07.19.16
$5.5 Billion
KEY EXECUTIVES:
Dr. Ulf M. Schneider, Chairman and CEO, Fresenius Group
Rice Powell, Chairman and CEO, Fresenius Medical Care
Ronald Kuerbitz, CEO, Fresenius Medical Care North America
William Valle, President, Kidney Care, Fresenius Medical Care North America
Mark Constanzo, President, Renal Therapies Group, Fresenius Medical Care North America
Mats Henriksson, CEO, Fresenius Kabi
John Ducker, President and CEO, Fresenius Kabi USA
Dean A. Gregory, President, Medical Devices, Region North America, Fresenius Kabi USA
NUMBER OF EMPLOYEES: 222,305 (Fresenius Group total)
GLOBAL HEADQUARTERS: Bad Homburg, Germany
The Gaza conflict has been raging on for over a decade now. In addition to many lives claimed, the conflict also wreaks havoc on the region’s economy—much of the local population is dependent on international aid to survive. The situation in Syria has had nearly identical effects, leaving millions of Syrians without the supplies they so drastically require.
Companioned with the economic situation is a sharp dropoff in the quality of medical care in both regions. Amidst this dire situation in April 2015, Fresenius Kabi donated lifesaving medicines to the aid organization action medeor e.V., which provides medication transfer to local relief organizations, supporting hospitals with infusion solutions in both Gaza and Syria. Both groups received 150,000 bottles, and Fresenius Kabi covered a portion of the transportation costs.
“We thank Fresenius Kabi for its generous support to the world’s most vulnerable people,” said Dirk Angemeer, action medeor’s head of export and purchasing department. “This donation really makes a difference. We are looking forward to continuing our successful cooperation.”
Fresenius Group is a parent company consisting of four business divisions, each responsible for their own worldwide operations: Fresenius Medical Care (dialysis products and services), Fresenius Kabi (therapy and care of the chronically ill, providing IV drugs, clinical nutrition, infusion therapy, medical devices/transfusion technology), Fresenius Helios (hospital operator), and Fresenius Vamed (manages projects and provides services for hospitals/healthcare facilities).
Most of Fresenius’ revenue isn’t derived from pure medical device offerings, but the gains achieved in fiscal year 2015 (ended Dec. 31) from the combination of the company’s dialysis products, medical devices/transfusion technology, and infusion therapy products were enough to keep Fresenius in the Top 30 with $5.5 billion in sales. Although Fresenius Group’s total sales of $27.6 billion marked an impressive 18.9 percent growth over the previous year, 2015 revenue from the aforementioned focus sectors actually fell 6.7 percent from 2014.
Fresenius Medical Care’s dialysis products experienced a 7 percent decline from 2014, with 3.35 billion Euros in sales. The North American market was not the cause for this—sales increased by 4 percent for that region. The main issue was the company’s international sales (which consists of Europe, Middle East, Africa, Asia-Pacific, and Latin America), which fell 9 percent from 2014. The company cited fluctuating currency translation as the main reason behind the decrease, but Fresenius didn’t seem to regard foreign monies as an issue, because executives claimed their focus was directed elsewhere—in further expanding its hospital network, and integrating acquisitions made in 2014.
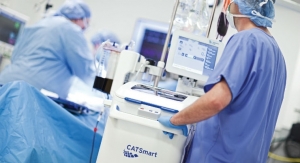
Fresenius Kabi had quite a strong fiscal year 2015, growing sales 15.7 percent to 5.9 billion euros. However, the company’s IV drugs are mainly responsible for this growth. Infusion therapy sales in 2015 totaled 914 million euros, falling 6 percent from the prior year. Medical devices/transfusion technology’s sales of 1.05 billion euros was the only medical device segment that represented an increase, though its not-insignificant 7.7 percent growth was not enough to offset dialysis and infusion therapy losses. The company cited U.S. sales of the Agilia infusion pump as the chief driver for growth in this sector. Also responsible for this increase was introduction of the CATSmart, a continuous autotransfusion system. CE mark approval was also achieved for the Agilia products ProNeo, Homecare, and Connect during the year.
In May 2015, Fresenius Medical Care AG & Co. KGaA (the dialysis products and services provider) launched multiFiltratePRO, a continuous renal replacement therapy system that treats acute kidney failure in critically ill patients in the ICU. The new system’s features support physicians and caregivers with high workloads and working pressure with a large touch screen monitor providing comprehensive information. The fluid is heated to the correct temperature by way of fully integrated fluid heaters, and a “Care Mode” helps reduce alarm fatigue by preventing unnecessary alarms.
“Over the time we expect to expand this new therapy platform,” said Dr. Olaf Schermeier, Fresenius’ CEO for global research and development. “The reliable and simple user interface helps reduce the demand on personnel while providing the highest treatment quality. And spending less time with handling complex technology offers the opportunity to pay more attention to the patient itself.”
Fresenius Medical Care also announced a partnership with Debiotech SA, a Swiss medical device developer with expertise in miniaturized medical equipment and devices. The partnership intended to develop a portfolio of peritoneal dialysis (PD) technologies that are lightweight and compact enough to dialyze at home. Worldwide, over 2.6 million end stage renal disease patients regularly undergo dialysis treatment. “We are dedicated to continuing to produce home dialysis machines that are as easy and convenient as possible for our customers to operate, because this is what they demand and deserve,” said Mark Costanzo, president of Fresenius Medical Care’s Renal Therapies Group.
KEY EXECUTIVES:
Dr. Ulf M. Schneider, Chairman and CEO, Fresenius Group
Rice Powell, Chairman and CEO, Fresenius Medical Care
Ronald Kuerbitz, CEO, Fresenius Medical Care North America
William Valle, President, Kidney Care, Fresenius Medical Care North America
Mark Constanzo, President, Renal Therapies Group, Fresenius Medical Care North America
Mats Henriksson, CEO, Fresenius Kabi
John Ducker, President and CEO, Fresenius Kabi USA
Dean A. Gregory, President, Medical Devices, Region North America, Fresenius Kabi USA
NUMBER OF EMPLOYEES: 222,305 (Fresenius Group total)
GLOBAL HEADQUARTERS: Bad Homburg, Germany
The Gaza conflict has been raging on for over a decade now. In addition to many lives claimed, the conflict also wreaks havoc on the region’s economy—much of the local population is dependent on international aid to survive. The situation in Syria has had nearly identical effects, leaving millions of Syrians without the supplies they so drastically require.
Companioned with the economic situation is a sharp dropoff in the quality of medical care in both regions. Amidst this dire situation in April 2015, Fresenius Kabi donated lifesaving medicines to the aid organization action medeor e.V., which provides medication transfer to local relief organizations, supporting hospitals with infusion solutions in both Gaza and Syria. Both groups received 150,000 bottles, and Fresenius Kabi covered a portion of the transportation costs.
“We thank Fresenius Kabi for its generous support to the world’s most vulnerable people,” said Dirk Angemeer, action medeor’s head of export and purchasing department. “This donation really makes a difference. We are looking forward to continuing our successful cooperation.”
Fresenius Group is a parent company consisting of four business divisions, each responsible for their own worldwide operations: Fresenius Medical Care (dialysis products and services), Fresenius Kabi (therapy and care of the chronically ill, providing IV drugs, clinical nutrition, infusion therapy, medical devices/transfusion technology), Fresenius Helios (hospital operator), and Fresenius Vamed (manages projects and provides services for hospitals/healthcare facilities).
Most of Fresenius’ revenue isn’t derived from pure medical device offerings, but the gains achieved in fiscal year 2015 (ended Dec. 31) from the combination of the company’s dialysis products, medical devices/transfusion technology, and infusion therapy products were enough to keep Fresenius in the Top 30 with $5.5 billion in sales. Although Fresenius Group’s total sales of $27.6 billion marked an impressive 18.9 percent growth over the previous year, 2015 revenue from the aforementioned focus sectors actually fell 6.7 percent from 2014.
Fresenius Medical Care’s dialysis products experienced a 7 percent decline from 2014, with 3.35 billion Euros in sales. The North American market was not the cause for this—sales increased by 4 percent for that region. The main issue was the company’s international sales (which consists of Europe, Middle East, Africa, Asia-Pacific, and Latin America), which fell 9 percent from 2014. The company cited fluctuating currency translation as the main reason behind the decrease, but Fresenius didn’t seem to regard foreign monies as an issue, because executives claimed their focus was directed elsewhere—in further expanding its hospital network, and integrating acquisitions made in 2014.
Fresenius Kabi had quite a strong fiscal year 2015, growing sales 15.7 percent to 5.9 billion euros. However, the company’s IV drugs are mainly responsible for this growth. Infusion therapy sales in 2015 totaled 914 million euros, falling 6 percent from the prior year. Medical devices/transfusion technology’s sales of 1.05 billion euros was the only medical device segment that represented an increase, though its not-insignificant 7.7 percent growth was not enough to offset dialysis and infusion therapy losses. The company cited U.S. sales of the Agilia infusion pump as the chief driver for growth in this sector. Also responsible for this increase was introduction of the CATSmart, a continuous autotransfusion system. CE mark approval was also achieved for the Agilia products ProNeo, Homecare, and Connect during the year.
In May 2015, Fresenius Medical Care AG & Co. KGaA (the dialysis products and services provider) launched multiFiltratePRO, a continuous renal replacement therapy system that treats acute kidney failure in critically ill patients in the ICU. The new system’s features support physicians and caregivers with high workloads and working pressure with a large touch screen monitor providing comprehensive information. The fluid is heated to the correct temperature by way of fully integrated fluid heaters, and a “Care Mode” helps reduce alarm fatigue by preventing unnecessary alarms.
“Over the time we expect to expand this new therapy platform,” said Dr. Olaf Schermeier, Fresenius’ CEO for global research and development. “The reliable and simple user interface helps reduce the demand on personnel while providing the highest treatment quality. And spending less time with handling complex technology offers the opportunity to pay more attention to the patient itself.”
Fresenius Medical Care also announced a partnership with Debiotech SA, a Swiss medical device developer with expertise in miniaturized medical equipment and devices. The partnership intended to develop a portfolio of peritoneal dialysis (PD) technologies that are lightweight and compact enough to dialyze at home. Worldwide, over 2.6 million end stage renal disease patients regularly undergo dialysis treatment. “We are dedicated to continuing to produce home dialysis machines that are as easy and convenient as possible for our customers to operate, because this is what they demand and deserve,” said Mark Costanzo, president of Fresenius Medical Care’s Renal Therapies Group.