Michael Barbella, Managing Editor05.13.20
It will be different next time. Much different, hopefully.
Future pandemics are unlikely to resemble the disjointed patchwork of COVID-19 mitigation efforts implemented worldwide by lawmakers and health officials. Instead, they possibly will involve more coordinated responses and widespread testing, as well as technological innovations inspired by design thinking.
Such thinking could result in redesigned public spaces that double as treatment areas during an outbreak and airports with more security screening lanes. Hospitals are likely to rethink conventional schematics too, potentially employing such pandemic-busting features as negative pressure zones, special patient evaluation areas, telehealth centers, and flexible-use rooms (i.e., those easily modified between acute care and ICU).
With the COVID-19 threat unlikely to abate in the fore- seeable future, design thinking could potentially become a powerful tool in helping to better manage and control forthcoming pandemics. The approach is already being used to improve healthcare equity and spawn creative solutions to medicine’s most pressing dilemmas.
MPO’s January/February feature, “Inventive Interaction,” explores the various forces at play in medtech R&D. Dorota Shortel, CEO of Simplexity Product Development, was among the various experts interviewed for the story. Her full input is provided in the following Q&A.
Barbella: Are startups better suited than their mid-size or large OEM cohorts to develop medical technology that addresses value-based healthcare solutions? If so, how? (Or, if not, why not?)
Dorota Shortel: In our experience, we see that startups are more likely to develop game-changing technology that addresses value-based healthcare solutions. The reason is that they are not constrained by a product line, core business, or reimbursement model that can sometimes occur at larger organizations, especially with the regulatory restrictions inherent to medical device development.
Barbella: Can a company effective tackle a healthcare need/problem and devise a solution based on its own internal R&D? Please explain.
Shortel: A company that is working directly with clients and understanding user needs in the healthcare space can absolutely tackle a healthcare problem. Understanding what the market needs and is willing to pay for is the key. In terms of developing a solution based on their internal R&D, a company can take it to a certain level, based on their core expertise. Usually the most successful model we’ve seen is when a company leverages their core expertise and hires partners to complement expertise that they may not have in-house.
Barbella: How can companies “innovate the process of innovation” to achieve more transformational products through R&D?
Shortel: I recently took a course called “Designing Organizations for Innovation and Creativity” via the Stanford Graduate School of Business, taught by Prof. Sarah Soule. One of my main takeaways is that companies need to actively design the architectures, routines, and culture that foster innovation. They cannot expect innovation to just happen because they want it to occur, rather it needs to be intentional. Some examples of this is allocating dedicated time for employees to innovate, having formal “hackathons” or innovation competitions, and evaluating every piece of customer feedback to see if it contains a snippet that could be a launching point for a possible new product.
Barbella: How will AI, big data, and digital health affect the R&D process going forward?
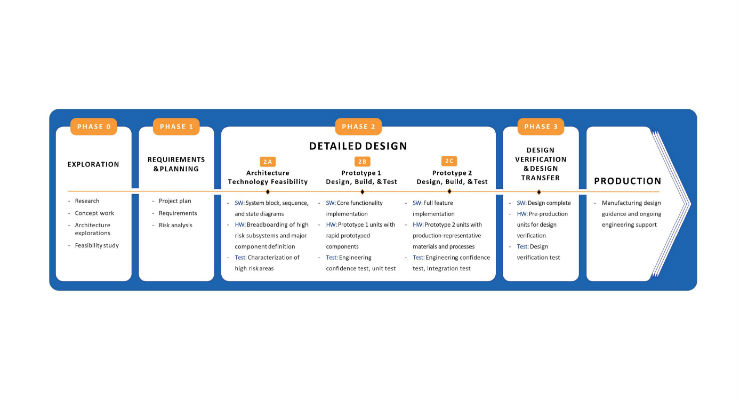
Shortel: The R&D process itself is often described as a phased, iterative approach, as described here. The high-level nature of how the process works will not change fundamentally. However, AI, big data, and digital health will mean that researchers and engineers are armed with a lot more information throughout the process. This means that they may be able to pull in data sets of user behaviors directly into their prototypes during the early phases of the process rather than waiting for a prototype to be complete before seeing how users would interact with it. With AI, prototypes will also be able to “learn” as they start to be put in user’s hands, providing more quantitative data to the developers, rather than relying on observation of user behavior.
Barbella: What is driving the need to outsource medtech R&D? Does the R&D process even need to be outsourced?
Shortel: The need to outsource medtech R&D is driven by two factors, time-to-market and expertise. Time-to-market is important for those companies realizing that getting a new product out and approved in the marketplace will give them an advantage in terms of user adoption and capturing market share. In that case of expertise, it helps the company to augment internal expertise with those of outside consultants and design firms. They get the benefit of leveraging knowledge across industries outside of their own and having expertise that is outside of their core applied to their products. It saves them the time and money of having to learn by pulling in best practices from the experts who have done it before. As to whether R&D really needs to be outsourced, if there are no schedule pressures nor expertise gaps, then it doesn’t have to, but in a majority of cases there is measurable value in outsourcing.
Future pandemics are unlikely to resemble the disjointed patchwork of COVID-19 mitigation efforts implemented worldwide by lawmakers and health officials. Instead, they possibly will involve more coordinated responses and widespread testing, as well as technological innovations inspired by design thinking.
Such thinking could result in redesigned public spaces that double as treatment areas during an outbreak and airports with more security screening lanes. Hospitals are likely to rethink conventional schematics too, potentially employing such pandemic-busting features as negative pressure zones, special patient evaluation areas, telehealth centers, and flexible-use rooms (i.e., those easily modified between acute care and ICU).
With the COVID-19 threat unlikely to abate in the fore- seeable future, design thinking could potentially become a powerful tool in helping to better manage and control forthcoming pandemics. The approach is already being used to improve healthcare equity and spawn creative solutions to medicine’s most pressing dilemmas.
MPO’s January/February feature, “Inventive Interaction,” explores the various forces at play in medtech R&D. Dorota Shortel, CEO of Simplexity Product Development, was among the various experts interviewed for the story. Her full input is provided in the following Q&A.
Barbella: Are startups better suited than their mid-size or large OEM cohorts to develop medical technology that addresses value-based healthcare solutions? If so, how? (Or, if not, why not?)
Dorota Shortel: In our experience, we see that startups are more likely to develop game-changing technology that addresses value-based healthcare solutions. The reason is that they are not constrained by a product line, core business, or reimbursement model that can sometimes occur at larger organizations, especially with the regulatory restrictions inherent to medical device development.
Barbella: Can a company effective tackle a healthcare need/problem and devise a solution based on its own internal R&D? Please explain.
Shortel: A company that is working directly with clients and understanding user needs in the healthcare space can absolutely tackle a healthcare problem. Understanding what the market needs and is willing to pay for is the key. In terms of developing a solution based on their internal R&D, a company can take it to a certain level, based on their core expertise. Usually the most successful model we’ve seen is when a company leverages their core expertise and hires partners to complement expertise that they may not have in-house.
Barbella: How can companies “innovate the process of innovation” to achieve more transformational products through R&D?
Shortel: I recently took a course called “Designing Organizations for Innovation and Creativity” via the Stanford Graduate School of Business, taught by Prof. Sarah Soule. One of my main takeaways is that companies need to actively design the architectures, routines, and culture that foster innovation. They cannot expect innovation to just happen because they want it to occur, rather it needs to be intentional. Some examples of this is allocating dedicated time for employees to innovate, having formal “hackathons” or innovation competitions, and evaluating every piece of customer feedback to see if it contains a snippet that could be a launching point for a possible new product.
Barbella: How will AI, big data, and digital health affect the R&D process going forward?
Shortel: The R&D process itself is often described as a phased, iterative approach, as described here. The high-level nature of how the process works will not change fundamentally. However, AI, big data, and digital health will mean that researchers and engineers are armed with a lot more information throughout the process. This means that they may be able to pull in data sets of user behaviors directly into their prototypes during the early phases of the process rather than waiting for a prototype to be complete before seeing how users would interact with it. With AI, prototypes will also be able to “learn” as they start to be put in user’s hands, providing more quantitative data to the developers, rather than relying on observation of user behavior.
Barbella: What is driving the need to outsource medtech R&D? Does the R&D process even need to be outsourced?
Shortel: The need to outsource medtech R&D is driven by two factors, time-to-market and expertise. Time-to-market is important for those companies realizing that getting a new product out and approved in the marketplace will give them an advantage in terms of user adoption and capturing market share. In that case of expertise, it helps the company to augment internal expertise with those of outside consultants and design firms. They get the benefit of leveraging knowledge across industries outside of their own and having expertise that is outside of their core applied to their products. It saves them the time and money of having to learn by pulling in best practices from the experts who have done it before. As to whether R&D really needs to be outsourced, if there are no schedule pressures nor expertise gaps, then it doesn’t have to, but in a majority of cases there is measurable value in outsourcing.