Sam Brusco, Associate Editor01.23.23
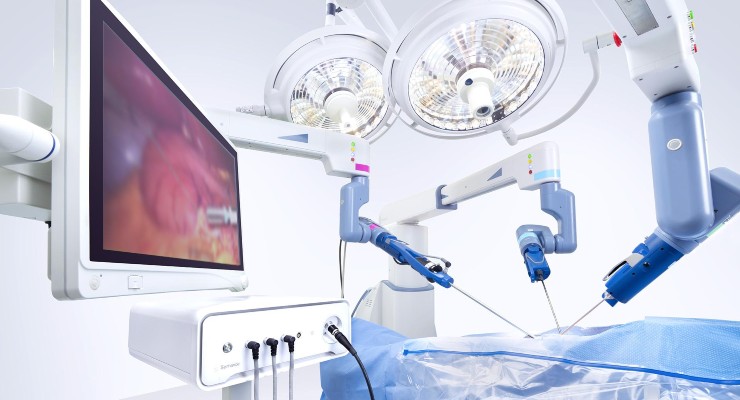
Asensus Surgical has received a CE mark to expand machine vision capabilities on its previously cleared Intelligent Surgical Unit (ISU).
The expanded ISU capabilities are now commercially available across the European Union, Japan, and U.S. Further, the Senhance robotic surgical system became one of the first to be approved the new, more rigorous EU Medical Device Regulation (MDR).
“We are thrilled to be able to offer these ground-breaking ISU capabilities to surgeons in the EU. Surgeon feedback from the expanded feature set across the U.S. and Japan has been tremendous, and we look forward to partnering with new and existing customers to help bring advanced real-time intraoperative digital tools into operating rooms throughout Europe,” Anthony Fernando, Asensus Surgical president and CEO told the press. “This is a significant milestone for the Company as our filing included a review of the Senhance Surgical System, both software and hardware, under the new, stricter EU MDR process. The fact that Senhance was one of the first robotic platforms to be cleared through the new process, in just under one year, is a testament to both the quality of our team and our technology.”
New features of the ISU include 3D measurement, digital tagging, image enhancement, and boosted intra-op camera control based on real-time data.
“I have used the first generation of the ISU with the Senhance Surgical System which seamlessly supports human-machine interaction and endoscopic camera control. This second generation of ISU features will bring surgeon support to a new level,” said Prof. (Saitama Med. Univ.) Dr. med. Dietmar Stephan of St. Marien-krankenhaus Siegen in Germany. The real-time measurement tools have broad applicability in surgery, and digital tagging will support communication between console and tableside indicating critical structures, e.g. clipping points or staple lines. With these new ISU features, I expect that the Senhance Surgical System will be a technology leader in the field of augmented intelligence in surgical robotics."
The expanded ISU capabilities are now commercially available across the European Union, Japan, and U.S. Further, the Senhance robotic surgical system became one of the first to be approved the new, more rigorous EU Medical Device Regulation (MDR).
“We are thrilled to be able to offer these ground-breaking ISU capabilities to surgeons in the EU. Surgeon feedback from the expanded feature set across the U.S. and Japan has been tremendous, and we look forward to partnering with new and existing customers to help bring advanced real-time intraoperative digital tools into operating rooms throughout Europe,” Anthony Fernando, Asensus Surgical president and CEO told the press. “This is a significant milestone for the Company as our filing included a review of the Senhance Surgical System, both software and hardware, under the new, stricter EU MDR process. The fact that Senhance was one of the first robotic platforms to be cleared through the new process, in just under one year, is a testament to both the quality of our team and our technology.”
New features of the ISU include 3D measurement, digital tagging, image enhancement, and boosted intra-op camera control based on real-time data.
“I have used the first generation of the ISU with the Senhance Surgical System which seamlessly supports human-machine interaction and endoscopic camera control. This second generation of ISU features will bring surgeon support to a new level,” said Prof. (Saitama Med. Univ.) Dr. med. Dietmar Stephan of St. Marien-krankenhaus Siegen in Germany. The real-time measurement tools have broad applicability in surgery, and digital tagging will support communication between console and tableside indicating critical structures, e.g. clipping points or staple lines. With these new ISU features, I expect that the Senhance Surgical System will be a technology leader in the field of augmented intelligence in surgical robotics."