Brian Reilly, Business Development Director, Biomaterials, NuSil, an Avantor Company10.18.18
When selecting a silicone adhesive to use for medical device manufacturing, many factors need to be considered, including the substrates, their surface energy and available bond sites, the relevant aspects of the manufacturing process, and the end-use application. Let’s review some of the key considerations in choosing and using silicone adhesives for medical devices.
Considering Adhesive Options
One-Part Room Temperature Vulcanizing (RTV) Silicone Adhesives
One-part moisture-cure room temperature vulcanizing (RTV) adhesives are the most common silicone adhesives used in medical devices. They can be applied directly and do not require premixing. Heat is not required to cure, making them ideal for temperature-sensitive components. They are also well-suited for creating a seal and for bonding silicone to other silicones, metals, some plastics, and glass.
Two-Part RTV Silicone Adhesives
Two-part RTV silicone adhesives are among the most versatile silicone polymer compounds. In general, the cure time for two-part RTV adhesives is much faster compared to one-part RTV adhesives. Because two-part RTV adhesives do not require moisture to cure, they can meet unique assembly requirements such as forming bonds at interfaces that have little or no access to air. Two-part RTV adhesives are also ideal for temperature-sensitive components and assembly processes where a relatively low temperature must be maintained.

As an option, two-part RTV adhesives can be supplied in ready-to-use cartridges where the material is dispensed through a static mix tip and air is not introduced.
High-Temperature Vulcanizing (HTV) Silicone Adhesives
HTV silicone adhesives are formulated specifically to cure with the application of heat and are only available in two-part formulations. HTV adhesives are ideal for bonding parts that are not heat-sensitive or when a quick cure is desired to save time in device assembly.
All the features and advantages of two-part RTV adhesives apply to HTV adhesive formulations, and the cure characteristics of HTV adhesives offer several additional advantages: They cure rapidly when applying heat and have a work time ranging from 15 minutes to greater than 24 hours, depending on the formulation.
Solvent-Based RTV and HTV Silicone Adhesives
Adhesives can be supplied dispersed in a compatible solvent to help facilitate a specific process or application of the adhesive. Its rheology is ideal for thin sections or small intricate areas where the adhesive needs to reach a location that might otherwise be hard to access. This is especially useful as today’s medical devices continue to get smaller in size, and the application of adhesives requires more precision. Dispersed adhesives are available in one- or two-part formulations using both RTV or HTV chemistries.

For ease of use, adhesives can be supplied dispersed in a compatible solvent to help facilitate a specific process or application of the adhesive.
Considering Substrates and Proper Surface Preparation
Proper preparation of the substrate surface is essential to forming a long-lasting bond and there are several variables to consider:
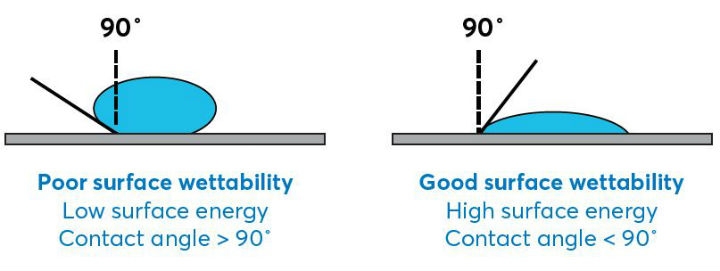
Figure 1
Other Important Factors
Measuring Adhesion
Many different physical, mechanical, and chemical forces act against a bond. Consequently, determining the acceptable failure point will differ from device to device and from manufacturer to manufacturer. Tests can be done in different conditions to simulate real-world use of the device to confirm that adhesion is suitable for the given application. It’s important to work with an adhesive manufacturer equipped to perform different tests to qualify adhesion, depending on the application. Adhesion tests can include several methods, such as lap shear, peel strength, and peel strength of cured bilayers from silicone dispersions.
Biocompatibility and Purity
Silicones used in medical adhesive applications should be biocompatible and conform with applicable ISO and USP testing protocols. Device designers should be sure to consider high-purity, medical-grade silicone adhesives with extensive regulatory support and Master Files submitted to the U.S. Food and Drug Administration (FDA) and international authorities. Master File (MAF) and GMP information on silicone adhesives support medical device manufacturers in their regulatory filings with details about ingredients, manufacturing, processing, packaging, and storage. In addition, the silicone adhesive manufacturer’s material should be designed and manufactured to strict purity standards. The process should start with carefully selected raw materials and utilize proven purification technology to produce the most refined intermediates and purest finished products, with low volatility levels.
Conclusion
Given the possible variations in premium silicone adhesive formulations and different application demands, it’s helpful to collaborate with a silicone adhesive manufacturer that offers a wide range of customization and product development services to meet specific device and silicone manufacturing requirements. The formulator should provide customization from the molecular level through formulation and packaging. Using state-of-the-art capabilities in R&D and molecular characterization, silicone chemistries can be modified to accommodate adhesion, substrate compatibility, performance characteristics, and cure profiles. Custom test methods can be adopted or developed to help translate the application needs and confirm that products meet specifications on a batch-to-batch level. For further customization, advanced manufacturing processes and proprietary equipment enable production in small batches up to very large batches at mass scale to support complete product commercialization.
Finally, medical device manufacturers should work with a silicone adhesive manufacturer that offers access to experienced chemists, engineers, regulatory experts, and technical specialists. This can be beneficial when trying to meet tight design and production parameters, giving medical device manufacturers a competitive advantage when working with medical silicone adhesives for demanding applications.
For additional resources and a downloadable white paper on the adhesives selection process, visit www.nusil.com/siliconeadhesives.
Considering Adhesive Options
One-Part Room Temperature Vulcanizing (RTV) Silicone Adhesives
One-part moisture-cure room temperature vulcanizing (RTV) adhesives are the most common silicone adhesives used in medical devices. They can be applied directly and do not require premixing. Heat is not required to cure, making them ideal for temperature-sensitive components. They are also well-suited for creating a seal and for bonding silicone to other silicones, metals, some plastics, and glass.
- Cure characteristics: Cure rates of one-part moisture-cure RTV adhesives are influenced by several factors, including humidity levels, cure temperature, thickness of adhesive applied, and surface area exposed to air. Moisture-laden airflow needs access to the adhesive in order to cure thoroughly, and “leaving groups” (byproducts of the curing process, such as mild acids, alcohols, or other volatile substances that evaporate from the material) need to be evacuated from the silicone.
- Rheology: These adhesives are available in self-leveling formulations that flatten or spread out over time, as well as thixotropic, or non-slump, formulations that hold shape over time.
- Application considerations: One-part RTV adhesives tend to have slower cure rates compared to two-part RTV adhesives. Due to leaving groups, the adhesive may shrink during cure. This type of adhesive is easily dispensed with no mixing required, and the product can be packaged in convenient resealable containers.
- Customization: One-part RTV adhesives have customizable attributes and their adhesion properties can be tuned to specific substrates. Viscosity can be adjusted to desired flow characteristics. Fillers can be added to match color or enhance properties such as radiopacity.
Two-Part RTV Silicone Adhesives
Two-part RTV silicone adhesives are among the most versatile silicone polymer compounds. In general, the cure time for two-part RTV adhesives is much faster compared to one-part RTV adhesives. Because two-part RTV adhesives do not require moisture to cure, they can meet unique assembly requirements such as forming bonds at interfaces that have little or no access to air. Two-part RTV adhesives are also ideal for temperature-sensitive components and assembly processes where a relatively low temperature must be maintained.
- Cure characteristics: The two-part RTV curing process is initiated by mixing two components together in a 1∶1 mix ratio and is not dependent on atmospheric moisture. The cure rate is influenced by heat, rather than exposure to air, and is not limited by the thickness of the adhesive because it cures uniformly. Practically no byproducts or leaving groups are released during the curing process.
- Rheology: Two-part RTV adhesives are available in non-slump formulations with moderate to high extrusion rates, enabling the adhesive to be easily dispensed.
- Application considerations: Cure rate can be accelerated by increasing temperature and shrinkage is minimal. Typical work times range from 15 minutes to two hours, depending on temperature. As an option, the product can be supplied in ready-to-use cartridges (where the material is dispensed through a static mix tip and air is not introduced).
- Customization: Similar to one-part RTV adhesives, two-part RTV adhesives can also be customized to meet specific end-application requirements.

As an option, two-part RTV adhesives can be supplied in ready-to-use cartridges where the material is dispensed through a static mix tip and air is not introduced.
High-Temperature Vulcanizing (HTV) Silicone Adhesives
HTV silicone adhesives are formulated specifically to cure with the application of heat and are only available in two-part formulations. HTV adhesives are ideal for bonding parts that are not heat-sensitive or when a quick cure is desired to save time in device assembly.
All the features and advantages of two-part RTV adhesives apply to HTV adhesive formulations, and the cure characteristics of HTV adhesives offer several additional advantages: They cure rapidly when applying heat and have a work time ranging from 15 minutes to greater than 24 hours, depending on the formulation.
Solvent-Based RTV and HTV Silicone Adhesives
Adhesives can be supplied dispersed in a compatible solvent to help facilitate a specific process or application of the adhesive. Its rheology is ideal for thin sections or small intricate areas where the adhesive needs to reach a location that might otherwise be hard to access. This is especially useful as today’s medical devices continue to get smaller in size, and the application of adhesives requires more precision. Dispersed adhesives are available in one- or two-part formulations using both RTV or HTV chemistries.
- Cure characteristics: All the features and advantages of one- and two-part RTV and HTV adhesives apply to dispersed adhesive formulations.
- Rheology: The dispersed silicone adhesive easily spreads out in a thin, uniform layer that conforms to the underlying shape.
- Customization: Dispersed adhesives also have customizable attributes to enhance process and performance characteristics.
- Application considerations: In a solvent system, a low-viscosity dispersion can be easily applied by spraying, wiping, dipping, or screen printing. Work times can be extended when the adhesive is solvated. The rate of solvent evaporation can be optimized with a combination of airflow, time, and heat. Cure can take place once the solvent has evaporated.

For ease of use, adhesives can be supplied dispersed in a compatible solvent to help facilitate a specific process or application of the adhesive.
Considering Substrates and Proper Surface Preparation
Proper preparation of the substrate surface is essential to forming a long-lasting bond and there are several variables to consider:
- Substrate cleaning: Proper cleaning methods remove contaminants that can directly impact the level of adhesion.
- Substrate surface finish: The surface finish can influence the wettability of the adhesive, thus affecting adhesion.
- Contact angle: The contact angle of the substrate surface helps quantify the wettability of the surface (i.e., the substrate’s acceptance of a coating). (See Figure 1)
- Surface treatments: To help increase the substrate’s surface energy for better adhesion, several methods can be considered. Plasma treatment bombards the substrate surface with ions of a gas such as argon. Corona discharge technique is another treatment method that uses increasing voltage cyclically to generate a plasma known as “corona discharge.” Adhesive primers can also provide a dramatic increase in adhesion.

Figure 1
Other Important Factors
Measuring Adhesion
Many different physical, mechanical, and chemical forces act against a bond. Consequently, determining the acceptable failure point will differ from device to device and from manufacturer to manufacturer. Tests can be done in different conditions to simulate real-world use of the device to confirm that adhesion is suitable for the given application. It’s important to work with an adhesive manufacturer equipped to perform different tests to qualify adhesion, depending on the application. Adhesion tests can include several methods, such as lap shear, peel strength, and peel strength of cured bilayers from silicone dispersions.
Biocompatibility and Purity
Silicones used in medical adhesive applications should be biocompatible and conform with applicable ISO and USP testing protocols. Device designers should be sure to consider high-purity, medical-grade silicone adhesives with extensive regulatory support and Master Files submitted to the U.S. Food and Drug Administration (FDA) and international authorities. Master File (MAF) and GMP information on silicone adhesives support medical device manufacturers in their regulatory filings with details about ingredients, manufacturing, processing, packaging, and storage. In addition, the silicone adhesive manufacturer’s material should be designed and manufactured to strict purity standards. The process should start with carefully selected raw materials and utilize proven purification technology to produce the most refined intermediates and purest finished products, with low volatility levels.
Conclusion
Given the possible variations in premium silicone adhesive formulations and different application demands, it’s helpful to collaborate with a silicone adhesive manufacturer that offers a wide range of customization and product development services to meet specific device and silicone manufacturing requirements. The formulator should provide customization from the molecular level through formulation and packaging. Using state-of-the-art capabilities in R&D and molecular characterization, silicone chemistries can be modified to accommodate adhesion, substrate compatibility, performance characteristics, and cure profiles. Custom test methods can be adopted or developed to help translate the application needs and confirm that products meet specifications on a batch-to-batch level. For further customization, advanced manufacturing processes and proprietary equipment enable production in small batches up to very large batches at mass scale to support complete product commercialization.
Finally, medical device manufacturers should work with a silicone adhesive manufacturer that offers access to experienced chemists, engineers, regulatory experts, and technical specialists. This can be beneficial when trying to meet tight design and production parameters, giving medical device manufacturers a competitive advantage when working with medical silicone adhesives for demanding applications.
For additional resources and a downloadable white paper on the adhesives selection process, visit www.nusil.com/siliconeadhesives.