Videos
U.S. Patent Granted to SciBase for Electrodes with Micro-Needles
U.S. Patent Granted to SciBase for Electrodes with Micro-Needles
The new patents all are valid through 2023.
09.10.15
SciBase, a Swedish medtech company that develops and markets a new method for early detection of malignant melanoma, has received approval from the U.S. Patent and Trademark Office regarding its patent application for its electrodes with micro-needles. The method already is approved in 12 European countries, Australia and Canada. All patents are valid through 2023.
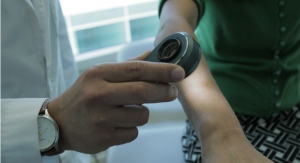
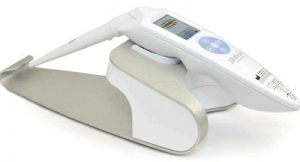
SciBase’s solution Nevisense is based on a technology called Electrical Impedance Spectroscopy (EIS), which uses the varying electrical properties of human tissue to categorize cellular structures and thereby detect malignancies. Malignant melanoma is a fast growing kind of skin cancer, which appears in the epidermis right under the cornified layer. Nevisense’s electrodes are equipped with microscopic needles that measure the electric impedance in the cells below, where malignant melanoma appears. The technique is essential for detecting the early stages of skin cancer.
EIS is a measure of the overall resistance within the tissue at alternating currents of various frequencies. It is measured by applying an unnoticeable alternating potential between the bars on the tip of the probe. In order to cover the lesion in both width and depth, the measurement is performed in 10 permutations covering both shallow measurements between neighboring electrode bars as well as deeper measurements between more distant electrode bars.EIS reflects different cellular properties in different frequency regions. In general, EIS measurements at low frequencies are affected by the extracellular environment, whereas both the intra- and extracellular environments affect measurements at higher frequencies. The frequencies used by Nevisense (1 kHz – 2.5 MHz) relate to clinically relevant properties, such as composition of intra- and extracellular environments, cell shape and size, and cell membrane composition, all of which are similar to those used by histopathologists to diagnose skin cancer.
Within seconds, an advanced algorithm is used to classify the lesion based on measurement data from both the lesion and the reference measurement. This classifier, developed in several iterations with data from multiple clinical studies, has proven to increase accuracy in melanoma detection in the pivotal study – the world’s largest clinical study of its kind.
”Right now, Nevisense is in the approval process for sales and marketing in the United States, which is a future key market for us. The approval of our patent is thus an important step in our global expansion. Electrodes with micro-needles can potentially be used to detect other types of cancer as well,” said Simon Grant, CEO of SciBase.
SciBase holds a total of 49 registered patents and currently have another 12 active patent applications pending in six patent families.
Skin cancer is the most common malignancy in the world, accounting for nearly half of all cancers, according to industry data. It has been estimated that nearly half of all Americans who live to age 65 will develop skin cancer at least once. Malignant melanoma is the most fatal form of skin cancer causing the majority (75 percent) of deaths related to skin cancer, although it only accounts for 4 percent of all skin cancer cases. Worldwide, doctors diagnose about 230,000 new cases of melanoma yearly.
Headquartered in Stockholm, Sweden, SciBase AB has developed a point-of-care device that helps doctors to detect malignant melanoma, the most dangerous type of skin cancer. SciBase was founded by Stig Ollmar, Associate Professor at The Karolinska Institute in Stockholm, Sweden. Nevisense is CE marked in Europe, has TGA approval in Australia, and is awaiting U.S. Food and Drug Administration clearance in the United States.
A demonstration of the Nevisense product can be seen in the video below:
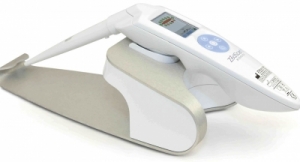
SciBase’s solution Nevisense is based on a technology called Electrical Impedance Spectroscopy (EIS), which uses the varying electrical properties of human tissue to categorize cellular structures and thereby detect malignancies. Malignant melanoma is a fast growing kind of skin cancer, which appears in the epidermis right under the cornified layer. Nevisense’s electrodes are equipped with microscopic needles that measure the electric impedance in the cells below, where malignant melanoma appears. The technique is essential for detecting the early stages of skin cancer.
EIS is a measure of the overall resistance within the tissue at alternating currents of various frequencies. It is measured by applying an unnoticeable alternating potential between the bars on the tip of the probe. In order to cover the lesion in both width and depth, the measurement is performed in 10 permutations covering both shallow measurements between neighboring electrode bars as well as deeper measurements between more distant electrode bars.EIS reflects different cellular properties in different frequency regions. In general, EIS measurements at low frequencies are affected by the extracellular environment, whereas both the intra- and extracellular environments affect measurements at higher frequencies. The frequencies used by Nevisense (1 kHz – 2.5 MHz) relate to clinically relevant properties, such as composition of intra- and extracellular environments, cell shape and size, and cell membrane composition, all of which are similar to those used by histopathologists to diagnose skin cancer.
Within seconds, an advanced algorithm is used to classify the lesion based on measurement data from both the lesion and the reference measurement. This classifier, developed in several iterations with data from multiple clinical studies, has proven to increase accuracy in melanoma detection in the pivotal study – the world’s largest clinical study of its kind.
”Right now, Nevisense is in the approval process for sales and marketing in the United States, which is a future key market for us. The approval of our patent is thus an important step in our global expansion. Electrodes with micro-needles can potentially be used to detect other types of cancer as well,” said Simon Grant, CEO of SciBase.
SciBase holds a total of 49 registered patents and currently have another 12 active patent applications pending in six patent families.
Skin cancer is the most common malignancy in the world, accounting for nearly half of all cancers, according to industry data. It has been estimated that nearly half of all Americans who live to age 65 will develop skin cancer at least once. Malignant melanoma is the most fatal form of skin cancer causing the majority (75 percent) of deaths related to skin cancer, although it only accounts for 4 percent of all skin cancer cases. Worldwide, doctors diagnose about 230,000 new cases of melanoma yearly.
Headquartered in Stockholm, Sweden, SciBase AB has developed a point-of-care device that helps doctors to detect malignant melanoma, the most dangerous type of skin cancer. SciBase was founded by Stig Ollmar, Associate Professor at The Karolinska Institute in Stockholm, Sweden. Nevisense is CE marked in Europe, has TGA approval in Australia, and is awaiting U.S. Food and Drug Administration clearance in the United States.
A demonstration of the Nevisense product can be seen in the video below:
Related Searches: