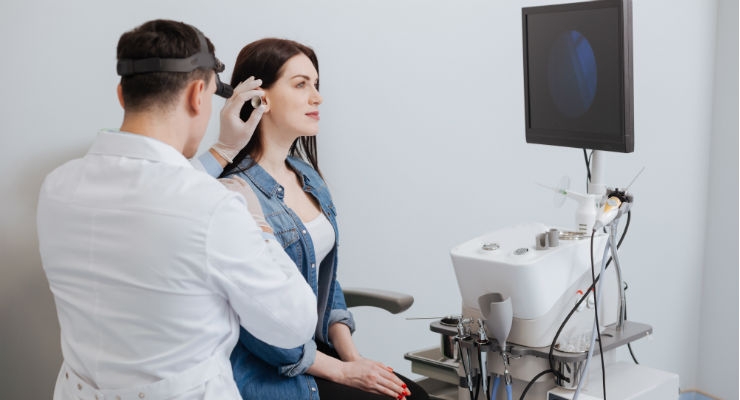
Nandini Roy Choudhury, Senior Research Consultant, Fact.MR01.14.19
With the advent of disruptive technology innovations, the ENT surgical devices landscape has evolved significantly in the areas of minimally invasive surgery, robotics, enhanced diagnostic imaging, and surgical visualization. Owing to the shift from traditional marketing model to care for delivery model in the medtech industry, over the past few years, several value-based ENT surgical devices have been introduced to improve patient care and aid in rapid recovery.
Augmented visualization, minimally invasive robotic surgery, 3D-guided surgery, diagnosis through high-quality microscopy, and digitalized interpretations are a few of the hot topics in the ENT surgical devices landscape.
Disorders related to ear, nose, and throat significantly decrease the quality of life and are the most common reasons for frequent hospital visits. Further, the number of hospital visits related to ENT problems continues to rise in the wake of the rising geriatric population. As for the global prevalence of different ENT disorders, for tinnitus the prevalence is found between 12-30 percent1 and for chronic rhinosinusitis between 5-16 percent2.
To accommodate the rising patient pool, ENT surgical devices are indispensable instruments in ENT clinics for the treatment of various ear-, nose- and throat-related disorders. Reliability and accuracy of these devices are highly important owing to the complexity of organs and their critical location. For instance, operations of sinus occur near the areas of eyes and brain, making them vulnerable to damage. However, improved imaging technology has significantly enhanced the display of the anatomical structures, thereby reducing the related risks of damage.
Minimal Invasion Aids in Cosmetic Appeal and Rapid Recovery
Minimally invasive procedures are highly embraced in the diagnosis and treatment of ENT disorders. Revolutionary minimally invasive ENT tools provide an alternative to major surgeries and involves less tissue damage, faster recovery, and reduced scarring as compared to major incisions.
ENT procedures, such as septoplasty, trans-oral robotic surgery, laryngeal surgery, tonsillectomy, and endoscopic sinus surgery have been performed with minimal invasion approaches over the past few years. Sophisticated instruments combined with robotics have transformed the treatment landscape of cancer associated with ear, nose, and throat. Tumor extraction via ear canal, transoral robotic surgery, and the potassium titanyl phosphate laser procedures are few of the highly reliable surgeries for cancer treatment.
Otorhinolaryngologists or ENT surgeons prefer laser surgery for the treatment of ENT disorders such as snoring, nasal obstruction, and cancer. Reliability and definitive results have led medical professionals, as well as patients, to increasingly rely on laser treatments. Among different ENT surgical devices, revenues associated with ENT surgical lasers were the highest and accounted for over one-fifth of the global share in 2018.
Advanced ENT visualization systems have enhanced the capabilities of laser treatments by providing high quality and optimal visualization for a smooth surgical process. Rising importance of visualization systems in ENT surgeries is likely to result in an impressive 9 percent growth in revenues of 2019.
Augmented Visualization and 3D Printing Technology
Developments in the medical imaging technology landscape have provided physicians with anatomical and functional data with greater specificity. While image-guided surgeries have been a conventional approach of surgeons, risks of potential errors have prevailed.
Augmented visualization (AR), on the other hand, can allow surgeons to annotate preoperative images and merge these with intra-operative images to broaden the scope of information to be used during actual surgical processes. AR also highlights critical “anti-targets” such as parts of the brain or important nerves and arteries, thereby reducing the surgical complications and associated morbidity cases.
With augmented visualization becoming a reality in ENT surgical approaches, leading providers have introduced augmented visualization in ENT surgical work platforms. Scopis GmbH, Leica Microsystems, Carl Zeiss, and Stryker are a few of the prominent players that have augmented visualization surgical systems for ENT surgeries in the market.
As ORs have become more and more connected, providers are introducing digital interfaces in the visualization system that provide an easy-to-use graphical user interface for medical professionals. In addition, a feature of integrated digital data management further aids in clinical data documentation.
Increasing penetration of 3D printing technology in the medtech industry is evident. Otolaryngologists use 3D-printed anatomical models for the pre-operational planning as well as for virtual planning and creation of surgical guides.
Globally, only a handful of case-series of patient data related to daily clinical practice of 3D printing in otorhinolaryngology is reported owing to the lack of comparative objective data. Albeit, at the budding stage, 3D printing holds potential in achieving imminent clinical applications in the field of ENT.
Regulatory Update
Medical technology innovations and clinical approvals go hand-in-hand. While major innovations lay in the pipeline, the ENT surgical devices industry has witnessed a number of FDA clearances in the past few years. A few examples of FDA clearances in 2017-2018 include Intersect ENT’s SINUVATM sinus implant, Medtronic’s surgical navigation system StealthStation, Acclarent’s TruDi ENT navigation system, and AERA Eustachian Tube Balloon Dilation System.
Medical devices including ENT surgical devices enter the market through the FDA 510(K) premarket clearance pathway. This forty-two-years-old regulatory law is set to undergo changes as announced by FDA in November 2018. The changes are likely to establish a more efficient medical devices clearance process wherein the baseline of decade-old predicate devices will be replaced by a contemporary baseline, which updates with technology advancements. Hence, the introduction of FDA 510(K) update is expected to introduce a new generation of safety, technology, and capabilities in medical devices.
The EU’s Medical Device Regulation (MDR) presented changes in 2017 that will come into force in 2020. EU manufacturers are vying to meet the changes in terms of recertification of legacy devices, as well as changes in the definition of regulated devices, safety measures, and risk management.
While the modernization of FDA 510(k) pathway and forthcoming enforcement of new EU MDR regulation is underway, the future of the ENT surgical devices market is likely to get impacted by these developments. While the new baseline can support the integration of disruptive technology innovations, higher standards of screening may impede or delay the development of ENT surgical devices in the coming years.
While the ENT surgical devices market was valued at over $2.3 billion in 2018, one-fourth of the global revenues were consolidated in Asia Pacific, excluding Japan (APEJ). Followed by North America, in terms of revenues, APEJ is presenting growing demand for medical technology owing to the presence of half the world’s population, increase in disease burden, income, and general awareness of health issues.
The medtech market in Asia Pacific portrays an expanding landscape while facing a shortage of supply and presence of divergent individual markets. While entering developed markets is easier due to established supply chain and favorable reimbursement policies, ENT surgical devices market players are seeking to broaden their reach and deliver high-quality products in developing markets.
All in all, the ENT surgical devices marketplace is growing at a steady pace wherein dynamism is maintained by the arrival of new devices with integrated exponential technologies that accommodate the care delivery focused business model of the medtech industry.
Note: The insights presented here are from a research study on ENT Surgical Devices Market by Fact.MR.
References
1 https://www.ncbi.nlm.nih.gov/pubmed/27246985
2 DOI: 10.1016/j.jaci.2011.08.004
Nandini Roy Choudhury is a senior research consultant for Fact.MR. She has a strong background in working across various verticals of healthcare (i.e., pharmaceuticals, medical devices, and healthcare IT). Within consulting engagements, Choudhury has experience in the development and advisory of corporate strategies in conjunction with the client’s top management. She has offered brand management and strategy consulting to two of China’s leading healthcare companies.
Augmented visualization, minimally invasive robotic surgery, 3D-guided surgery, diagnosis through high-quality microscopy, and digitalized interpretations are a few of the hot topics in the ENT surgical devices landscape.
Disorders related to ear, nose, and throat significantly decrease the quality of life and are the most common reasons for frequent hospital visits. Further, the number of hospital visits related to ENT problems continues to rise in the wake of the rising geriatric population. As for the global prevalence of different ENT disorders, for tinnitus the prevalence is found between 12-30 percent1 and for chronic rhinosinusitis between 5-16 percent2.
To accommodate the rising patient pool, ENT surgical devices are indispensable instruments in ENT clinics for the treatment of various ear-, nose- and throat-related disorders. Reliability and accuracy of these devices are highly important owing to the complexity of organs and their critical location. For instance, operations of sinus occur near the areas of eyes and brain, making them vulnerable to damage. However, improved imaging technology has significantly enhanced the display of the anatomical structures, thereby reducing the related risks of damage.
Minimal Invasion Aids in Cosmetic Appeal and Rapid Recovery
Minimally invasive procedures are highly embraced in the diagnosis and treatment of ENT disorders. Revolutionary minimally invasive ENT tools provide an alternative to major surgeries and involves less tissue damage, faster recovery, and reduced scarring as compared to major incisions.
ENT procedures, such as septoplasty, trans-oral robotic surgery, laryngeal surgery, tonsillectomy, and endoscopic sinus surgery have been performed with minimal invasion approaches over the past few years. Sophisticated instruments combined with robotics have transformed the treatment landscape of cancer associated with ear, nose, and throat. Tumor extraction via ear canal, transoral robotic surgery, and the potassium titanyl phosphate laser procedures are few of the highly reliable surgeries for cancer treatment.
Otorhinolaryngologists or ENT surgeons prefer laser surgery for the treatment of ENT disorders such as snoring, nasal obstruction, and cancer. Reliability and definitive results have led medical professionals, as well as patients, to increasingly rely on laser treatments. Among different ENT surgical devices, revenues associated with ENT surgical lasers were the highest and accounted for over one-fifth of the global share in 2018.
Advanced ENT visualization systems have enhanced the capabilities of laser treatments by providing high quality and optimal visualization for a smooth surgical process. Rising importance of visualization systems in ENT surgeries is likely to result in an impressive 9 percent growth in revenues of 2019.
Augmented Visualization and 3D Printing Technology
Developments in the medical imaging technology landscape have provided physicians with anatomical and functional data with greater specificity. While image-guided surgeries have been a conventional approach of surgeons, risks of potential errors have prevailed.
Augmented visualization (AR), on the other hand, can allow surgeons to annotate preoperative images and merge these with intra-operative images to broaden the scope of information to be used during actual surgical processes. AR also highlights critical “anti-targets” such as parts of the brain or important nerves and arteries, thereby reducing the surgical complications and associated morbidity cases.
With augmented visualization becoming a reality in ENT surgical approaches, leading providers have introduced augmented visualization in ENT surgical work platforms. Scopis GmbH, Leica Microsystems, Carl Zeiss, and Stryker are a few of the prominent players that have augmented visualization surgical systems for ENT surgeries in the market.
As ORs have become more and more connected, providers are introducing digital interfaces in the visualization system that provide an easy-to-use graphical user interface for medical professionals. In addition, a feature of integrated digital data management further aids in clinical data documentation.
Increasing penetration of 3D printing technology in the medtech industry is evident. Otolaryngologists use 3D-printed anatomical models for the pre-operational planning as well as for virtual planning and creation of surgical guides.
Globally, only a handful of case-series of patient data related to daily clinical practice of 3D printing in otorhinolaryngology is reported owing to the lack of comparative objective data. Albeit, at the budding stage, 3D printing holds potential in achieving imminent clinical applications in the field of ENT.
Regulatory Update
Medical technology innovations and clinical approvals go hand-in-hand. While major innovations lay in the pipeline, the ENT surgical devices industry has witnessed a number of FDA clearances in the past few years. A few examples of FDA clearances in 2017-2018 include Intersect ENT’s SINUVATM sinus implant, Medtronic’s surgical navigation system StealthStation, Acclarent’s TruDi ENT navigation system, and AERA Eustachian Tube Balloon Dilation System.
Medical devices including ENT surgical devices enter the market through the FDA 510(K) premarket clearance pathway. This forty-two-years-old regulatory law is set to undergo changes as announced by FDA in November 2018. The changes are likely to establish a more efficient medical devices clearance process wherein the baseline of decade-old predicate devices will be replaced by a contemporary baseline, which updates with technology advancements. Hence, the introduction of FDA 510(K) update is expected to introduce a new generation of safety, technology, and capabilities in medical devices.
The EU’s Medical Device Regulation (MDR) presented changes in 2017 that will come into force in 2020. EU manufacturers are vying to meet the changes in terms of recertification of legacy devices, as well as changes in the definition of regulated devices, safety measures, and risk management.
While the modernization of FDA 510(k) pathway and forthcoming enforcement of new EU MDR regulation is underway, the future of the ENT surgical devices market is likely to get impacted by these developments. While the new baseline can support the integration of disruptive technology innovations, higher standards of screening may impede or delay the development of ENT surgical devices in the coming years.
While the ENT surgical devices market was valued at over $2.3 billion in 2018, one-fourth of the global revenues were consolidated in Asia Pacific, excluding Japan (APEJ). Followed by North America, in terms of revenues, APEJ is presenting growing demand for medical technology owing to the presence of half the world’s population, increase in disease burden, income, and general awareness of health issues.
The medtech market in Asia Pacific portrays an expanding landscape while facing a shortage of supply and presence of divergent individual markets. While entering developed markets is easier due to established supply chain and favorable reimbursement policies, ENT surgical devices market players are seeking to broaden their reach and deliver high-quality products in developing markets.
All in all, the ENT surgical devices marketplace is growing at a steady pace wherein dynamism is maintained by the arrival of new devices with integrated exponential technologies that accommodate the care delivery focused business model of the medtech industry.
Note: The insights presented here are from a research study on ENT Surgical Devices Market by Fact.MR.
References
1 https://www.ncbi.nlm.nih.gov/pubmed/27246985
2 DOI: 10.1016/j.jaci.2011.08.004
Nandini Roy Choudhury is a senior research consultant for Fact.MR. She has a strong background in working across various verticals of healthcare (i.e., pharmaceuticals, medical devices, and healthcare IT). Within consulting engagements, Choudhury has experience in the development and advisory of corporate strategies in conjunction with the client’s top management. She has offered brand management and strategy consulting to two of China’s leading healthcare companies.