11.15.11
A new partnership is expanding Boston Scientific Corp.’s catheter business in Europe.
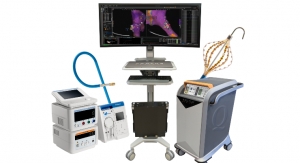
The Natick, Mass.-based company will market the CRI Amigo system, a remote-controlled catheter system and related accessories in select European countries through an exclusive agreement with Catheter Robotics Inc. in Mount Olive, N.J.
The Amigo system is compatible with certain Boston Scientific and other commercially available catheters and allows physicians to remotely maneuver diagnostic and ablation catheters designed to treat common cardiac arrhythmias during electrophysiology procedures.
Boston Scientific plans to market the Amigo system in Austria, Belgium, France, Germany, Luxembourg, the Netherlands, Portugal, Spain and Switzerland.
"To perform successful ablation procedures, electrophysiologists require precise control over catheter tip placement," said Jean Paul Albenque, M.D., Clinique Pasteur in Toulouse, France.
The Amigo system is designed to integrate easily into existing catheter labs for use in simple and complex procedures to diagnose and treat cardiac arrhythmias, according to the company. It connects to the handle of specific electrophysiological catheters and lets the physician remotely operate the catheter with a controller. The system is intended to allow full operation of all catheter functions while reducing a physician's radiation exposure during the procedure.
The Amigo system is compatible with Boston Scientific's Blazer and
Blazer Open-Irrigated radiofrequency ablation catheters and Chilli
II fluid cooled RF ablation catheter. The Blazer open-irrigated catheter was launched in CE Mark countries in May.
"The Amigo system complements our leading line of EP catheters, offering electrophysiologists an intuitive solution for remote operation while retaining the full functionality of their preferred ablation catheter platforms," said Michael Onuscheck, president of Boston Scientific’s European operations. "This agreement with Catheter Robotics is an important step in our continued focus on advancing cardiac ablation solutions for European physicians and their patients."
According to Boston Scientific, atrial fibrillation— an arrhythmia associated with a rapid rhythm in the upper chambers of the heart—affects more than 4.5 million Europeans. Patients most often are treated with anti-arrhythmic drugs. Ablation with an RF ablation catheter is increasingly becoming an option for patients who cannot tolerate the medication.
In the United States, the Blazer device is an investigational device and has not yet been approved by the U.S. Food and Drug Administration.
The Natick, Mass.-based company will market the CRI Amigo system, a remote-controlled catheter system and related accessories in select European countries through an exclusive agreement with Catheter Robotics Inc. in Mount Olive, N.J.
The Amigo system is compatible with certain Boston Scientific and other commercially available catheters and allows physicians to remotely maneuver diagnostic and ablation catheters designed to treat common cardiac arrhythmias during electrophysiology procedures.
Boston Scientific plans to market the Amigo system in Austria, Belgium, France, Germany, Luxembourg, the Netherlands, Portugal, Spain and Switzerland.
"To perform successful ablation procedures, electrophysiologists require precise control over catheter tip placement," said Jean Paul Albenque, M.D., Clinique Pasteur in Toulouse, France.
The Amigo system is designed to integrate easily into existing catheter labs for use in simple and complex procedures to diagnose and treat cardiac arrhythmias, according to the company. It connects to the handle of specific electrophysiological catheters and lets the physician remotely operate the catheter with a controller. The system is intended to allow full operation of all catheter functions while reducing a physician's radiation exposure during the procedure.
The Amigo system is compatible with Boston Scientific's Blazer and
Blazer Open-Irrigated radiofrequency ablation catheters and Chilli
II fluid cooled RF ablation catheter. The Blazer open-irrigated catheter was launched in CE Mark countries in May.
"The Amigo system complements our leading line of EP catheters, offering electrophysiologists an intuitive solution for remote operation while retaining the full functionality of their preferred ablation catheter platforms," said Michael Onuscheck, president of Boston Scientific’s European operations. "This agreement with Catheter Robotics is an important step in our continued focus on advancing cardiac ablation solutions for European physicians and their patients."
According to Boston Scientific, atrial fibrillation— an arrhythmia associated with a rapid rhythm in the upper chambers of the heart—affects more than 4.5 million Europeans. Patients most often are treated with anti-arrhythmic drugs. Ablation with an RF ablation catheter is increasingly becoming an option for patients who cannot tolerate the medication.
In the United States, the Blazer device is an investigational device and has not yet been approved by the U.S. Food and Drug Administration.