Michael Barbella, Managing Editor02.01.22
Timing truly is central to business success.
Microsoft, for example, could very well have been the world’s first trillion-dollar company had the public warmed to its tablet at the millennium’s dawning. Similarly, the world was not quite ready to embrace the future when Bell Labs unveiled its Picturephone in 1964 (imagine how different life would be today had it become commonplace back then).
Surprisingly, biotechnology firm Vaxxas hasn’t encountered that same dead end despite its invention being a bit ahead of its own time. The privately-held company is developing needle-free vaccination technology originally hatched by the Australian Institute of Bioengineering & Nanotechnology at The University of Queensland.
The technology uses a proprietary high-density micro-projection array patch (HD-MAP) to streamline and improve vaccine delivery. The postage stamp-sized silicon patch contains tens of thousands of projections 200-300 microns in length that release vaccine antigens directly to immune cells sitting just below the skin’s surface.
Preclinical studies have shown the Nanopatch to be considerably more effective than conventional vaccine delivery systems, with as little as 1/100th of its dose eliciting the same immune response as a “full” portion through intramuscular injection. Moreover, Nanopatch’s dry-coating technology eliminates the need for vaccine refrigeration during storage and transportation, thereby eliminating the resource burden of maintaining a cold chain.
“Based on our results, we believe that Vaxxas’ HD-MAP could offer a compelling solution that importantly could use less vaccine and potentially could be readily distributed without refrigeration for self-administration,” David A. Muller, Advance Queensland Industry Research Fellow, School of Chemistry and Molecular Biosciences, The University of Queensland, said last June. “This combination could make the HD-MAP extremely well suited to support the massive need for global population vaccination and indeed, we believe that HD-MAP offers a superior alternative to conventional needle-and-syringe.”
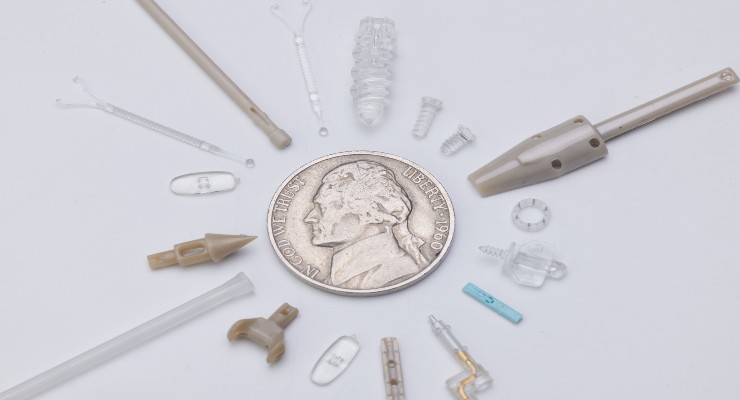
That superiority lies in the microscopic projections responsible for delivering vaccines. Those projections likely were created through micromolding, a highly specialized manufacturing process that produces extremely small, high-precision thermoplastic components with micron tolerances. This technique has become integral to medical device manufacturing of late as devices continue to shrink in size and scale.
MPO’s feature “Big Shots” details the trends and market forces driving micromolding in the medical device industry. Kenny Freitag, sales manager, Specialty Molding and Medical Tubing, at Spectrum Plastics Group, was among the more than one dozen experts interviewed for the feature. His full input is provided in the following Q&A:
Michael Barbella: What are the latest trends in micromolding technology and services?
Kenny Freitag: Companies are designing new products that feature miniaturization, high precision, and tight tolerances—all of which require micromolding. This is especially true for small parts in the life sciences and medical device industries, including implantables. Customers are also asking for new exotic, highly engineered materials rather than standard thermoplastics, especially for high-temperature applications. As features get smaller and smaller, Spectrum continuously improves its system controls and technologies to enable even greater control over shot size, measuring systems, and equipment monitors.
Barbella: What are customers demanding or expecting of their micromolded products and have these demands/expectations changed in recent years?
Freitag: Customers want precision and accuracy. They seek molders that will share their deep knowledge and experience about material science and molding technologies during the design for manufacturing (DFM) process to make their designs better and easier to produce. They expect us to find solutions to their toughest design challenges—smaller parts, advanced materials, improved functionality, tighter tolerances, improved outcomes, and even regulatory assistance. Speed is also in the highest demand—both in manufacturing the product and getting it to market—so vertically integrated services are always an added benefit for customers.
Barbella: How have advances in materials impacted micromolding technology?
Freitag: Improving functionality and performance of a product is not always just a matter of relying on a better technology—it can also be accomplished using advanced materials that have specific chemical and physical properties. There is steady advancement in new or improved materials that can be micromolded with high precision—for example, polyether ether ketone, or PEEK, can be formulated with different fillers that enhance mechanical performance. Micromolding PEEK, however, requires a solid understanding of its melt characteristics, such as flow path. Temperature must also be tightly controlled. Using highly engineered materials that enhance functionality also gives engineers a wider range of design options.
Barbella: Please discuss the challenges and complexities involved in micromolding tooling design. How can these challenges be overcome?
Freitag: Probably the biggest challenge is miniaturizing parts and duplicating them in a high-volume production and manufacturing environment. It is absolutely imperative to create robust and repeatable processes for mircomolding parts. Mold designs evolve as materials evolve. Miniaturization creates challenges with understanding material flow and shear, for example. Automation can certainly improve micromolding efficiency, but only if unit volumes are high enough. Tooling is very complex, so there is a real issue with finding tooling shops and vendors with skilled personnel.
Barbella: Design for manufacturability (DFM) is critically important in micromolding. How is this different than conventional DFM?
Freitag: It is mostly a challenge of scale. Because we are making smaller parts, we are putting more thought into the tool design and how we are going to manufacture these miniaturized components. Plastic mold flow analysis is different for a small part since less plastic is involved, therefore requiring more tooling design time and iterations. Perhaps the greatest focus is on mold design to ensure smooth and cost-effective production—with less space for gates and runners, sometimes the mold needs to be redesigned to better use the limited space.
Barbella: What medtech specialty (cardiology, wearables, orthopedics, etc.) presents the greatest challenge in to producing micro-molded parts and why? Which present the greatest opportunity?
Freitag: Minimally invasive procedures such as those used in cardiology and structural heart applications present the greatest challenge due to the fact the improved access and delivery systems are allowing device designers to incorporate more and smaller features. For example, a catheter that fits within a narrow vessel may house multiple lumens, each of which will contain wires, sutures, or electrodes. The corresponding molded components at the distal tip or housed within the handle of the delivery system will likewise have a high density of tiny features and cavities to manage and guide those respective components. This is where micromolding capabilities, alongside advanced extrusion and catheter manufacturing, becomes critical to the success of next-generation interventional devices.
Barbella: How do quality systems play a part in micromolding? Why is this so important part of the process?
Freitag: Measurement systems and metrology must be capable of detecting and/or fixing variances in these high-precision, tight-tolerance manufacturing environments. Integrated measurements are critical to the micromolding process. An efficient approach is measuring parts optically while they are being produced in-line, rather than offline. Non-contact vision inspection systems can detect flaws as small as a few microns. Any variances can be identified in real time and fixed, minimizing defective parts, material waste, and downtime.
Barbella: How might the medtech micromolding industry evolve over the next five years?
Freitag: Parts will continue to become smaller and more complex, challenging the limits of molding technologies. Materials will be developed that are specifically for micromolding processes that have greater functionality and can hold tighter tolerances. Ultimately, the Internet of things will allow the integration of sensor technologies, machine-to-machine communication, artificial intelligence, and cloud-based platforms to become integrated into micromolding systems, maximize production quality and efficiency, and reduce operational costs and downtime. Different manufacturing technologies (machining, lasers, additive manufacturing, and molding) are also being combined in creative ways to create “hybrid equipment” where these technologies can complement each other in making a part.
Microsoft, for example, could very well have been the world’s first trillion-dollar company had the public warmed to its tablet at the millennium’s dawning. Similarly, the world was not quite ready to embrace the future when Bell Labs unveiled its Picturephone in 1964 (imagine how different life would be today had it become commonplace back then).
Surprisingly, biotechnology firm Vaxxas hasn’t encountered that same dead end despite its invention being a bit ahead of its own time. The privately-held company is developing needle-free vaccination technology originally hatched by the Australian Institute of Bioengineering & Nanotechnology at The University of Queensland.
The technology uses a proprietary high-density micro-projection array patch (HD-MAP) to streamline and improve vaccine delivery. The postage stamp-sized silicon patch contains tens of thousands of projections 200-300 microns in length that release vaccine antigens directly to immune cells sitting just below the skin’s surface.
Preclinical studies have shown the Nanopatch to be considerably more effective than conventional vaccine delivery systems, with as little as 1/100th of its dose eliciting the same immune response as a “full” portion through intramuscular injection. Moreover, Nanopatch’s dry-coating technology eliminates the need for vaccine refrigeration during storage and transportation, thereby eliminating the resource burden of maintaining a cold chain.
“Based on our results, we believe that Vaxxas’ HD-MAP could offer a compelling solution that importantly could use less vaccine and potentially could be readily distributed without refrigeration for self-administration,” David A. Muller, Advance Queensland Industry Research Fellow, School of Chemistry and Molecular Biosciences, The University of Queensland, said last June. “This combination could make the HD-MAP extremely well suited to support the massive need for global population vaccination and indeed, we believe that HD-MAP offers a superior alternative to conventional needle-and-syringe.”
That superiority lies in the microscopic projections responsible for delivering vaccines. Those projections likely were created through micromolding, a highly specialized manufacturing process that produces extremely small, high-precision thermoplastic components with micron tolerances. This technique has become integral to medical device manufacturing of late as devices continue to shrink in size and scale.
MPO’s feature “Big Shots” details the trends and market forces driving micromolding in the medical device industry. Kenny Freitag, sales manager, Specialty Molding and Medical Tubing, at Spectrum Plastics Group, was among the more than one dozen experts interviewed for the feature. His full input is provided in the following Q&A:
Michael Barbella: What are the latest trends in micromolding technology and services?
Kenny Freitag: Companies are designing new products that feature miniaturization, high precision, and tight tolerances—all of which require micromolding. This is especially true for small parts in the life sciences and medical device industries, including implantables. Customers are also asking for new exotic, highly engineered materials rather than standard thermoplastics, especially for high-temperature applications. As features get smaller and smaller, Spectrum continuously improves its system controls and technologies to enable even greater control over shot size, measuring systems, and equipment monitors.
Barbella: What are customers demanding or expecting of their micromolded products and have these demands/expectations changed in recent years?
Freitag: Customers want precision and accuracy. They seek molders that will share their deep knowledge and experience about material science and molding technologies during the design for manufacturing (DFM) process to make their designs better and easier to produce. They expect us to find solutions to their toughest design challenges—smaller parts, advanced materials, improved functionality, tighter tolerances, improved outcomes, and even regulatory assistance. Speed is also in the highest demand—both in manufacturing the product and getting it to market—so vertically integrated services are always an added benefit for customers.
Barbella: How have advances in materials impacted micromolding technology?
Freitag: Improving functionality and performance of a product is not always just a matter of relying on a better technology—it can also be accomplished using advanced materials that have specific chemical and physical properties. There is steady advancement in new or improved materials that can be micromolded with high precision—for example, polyether ether ketone, or PEEK, can be formulated with different fillers that enhance mechanical performance. Micromolding PEEK, however, requires a solid understanding of its melt characteristics, such as flow path. Temperature must also be tightly controlled. Using highly engineered materials that enhance functionality also gives engineers a wider range of design options.
Barbella: Please discuss the challenges and complexities involved in micromolding tooling design. How can these challenges be overcome?
Freitag: Probably the biggest challenge is miniaturizing parts and duplicating them in a high-volume production and manufacturing environment. It is absolutely imperative to create robust and repeatable processes for mircomolding parts. Mold designs evolve as materials evolve. Miniaturization creates challenges with understanding material flow and shear, for example. Automation can certainly improve micromolding efficiency, but only if unit volumes are high enough. Tooling is very complex, so there is a real issue with finding tooling shops and vendors with skilled personnel.
Barbella: Design for manufacturability (DFM) is critically important in micromolding. How is this different than conventional DFM?
Freitag: It is mostly a challenge of scale. Because we are making smaller parts, we are putting more thought into the tool design and how we are going to manufacture these miniaturized components. Plastic mold flow analysis is different for a small part since less plastic is involved, therefore requiring more tooling design time and iterations. Perhaps the greatest focus is on mold design to ensure smooth and cost-effective production—with less space for gates and runners, sometimes the mold needs to be redesigned to better use the limited space.
Barbella: What medtech specialty (cardiology, wearables, orthopedics, etc.) presents the greatest challenge in to producing micro-molded parts and why? Which present the greatest opportunity?
Freitag: Minimally invasive procedures such as those used in cardiology and structural heart applications present the greatest challenge due to the fact the improved access and delivery systems are allowing device designers to incorporate more and smaller features. For example, a catheter that fits within a narrow vessel may house multiple lumens, each of which will contain wires, sutures, or electrodes. The corresponding molded components at the distal tip or housed within the handle of the delivery system will likewise have a high density of tiny features and cavities to manage and guide those respective components. This is where micromolding capabilities, alongside advanced extrusion and catheter manufacturing, becomes critical to the success of next-generation interventional devices.
Barbella: How do quality systems play a part in micromolding? Why is this so important part of the process?
Freitag: Measurement systems and metrology must be capable of detecting and/or fixing variances in these high-precision, tight-tolerance manufacturing environments. Integrated measurements are critical to the micromolding process. An efficient approach is measuring parts optically while they are being produced in-line, rather than offline. Non-contact vision inspection systems can detect flaws as small as a few microns. Any variances can be identified in real time and fixed, minimizing defective parts, material waste, and downtime.
Barbella: How might the medtech micromolding industry evolve over the next five years?
Freitag: Parts will continue to become smaller and more complex, challenging the limits of molding technologies. Materials will be developed that are specifically for micromolding processes that have greater functionality and can hold tighter tolerances. Ultimately, the Internet of things will allow the integration of sensor technologies, machine-to-machine communication, artificial intelligence, and cloud-based platforms to become integrated into micromolding systems, maximize production quality and efficiency, and reduce operational costs and downtime. Different manufacturing technologies (machining, lasers, additive manufacturing, and molding) are also being combined in creative ways to create “hybrid equipment” where these technologies can complement each other in making a part.