Sam Brusco, Associate Editor02.03.23
Boston Scientific has gained U.S. Food and Drug Administration (FDA) approval for its LithoVue Elite single-use digital flexible ureteroscope, which according to the company is the first that can monitor intrarenal pressure in real-time during ureteroscopy procedures.
"Multiple studies have shown the importance of understanding intrarenal pressure during ureteroscopy procedures to mitigate potential complications, but until now, urologists have lacked an easy way to measure pressure in real-time," Ben Chew, M.D., M.Sc., associate professor, University of British Columbia, told the press. "This device represents an important and needed advancement in single-use ureteroscopes, which could help improve patient care and potentially lead to a clearer understanding of the impact of elevated intrarenal pressure on patient outcomes."
LithoVue Elite is comprised on the single-use digital flexible ureteroscope and StoneSmart Connect console. The new system build on many features of the original LithoVue.
A high-resolution digital chip and advanced imaging software aims to generate faster, sharper image quality and greater depth of field. Two programmable buttons on the ureteroscope handle allow direct video control and image capture without needing to coordinate with staff.
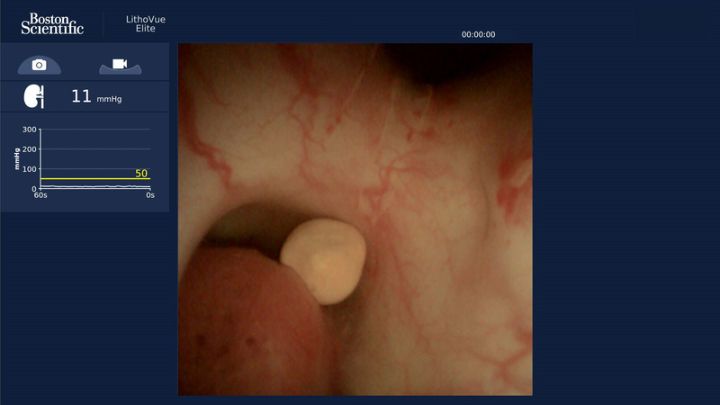
LithoVue Eilte monitor image.
The StoneSmart Connect console, a compact processing unit, works with existing operating room visualization towers and endoscopic monitors to reduce clutter and capital footprint as well.
"We're at a critical moment in kidney stone care where patient demand is outpacing the number of urologists needed to address it," said Meghan Scanlon, senior VP and president, Urology, Boston Scientific. "We designed the next-generation LithoVue Elite System to bring a greater level of precision and functionality in the way urologists diagnose and treat kidney stones, an increasingly common condition among adults. We are committed to developing clinical solutions, like the LithoVue Elite System, that are intended to make procedures more efficient, empower surgical decisions, optimize patient care and ultimately improve the treatment of kidney stone disease globally."
"Multiple studies have shown the importance of understanding intrarenal pressure during ureteroscopy procedures to mitigate potential complications, but until now, urologists have lacked an easy way to measure pressure in real-time," Ben Chew, M.D., M.Sc., associate professor, University of British Columbia, told the press. "This device represents an important and needed advancement in single-use ureteroscopes, which could help improve patient care and potentially lead to a clearer understanding of the impact of elevated intrarenal pressure on patient outcomes."
LithoVue Elite is comprised on the single-use digital flexible ureteroscope and StoneSmart Connect console. The new system build on many features of the original LithoVue.
A high-resolution digital chip and advanced imaging software aims to generate faster, sharper image quality and greater depth of field. Two programmable buttons on the ureteroscope handle allow direct video control and image capture without needing to coordinate with staff.

LithoVue Eilte monitor image.
The StoneSmart Connect console, a compact processing unit, works with existing operating room visualization towers and endoscopic monitors to reduce clutter and capital footprint as well.
"We're at a critical moment in kidney stone care where patient demand is outpacing the number of urologists needed to address it," said Meghan Scanlon, senior VP and president, Urology, Boston Scientific. "We designed the next-generation LithoVue Elite System to bring a greater level of precision and functionality in the way urologists diagnose and treat kidney stones, an increasingly common condition among adults. We are committed to developing clinical solutions, like the LithoVue Elite System, that are intended to make procedures more efficient, empower surgical decisions, optimize patient care and ultimately improve the treatment of kidney stone disease globally."













