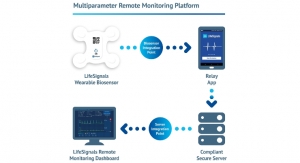
Bernard Tremaine, Owner, MDRS UK03.01.16
If your medical devices that are intended for CE Marking are already cleared for marketing by the U.S. Food and Drug Administration and do not have any innovative, new, or unique Indications to Use, then the devices would likely fit within the European Class IIa or IIb classifications. This article will therefore focus on these 510(k)-type devices while upcoming features will cover the lower-risk Class I devices [which would normally be 510(k) exempt] as well as the higher-risk Class III devices. The first part of this series can be viewed here.
The first step would be to produce a matrix linking the Index/Contents of the data in the 510(k) submission to the Index/Content of the CE Marking Technical File. The second step would then be to identify and list the missing data that the 510(k) submission file needs to make it EU compliant. The amended data file would then serve as the CE Marking Technical File submission data. The sections of a 510(k) submission, where deficiencies are mostly identified by European Notified Bodies when conducting a review against the CE Marking requirements, are listed below as 1 through 5. This is by no means an exhaustive list of where deficiencies could be found, but these are the areas where non-conformances most commonly occur when Notified Body reviews are undertaken.
Tech File Contents/Index
The EU Tech File Index would contain the following sections:
1. Standards, Specifications, MEDDEVs, etc.
This section is the single most important section of the Tech File, as it is the section where all the applicable Standards, Specifications, Formulations, and EU MEDDEVs that apply in full or part to the devices are identified. It is vitally important that all the “main” standards applicable to the devices are identified, together with any particular, secondary, and other standards that might also apply. Whatever method a manufacturer has to assist in the identification of applicable standards, the EU Commission’s website of harmonized standards should be used as part of that process. It is also important to ensure that the latest version of applicable standards is being applied and where this is not the case, then a rationale should be documented as justification.
Of equal importance is the identification of all applicable EU MEDDEV guidance documents. These “guidance” documents are published by the EU Commission and are so important that even though they are referred to as being “guidance” documents, application of their content is a mandatory requirement of CE Marking.
The main MEDDEV guidance documents cover the important areas of clinical data and vigilance. A subsequent article in this series will cover application of the MEDDEV on clinical data.
2. Essential Requirements
An Essential Requirements Checklist (ERC) is a mandatory requirement of any CE Marking compliance strategy. An ERC is simply a “signpost” to which compliance data/document is the evidence of compliance with each applicable Essential Requirement. An example relating to ER number 6 is provided below:

There are around 60 individual ERs and a compliant ERC would annotate each ER either applicable or not applicable (A/N). The third column would list the Standard or Specification, where applicable, which had been applied in order to meet the ER. Then the fourth column would give the reference ID of the Report/Document/Data that is the actual evidence of compliance with the particular Essential Requirement.
3. Labeling
The third most significant area where deficiencies occur is in labeling. Pitfalls include:
4. Post Sales Issues
Post sales issues must be controlled by documented procedures, which are reviewed and updated regularly and “challenged” to ensure their effectiveness. They must cover any additional or national requirements that apply to some EU countries. The important issue of vigilance reporting is covered by one of the MEDDEVs and is continually being reviewed and amended in order to make the process more effective. This particular MEDDEV (2.12/1) titled “Guidelines on a Medical Devices Vigilance System” is currently at version number 8.
Postmarket feedback is vitally important and must be shown to be effective in meeting the stated objectives of the process. The inputs to the process must include the correct balance of proactive and reactive elements and the outputs must feed into the design control, risk management, clinical evaluation, and improvement processes, and be shown to be effective.
Bernard Tremaine is the former CEO of MDQAS Ltd. (1994 to 2015 August). Bernard has 40+ years experience in the healthcare industry, with half of this time being spent at MEI in Milwaukee, Wis., where he held various positions in MEI’s European operations.
The first step would be to produce a matrix linking the Index/Contents of the data in the 510(k) submission to the Index/Content of the CE Marking Technical File. The second step would then be to identify and list the missing data that the 510(k) submission file needs to make it EU compliant. The amended data file would then serve as the CE Marking Technical File submission data. The sections of a 510(k) submission, where deficiencies are mostly identified by European Notified Bodies when conducting a review against the CE Marking requirements, are listed below as 1 through 5. This is by no means an exhaustive list of where deficiencies could be found, but these are the areas where non-conformances most commonly occur when Notified Body reviews are undertaken.
Tech File Contents/Index
The EU Tech File Index would contain the following sections:
- Declaration of Conformity
- Declaration of non-use of medicinal products
- Intended Purpose
- Performance Claims
- Product Description, including any variants and accessories
- Documented EU Classification and Attestation Route
- Manufacturing Flow Chart
- Full QA spec Index (DMR)
- Responsibilities and Authorities
- Device Life and Stability
- 1. Standards, Specifications, MEDDEVs, etc. applied in full or part
- 2. Essential Requirements (an ERC)
- Design Input Specified Requirements
- Design Verification and Validation including Certificates and Accreditations
- Risk Management
- Biocompatibility data
- 3. Labeling (including labels, instructions for use, controls, and language translation process)
- 4. Post Sales Issues (including post sales procedures, postmarket feedback process, product recall, vigilance reporting, and significant change notification process)
- 5. Clinical Data
1. Standards, Specifications, MEDDEVs, etc.
This section is the single most important section of the Tech File, as it is the section where all the applicable Standards, Specifications, Formulations, and EU MEDDEVs that apply in full or part to the devices are identified. It is vitally important that all the “main” standards applicable to the devices are identified, together with any particular, secondary, and other standards that might also apply. Whatever method a manufacturer has to assist in the identification of applicable standards, the EU Commission’s website of harmonized standards should be used as part of that process. It is also important to ensure that the latest version of applicable standards is being applied and where this is not the case, then a rationale should be documented as justification.
Of equal importance is the identification of all applicable EU MEDDEV guidance documents. These “guidance” documents are published by the EU Commission and are so important that even though they are referred to as being “guidance” documents, application of their content is a mandatory requirement of CE Marking.
The main MEDDEV guidance documents cover the important areas of clinical data and vigilance. A subsequent article in this series will cover application of the MEDDEV on clinical data.
2. Essential Requirements
An Essential Requirements Checklist (ERC) is a mandatory requirement of any CE Marking compliance strategy. An ERC is simply a “signpost” to which compliance data/document is the evidence of compliance with each applicable Essential Requirement. An example relating to ER number 6 is provided below:
3. Labeling
The third most significant area where deficiencies occur is in labeling. Pitfalls include:
- Not having identified and applied the European harmonized standard that governs “Symbols used on medical devices” (EN 980 & EN 15223-1)
- Not having identified and applied the necessary symbol requirements from ER numbers 13.3.(x) and 13.6.(x) and other relevant articles and clauses of the directive (for example, Article 17 and Annex xii)
- Not having identified any additional requirements from any particular standards that might be applicable to the devices in question (for example, “60601-2-xx Part 2s,” where the device is electrical in nature).
- Not having identified any national specific additional requirements, including any necessary language translations
- Not having a procedure to control identification, application, and change of all the above, including necessary language translations
4. Post Sales Issues
Post sales issues must be controlled by documented procedures, which are reviewed and updated regularly and “challenged” to ensure their effectiveness. They must cover any additional or national requirements that apply to some EU countries. The important issue of vigilance reporting is covered by one of the MEDDEVs and is continually being reviewed and amended in order to make the process more effective. This particular MEDDEV (2.12/1) titled “Guidelines on a Medical Devices Vigilance System” is currently at version number 8.
Postmarket feedback is vitally important and must be shown to be effective in meeting the stated objectives of the process. The inputs to the process must include the correct balance of proactive and reactive elements and the outputs must feed into the design control, risk management, clinical evaluation, and improvement processes, and be shown to be effective.
Bernard Tremaine is the former CEO of MDQAS Ltd. (1994 to 2015 August). Bernard has 40+ years experience in the healthcare industry, with half of this time being spent at MEI in Milwaukee, Wis., where he held various positions in MEI’s European operations.