was enhanced with a new modern, sleek exterior, including new covers and soft, patient-soothing lighting around the bore, according to the company. The company also claims the Titan 8-channel is a “workhorse” for most clinical applications.
In August last year, Toshiba received FDA clearance for its high-density 16-element flexible coil system, developed in partnership with Pewaukee, Wis.-based Neocoil, a medical imaging device company that specializes in magnetic resonance imaging radiofrequency (RF) coils. The new coil system makes it easier for clinicians to complete high-quality exams and improve diagnostic efficiency, the company claims.
“The 16-element MR coils feature a flexible design, enabling clinicians to use the coils for various exam types,” said Steve Nichols, chief operating officer, NeoCoil. “The lightweight coils come in two sizes and wrap around a patient’s anatomy, accommodating patients of various sizes.”
Available for the Vantage Titan 1.5T, the 16-element flexible coils conform more closely to the anatomy, greatly improving signal-to-noise ratio. The coils are available in medium and large sizes and ideally are suited for general orthopedic and body imaging of large and small patients. Where standard coils are not optimal, the flexible coils can be used for general-purpose as well as head, neck and spine imaging. Large patients are not forced into the hard plastic of traditional rigid coils. Adding to its ultrasound product line, following FDA clearance, the company rolled out the Aplio 500 and 300 ultrasound systems. The Aplio 500 and Aplio 300 combine advanced visualization capabilities, workflow automation tools and improved ergonomics for improved departmental efficiency and greater patient access, according to the company.
The Aplio 500 offers advanced visualization features, including Fly Thru and Smart Fusion. Fly Thru, which the company says is an industry first, uses four-dimensional ultrasound to “fly through” interiors of fluid-filled ducts and vessels for better exploration of lesions and masses and to assist in planning interventional procedures. Smart Fusion combines the best in imaging and synchronizes computed tomography (CT) or magnetic resonance with ultrasound, helping to locate hard-to-find lesions and aiding in ultrasound-guided biopsies. The Aplio 300 offers exceptional image quality in a smaller, more versatile platform designed to be a clinical workhorse for all routine ultrasound exams.
“Toshiba’s new ultrasound systems are designed to meet the clinical needs of hospitals everywhere,” said Tomohiro Hasegawa, director, Ultrasound Business Unit, Toshiba. “Aplio 500 has advanced features to help improve ultrasound diagnoses, while the Aplio 300 is a versatile system designed for many routine exams.”
Toshiba received FDA clearance of the Aquilion Prime 80 series CT system, the latest addition to the Aquilion CT product line. Producing higher-quality clinical images and reducing radiation exposure, the system can generate 80 unique slices per rotation, according to the company. The system is designed with in-field upgradeability to 160 slices, giving healthcare facilities the ability to perform a wide variety of advanced clinical procedures today and to grow as clinical needs expand. The combination of a 660-pound patient-weight-capacity couch with a large gantry bore makes the Aquilion Prime 80 series a good system for routine clinical scanning or bariatric patient studies, the company claims.
“The PRIME 80 series is an example of how Toshiba constantly develops new technologies to improve safety in imaging and patient care,” said Tim Nicholson, senior manager, Market Development, CT Business Unit, Toshiba. “Giving hospitals the opportunity to conduct advanced exams in mere seconds and grow clinical abilities based on patient needs results in improved patient care and overall departmental efficiency.”
Also in fiscal 2012, the FDA cleared the Adaptive Iterative Dose Reduction (AIDR) 3D (AIDR 3D), the company’s newest dose-reduction technology. AIDR 3D is Toshiba’s third-generation iterative dose reconstruction software, which has been enhanced to reduce radiation dose compared to conventional scanning. It is a sophisticated algorithm designed to work in both the raw data and image data space, reducing noise while attempting to maintain image quality. AIDR 3D can be integrated with SureExposure 3D, a software that calculates the minimum radiation exposure required for every exam. It adjusts the dose for each patient based on a pre-set, targeted level of image quality.
On the management side, Toshiba America Medical Systems named Satrajit Misra senior director of the CT Business Unit, where he will implement tactical and strategic marketing plans for Toshiba’s CT product line. Prior to joining Toshiba, Misra served as senior director and head of product marketing for Nuclear Medicine at Philips Healthcare and was a medical systems director of business management at Siemens. Toshiba America Medical Systems also named Calum Cunningham as vice president of
business operations. Cunningham will oversee a range of activities, from national quotes and bids, to market data submissions and reporting. He also is responsible for sales logistics management, including warehouse logistics, customer site planning, installation support, clinical applications and service training. Cunningham joined Toshiba in 2009 as senior vice president and general manager of Toshiba Medical Visualization Systems. Upon Toshiba’s merger with Minnetonka, Minn.-based Vital Images Inc. in 2011, he was named executive vice president of corporate and business development at Vital Images.
By the numbers, Toshiba’s healthcare unit had a good year, increasing sales 6.6 percent (in yen) to approximately $3.97 billion. The division reported operating income of approximately $251.4 million, up 37.8 percent.