Spectranetics Strengthens Peripheral Vascular Portfolio with Distribution Partnership
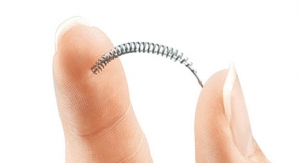
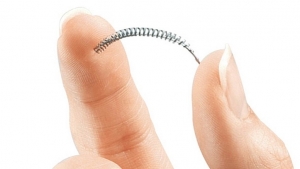
Spectranetics Corporation has formed a partnership agreement to commercialize its TAPAS catheter, a therapeutic infusion system that recently received U.S. Food and Drug Administration (FDA) 510(k) clearance. The product, intended for targeted infusion of therapeutic and diagnostic agents in the peripheral vasculature, is being launched through an agreement with ThermopeutiX Inc., in a limited number of U.S.-based and European hospitals.
The TAPAS catheter features two compliant occlusion balloons that enabletargeted local delivery of any physician-specified agent. One of the device’s unique features is the ability to adjust the treatment zone up to 300 millimeters, allowing for the treatment of long vessels with only one device and, in certain cases, multiple lesions with one device. The medication can beremoved from the catheter after treatment, providing localized intravascular treatment without systemic runoff. The TAPAS catheter can be used in conjunction with Spectranetics’ laser atherectomy or other interventional devices, according to the company. Clinical studies are currently underway to investigate whether debulking with laser or other atherectomy devices prior to drug delivery improves patient outcomes.
“I am very excited to begin working with the new TAPAS catheter,” said Lawrence Garcia, an M.D. at St. Elizabeth’s Medical Center in Boston, Mass. “Endovascular treatment is becoming an increasingly
targeted therapy approach for our patients. We currently do not have a device that can provide local drug delivery while minimizing systemic effects and be adaptable to a wide variety of patients and anatomies.
Although we are years away from the availability of other targeted therapeutic and diagnostic delivery systems in the United States, the TAPAS catheter provides anopportunity for local drug delivery for our patient needs now.”
The first procedures were performed by Eric Dippel, M.D., at Trinity Medical Center in Bettendorf, Iowa. “The TAPAS catheter provides a flexible platform for a variety of applications,” Dippel noted. “I was able to use the product to deliver different drugs tailored to the patients’ clinical situations.”
Spectranetics President and CEO Scott Drake said the partnership with ThermopeutiX will enable doctors to address a greater number of patients with peripheral vascular disease. He called the TAPAS catheter a “novel approach to delivering therapeutic agents” to the peripheral vasculature and said the company’s agreement is an “important next step” in helping clinicians address the challenges of peripheral vascular disease. “The TAPAS catheter…fits nicely with our portfolio of devices that allow physicians to cross, prepare and remove stenoses while preserving native vessels,”he said.
Based in Colorado Springs, Colo., Spectranetics develops single-use medicaldevices used in minimally invasive procedures within the cardiovascular system. The company’s products are sold in 40 countries throughout the world and are used to treat arterial blockages in both the heart and legs. Spectranetics devices also are used to help remove pacemaker and defibrillator leads.
The company’s vascular intervention products include a range of peripheral and cardiac laser catheters for ablation ofoccluded arteries above and below the knee and with coronary arteries. Spectranetics also markets aspiration and thrombectomy catheters for the removal of thrombus and support catheters to facilitate the crossing of coronary and peripheral arterial blockages.
ThermopeutiX is a private company based in San Diego, Calif., that has developed a dual-lumen catheter system that removes warm blood from the body through one lumen and delivers cold blood to the brain through a second lumen, according to a brief summary in Elsevier Business Intelligence. The technology is designed to be used primarily by interventional cardiologists and interventional radiologists to treat stroke, cardiac arrest and trauma.
Bayer HealthCare OpensNew Facility
The new year is turning out to be a busy one for Bayer HealthCare. Within the first few weeks of 2012, the Bayer AG subsidiary announced a new lab-sharing arrangement to help promising life sciences startup companies, and opened a newly expanded facility that will be used to research and develop medical devices for vascular occlusions.
The new facility mostly will be used by the company’s interventional franchise within its medical care division’s radiology and interventional business unit. It islocated in Coon Rapids, Minn.
“This expansion nearly doubles ourinfrastructure in Minnesota … and provides room for future growth in our cardiovascular and peripheral vascular disease device lines,” said Alan Main, president of Bayer HealthCare’s medical care business.
The 80,000-square-foot building houses research and development laboratories as well as manufacturing clean rooms. Executives hope that scientists and researchers take advantage of the new facility as the company moves to expand production of Cotavence, a European Union-blessed drug-eluting balloon treatment for peripheral artery disease. Bayer is hoping to make its drug-eluting treatment available worldwide.
New Medical Fibers Developed by Adhesives Research
There’s a new line of medical fibers on the market from Adhesives Research Inc. The Glen Rock, Pa.-based company has unveiled its ARmicron engineered medical fiber technology, a product designed specifically for wound healing, implantable devices and medical textiles. ARmicron’s engineered medical fiber technology was developed using the a melt-spin process known as high-definition micro-extrusion (HDME). Through this process, monofilament, multifilament and fine denier fibers are manufactured to customers’ precise specifications from a range of biocompatible, non-resorbable or bioresorbable polymers with tailored physical properties. The technology enables the development of multi-component fibers with highlyresolved internal domains or external submicron service architectural definitions that may be beneficial for directional cellularingrowth, according to the company.
Historically, medical fibers have been used in surgical meshes, sutures, stents and grafts. Adhesive Research can produce fibers with multi-polymer gradients, isolated cross sectional domains and precise internal and external surface structure definition to bring added benefits to other medical fiber applications. The firm’s HDME technology offers enhanced fiber design with increased manufacturing and supply chain control for medical devices, as well as a new design and functional flexibility for applications in controlled drug delivery and tissue engineering.
Founded in 1961, Adhesives Research designs, develops and manufactures custom components for companies serving the medical device and diagnostic, pharmaceutical, industrial, electronics, pulp and paper, engineered tapes, engineered fiber technologies and brand protection industries. The company is comprised of two businesses—Adhesives Research, which serves the medical device and diagnostics, pharmaceutical, electronics, engineered tapes, authentication and splicing markets; and ARx LLC, which develops and manufactures drug delivery platforms for the pharmaceutical and biotechnology industries.
A Year to Remember forC&J Industries
C&J Industries Inc. has much to celebrate this year. Not only is the contract manufacturer and injection molding company turning 50, it also recently completed its largest renovation project to date, a $6.8 million expansion of its facility in Meadville, Pa.
The expansion has increased the company’s Class VIII clean room molding and assembly space by more than 50 percent. The new assembly room, according to a brief news release from C&J, offers a complete suite of secondary services such as pad printing, sonic welding, heat staking and leak testing as well as manual andautomated assembly.
C&J has purchased best-in-class all-electric molding presses that have helped improve process control and reduced energy consumption. The addition of the newest molding presses brings the company’s total number of presses to 42; the machines provide a range of 20 tons to 720 tons of clamping pressure. Besides its newly-enlarged molding and assembly space, C&J also has its own in-house tool shop.
The company was founded by two toolmakers who named their new venture Meadville Precision Tool and Mold. The pair, according to company history, quickly gained notoriety for building high-precision plastic injection molded components and quality injection molds for such companies as AMP, Western Electric and Packard Electric. Fate stepped in during the early 1970s to introduce the firm to the plastic injection molding business. A historical account of the encounter appears on the company’s Website: “In the early 1970s (this is the stuff of legend) one of our customers commented that they were very pleased with the injection molds that we produced, however, they wanted us to qualify the tools prior to shipment. We responded, ‘But how can we qualify the molds—we don't have any plastic injection molding presses?’ Shortly thereafter a truck arrived at our loading dock and the truck driver had us sign for two slightly used injection molding presses. We were now in the plastic injection molding business.” Today, C&J specializes in the contract manufacturing of complex, high-precision injection molded components and assemblies for medical OEMs.
|
ISO CERTIFICATIONS: • AB SCIEX, a Framingham, Mass.-based developer of life sciences analytical technology, has achieved ISO 13485:2003 certification. The achievement covers the company’s manufacturing facility in Singapore as well as its research and development design center in Toronto, Ontario, Canada. “Achieving the ISO 13485 certification is an important measure toward realizing the vision of our LC/MS/MS systems to be cleared for use in clinics,” President Rainer Blair said. “The potential impact of mass spectrometry technologies on clinical diagnostics is significant.” • ISO 13485:2003 certification has been given to Cyberbond LLC, a global manufacturer of industrial adhesives headquartered in Batavia, Ill. The 14-year-old company operates facilities on six of the world’s seven continents. • ISO 13485:2003/AC:2009 certification has been awarded to Grifols, a Spanish pharmaceutical firm that develops and produces plasma-derived therapies and manufactures hospital pharmacy products, intravenous solutions, diagnostic tools and medical devices. The company is based in Barcelona, Spain, and operates industrial facilities in Melbourne, Australia; Guadalajara, Mexico; Murcia, Spain; Düdinquen, Switzerland; Los Angeles, Calif.; Melville, N.Y.; and Clayton, N.C. • Interstate Specialty Products, a Sutton, Mass.-headquartered manufacturer of high-performance custom gaskets and provider of precision custom die cutting services, has achieved ISO 13485:2003 certification. President John Savickas said the certification shows his company’s “commitment to continuous improvement, ability to meet customers’ needs and [dedication] to advancing its growth in the global marketplace.” • MedPro Safety Products Inc., a Lexington, Ky.-based developer of medication delivery and blood collection systems, has received ISO 13485 recertification. MedPro Chairman and CEO W. Craig Turner said the company expanded the scope of its latest certificate to include the design and manufacture of fluid delivery devices. “The updated scope reflected our expanding portfolio which will allow MedPro to further address three compelling yet unmet needs in the market: clinician safety, patient safety and community safety.” • ISO 13485:2003 certification has been granted to Nextremity Solutions LLC, a privately held medical device firm based in Colts Neck, N.J. The surgeon-founded company claims it offers a novel approach to the surgical correction of small bone deformity, degeneration and trauma. • Vishay Intertechnology Inc., a Malvern, Pa.-based manufacturer of discrete semiconductors (diodes, MOSFETs and infrared optoelectronics) and passive electronic components (resistors, inductors and capacitors), has achieved ISO 13485:2003 certification at its Yankton, S.D., facility. The company’s plant in the Mount Rushmore State makes inductors and transformers for implantable cardiac management devices such as pacemakers and defibrillators. |
|
BIOTECH in BRIEF The story of CytoSorbents Corporation has the potential to become a classic rags-to-riches tale. The Monmouth Junction, N.J.-based operating company of MedaSorb Technologies Corporation is introducing blood purification technology in Germany that could help the firm become a global leader in blood treatment/purification. Its HemoDefend platform removes toxins from blood and physiologic fluids by capturing harmful substances based on size and surface adsorption and specifically targeting antibodies, foreign antigens, cytokines, inflammatory mediators, free hemoglobin and bioactive lipids that potentially can be dangerous. Last year, the company received CE Mark approval to use its HemoDefend technology to remove high levels of cytokines from the bloodstream. The company currently is working to get additional approvals for the technology as well as find a partner that can help finance its climb to the top…A similar ascent is underway at American CryoStem Corporation, which has patented a way to collect, process and store adipose tissue (fat) derived adult stem cells for future use cellular treatments and therapies. The Red Bank, N.J.-based publicly-held firm has opened a new U.S. Food and Drug Administration-registered cGTP/cGMP tissue processing facility in Mount Laurel, N.J. Executives claim the company already has processed more than 100 fat tissue samples from patients for long-term storage…Flying under the radar is Proteus Biomedical Inc.’s pathway to commercial success, which involves the United Kingdom launch of its first product, Helius. Based on the company’s Raisin System, a sensor-enabled technology platform that collects and aggregates various behavioral, physiologic and therapeutic metrics, the Helius is described as a product that provides assurance and peace of mind to patients juggling complex medication regimens and health issues. Sold in retail pharmacies, Helius combines sensor-enabled tablets, a wearable physiologic monitor and a mobile health platform that enables patients to remain independent in their own homes…A new home is in store for genetic research firm Sequenom Inc. The San Diego, Calif.-based company is investing $18.7 million in a molecular diagnostics laboratory inside Research Triangle Park, N.C. Sequenom is developing a non-invasive test for Down Syndrome called the MaterniT21, which checks for the extra copy of chromosome 21 (trisomy 21)—the most common cause of the condition. Sequenom’s move is expected to generate 242 new jobs for the Park by 2015…New biotechnology jobs could be in store for Illinois, thanks to a deal between several Prairie State biotech groups and China’s Bio Pharmaceuticals Association. An agreement announced last fall will allow researchers and biotechnology companies in Illinois and the Middle Kingdom to “enhance business and education partnerships and collaborate on the scientific breakthroughs that will accelerate company growth and foster the creation of jobs,” according to a statement from Gov. Pat Quinn’s office. The state’s biotechnology industry, Quinn claims, employs 58,000 and is home to six of the top 20 pharmaceutical and life sciences companies…While those are impressive numbers, Massachusetts reigns supreme in biopharmaceutical employment. According to a recent report, the Commonwealth had the nation’s highest concentration of biopharmaceutical workers in 2010 (48,000) and attracted 23 percent of all U.S. venture capital dollars that year. A little more than half of those workers (26,000) are involved in R&D efforts. The report claims that Massachusetts only trailed California in biotech R&D employment growth from 2007 to 2010…The Treasure Coast of Florida is aiming to create its own viable biotechnology hub but the area is off to a slow start. As of December 2010, only 362 people worked in the biotechnology sector, comprising 0.2 percent of the local workforce, according to data from the Sunshine State’s Agency for Workforce Innovation. That figure more than quadrupled from a December 2009 estimate of 80, and the number could keep climbing if economic development officials’ plans come to fruition. Larry Pelton, president of the St. Lucie Economic Development Council, said he expects five new companies in the next five years to move to the Coast (identified as Indian River, St. Lucie and Martin counties), generating at least 4,000 to 5,000 new jobs. “Five years from now is going to be really exciting,” he said in published reports. “I think 10 years from now is going to be enormous. You won’t recognize our county.” |