Chris Delporte, Editor10.17.13
“Far and away the best prize that life has to offer is the chance to work hard at work worth doing.”
Though global in its reach and scope, the medical device industry at its most fundamental—without a doubt—is a truly American success story. That’s not patriotic hyperbole. The United States accounts for more than 40 percent of the global medtech market. It also has a positive trade balance in medical devices, exporting $5.4 billion more than in imports (the industry exports approximately $33 billion in goods annually).
It represents one of the healthiest manufacturing sectors in the country. According to the Advanced Medical Technology Association (AdvaMed), the industry’s largest lobby group, the 7,000 firms that make up the U.S. medical device industry directly employ almost 423,000 people. Indirectly, accounting for peripheral jobs and services, the medtech sector has an overall impact on nearly 2 million U.S. workers. The industry’s average annual salary is $58,000 (the overall U.S. average is approximately $48,000, according to the U.S. Bureau of Economic Analysis), and the U.S. Bureau of Labor Statistics predicts job growth in medtech. For example, medical device sales jobs are expected to grow 14.3 percent between now and 2020.
Incidentally, it’s an industry built strong on the innovation of small companies. AdvaMed’s data notes that 62 percent of device companies have fewer than 20 employees and only 2 percent employ more than 500.
These numbers paint the picture of a robust industry going about its lifesaving work. But what are these industry professionals—salespeople, engineers, manufacturing technicians, reimbursement specialists, regulatory and quality managers, and so many others—thinking? Beyond the dollars-and-cents data of who earns how much, is there an industry mood? Of course, that’s a little more difficult to capture.
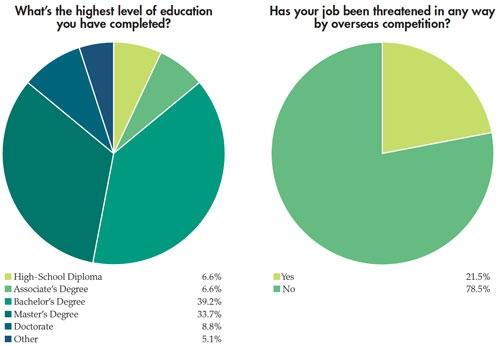
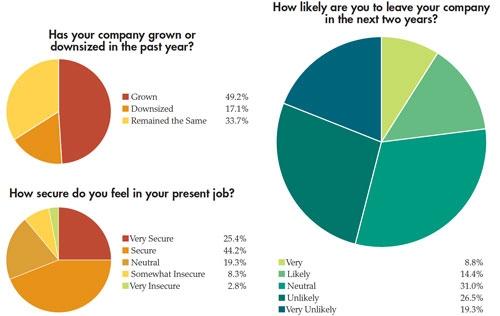
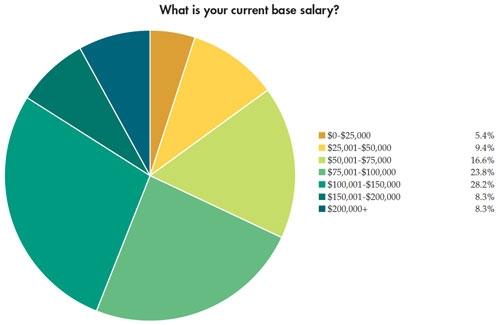
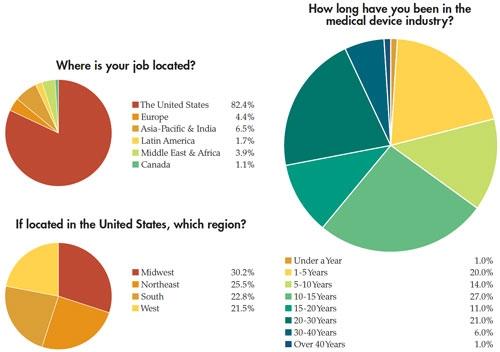
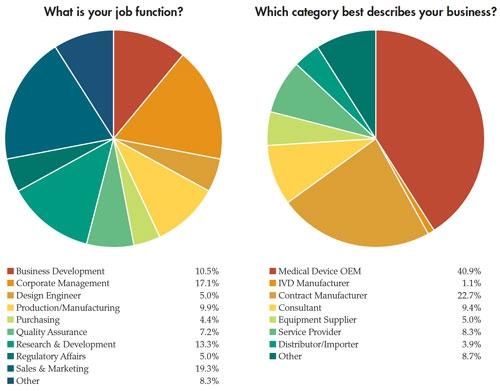 In addition to the paycheck picture, Medical Product Outsourcing’s annual salary survey attempts to take a snapshot of what our readers are feeling about their jobs and the state of the industry. This year’s survey, despite the dreaded medical device tax now in place, seems to reflect—as in years past—a mostly optimistic, well-educated group of professionals with years of industry experience. This year’s pool of survey takers predominantly was male (82 percent). Thirty-nine percent have a four-year college degree and nearly 34 percent have an advanced degree. More than half of the 181 respondents make more than $75,000 per year, and the majority has been in the medical device industry for more than 10 years. In fact, 21 percent have 20-30 years of experience. Nearly 70 percent feel “secure” or “very secure” in their current jobs.
In addition to the paycheck picture, Medical Product Outsourcing’s annual salary survey attempts to take a snapshot of what our readers are feeling about their jobs and the state of the industry. This year’s survey, despite the dreaded medical device tax now in place, seems to reflect—as in years past—a mostly optimistic, well-educated group of professionals with years of industry experience. This year’s pool of survey takers predominantly was male (82 percent). Thirty-nine percent have a four-year college degree and nearly 34 percent have an advanced degree. More than half of the 181 respondents make more than $75,000 per year, and the majority has been in the medical device industry for more than 10 years. In fact, 21 percent have 20-30 years of experience. Nearly 70 percent feel “secure” or “very secure” in their current jobs.
The figures outlined above and the ones reflected in the charts and graphs in this article are impressive—and seem positive. But when asked in a few questions to elaborate on their feelings, the responses belied the confidence of the non-open-ended questions.
When asked how the U.S. and/or world economy has impacted their jobs, most of the responses—surprisingly—were of the “not much at all” variety, while many said their jobs were “absolutely,” “definitely,” or even “horribly” affected. At one end of the spectrum, one respondent said: “Our manufacturing facility has been doing well despite the economy. We have made record profits the last few months.” Yet another wasn’t as bullish, noting that “Lack of orders from customers has led to layoffs, lack of bonuses, and lack of raises for several years.” One of the contract manufacturers taking the survey said, “Business is good. More companies are outsourcing their product development.” Another said outsourcing (I wonder if they meant offshoring) was “killing U.S.
competitiveness.” Yet, despite the wide disparity in responses to this question, nearly 49 percent of those surveyed reported business growth in the last year, with nearly 79 percent not feeling the impact of overseas competition.
The question “What concerns you most, if anything, about the medical device industry at present?” produced some of the most interesting responses.
 More than a quarter of the respondents to this question noted that despite not being immediately impacted thus far, they were still waiting to see how the medical device tax would affect their businesses. Others said the Affordable Care Act, commonly called Obamacare, and its effect on patient and payers is dangling a huge question mark above the medtech market. Many predicted the worst. Some were a little more pragmatic in their approach.
More than a quarter of the respondents to this question noted that despite not being immediately impacted thus far, they were still waiting to see how the medical device tax would affect their businesses. Others said the Affordable Care Act, commonly called Obamacare, and its effect on patient and payers is dangling a huge question mark above the medtech market. Many predicted the worst. Some were a little more pragmatic in their approach.
One respondent wrote, “The reform does not concern me. It is the lack of research done before it was proposed and approved. This country needs something, but something well-vetted that does not hinder healthcare facilities or medical device companies from growing innovation and profit, something beneficial to those who really need healthcare.”
Regulatory “unknowns” also were top-reported concerns.
One person responded simply: “more taxes and stricter regulation.” Many expressed their fear that the “burdensome” (that word was used a lot) regulatory environment at the U.S. Food and Drug Administration (FDA) would impact the country’s status as a medtech leader and innovator.
 “The overly long FDA approval process is driving medical device development overseas, making it difficult for U.S. manufacturers to be involved in early stage development and prototyping/trial work,” wrote one industry professional. “This, in effect, locks us out of being able to help shape and innovate new medical devices coming to market and relegates domestic manufacturers to ‘commodity production’ status.”
“The overly long FDA approval process is driving medical device development overseas, making it difficult for U.S. manufacturers to be involved in early stage development and prototyping/trial work,” wrote one industry professional. “This, in effect, locks us out of being able to help shape and innovate new medical devices coming to market and relegates domestic manufacturers to ‘commodity production’ status.”
Another person noted that he or she was most concerned about “the effect of more stringent regulatory requirements—which are necessary—on smaller companies trying to get new products to market.”
Many respondents’ reactions reflected nervousness about the number of issues swirling at once in the medical device industry at present.
“So many [things] are moving—for example changing patent law, other regulatory issues, funding scenarios, and the instability of large device and pharma companies all cause concern,” wrote one respondent.
In fact, reductions in venture capital funding were cited by about 15 percent of those surveyed.
According to one of the respondents of this year’s MPO survey, the state of the industry and healthcare overall has become “a race to the bottom” that gets in the way of—as Teddy Roosevelt put it—“work worth doing.”
“I’m worried about the chilling effect [of] the healthcare law, the [device] tax, FDA regulations, [and] limited funding are going to have on innovation and patient care,” he or she wrote. “It’s not about just providing cheap care. It’s about the best care, which brings down costs overall in a perfect world. We need to truly transform the system for quality care and outcomes. That will benefit physicians, patients and the device companies.”
—Teddy Roosevelt
Though global in its reach and scope, the medical device industry at its most fundamental—without a doubt—is a truly American success story. That’s not patriotic hyperbole. The United States accounts for more than 40 percent of the global medtech market. It also has a positive trade balance in medical devices, exporting $5.4 billion more than in imports (the industry exports approximately $33 billion in goods annually).
It represents one of the healthiest manufacturing sectors in the country. According to the Advanced Medical Technology Association (AdvaMed), the industry’s largest lobby group, the 7,000 firms that make up the U.S. medical device industry directly employ almost 423,000 people. Indirectly, accounting for peripheral jobs and services, the medtech sector has an overall impact on nearly 2 million U.S. workers. The industry’s average annual salary is $58,000 (the overall U.S. average is approximately $48,000, according to the U.S. Bureau of Economic Analysis), and the U.S. Bureau of Labor Statistics predicts job growth in medtech. For example, medical device sales jobs are expected to grow 14.3 percent between now and 2020.
Incidentally, it’s an industry built strong on the innovation of small companies. AdvaMed’s data notes that 62 percent of device companies have fewer than 20 employees and only 2 percent employ more than 500.
These numbers paint the picture of a robust industry going about its lifesaving work. But what are these industry professionals—salespeople, engineers, manufacturing technicians, reimbursement specialists, regulatory and quality managers, and so many others—thinking? Beyond the dollars-and-cents data of who earns how much, is there an industry mood? Of course, that’s a little more difficult to capture.
The figures outlined above and the ones reflected in the charts and graphs in this article are impressive—and seem positive. But when asked in a few questions to elaborate on their feelings, the responses belied the confidence of the non-open-ended questions.
When asked how the U.S. and/or world economy has impacted their jobs, most of the responses—surprisingly—were of the “not much at all” variety, while many said their jobs were “absolutely,” “definitely,” or even “horribly” affected. At one end of the spectrum, one respondent said: “Our manufacturing facility has been doing well despite the economy. We have made record profits the last few months.” Yet another wasn’t as bullish, noting that “Lack of orders from customers has led to layoffs, lack of bonuses, and lack of raises for several years.” One of the contract manufacturers taking the survey said, “Business is good. More companies are outsourcing their product development.” Another said outsourcing (I wonder if they meant offshoring) was “killing U.S.
competitiveness.” Yet, despite the wide disparity in responses to this question, nearly 49 percent of those surveyed reported business growth in the last year, with nearly 79 percent not feeling the impact of overseas competition.
The question “What concerns you most, if anything, about the medical device industry at present?” produced some of the most interesting responses.
One respondent wrote, “The reform does not concern me. It is the lack of research done before it was proposed and approved. This country needs something, but something well-vetted that does not hinder healthcare facilities or medical device companies from growing innovation and profit, something beneficial to those who really need healthcare.”
Regulatory “unknowns” also were top-reported concerns.
One person responded simply: “more taxes and stricter regulation.” Many expressed their fear that the “burdensome” (that word was used a lot) regulatory environment at the U.S. Food and Drug Administration (FDA) would impact the country’s status as a medtech leader and innovator.
Many respondents’ reactions reflected nervousness about the number of issues swirling at once in the medical device industry at present.
“So many [things] are moving—for example changing patent law, other regulatory issues, funding scenarios, and the instability of large device and pharma companies all cause concern,” wrote one respondent.
In fact, reductions in venture capital funding were cited by about 15 percent of those surveyed.
According to one of the respondents of this year’s MPO survey, the state of the industry and healthcare overall has become “a race to the bottom” that gets in the way of—as Teddy Roosevelt put it—“work worth doing.”
“I’m worried about the chilling effect [of] the healthcare law, the [device] tax, FDA regulations, [and] limited funding are going to have on innovation and patient care,” he or she wrote. “It’s not about just providing cheap care. It’s about the best care, which brings down costs overall in a perfect world. We need to truly transform the system for quality care and outcomes. That will benefit physicians, patients and the device companies.”



























