Sam Brusco, Associate Editor12.05.22
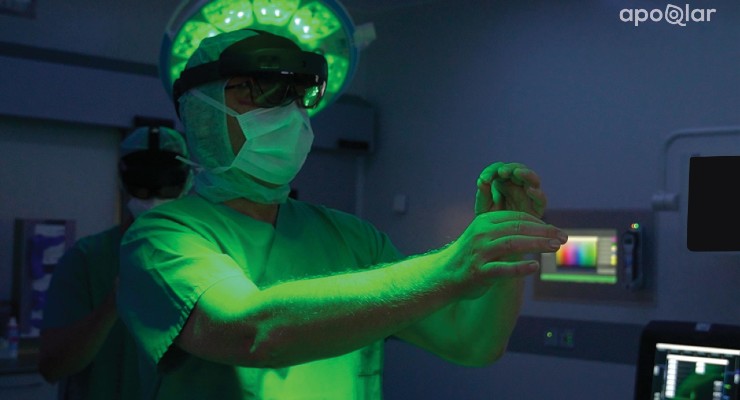
German medtech company apoQlar has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for VSI HoloMedicine, a mixed-reality software that helps plan complex procedures using immersive 3D holographic technology.
Availability is expected in Q2 2023 and the company is raising a Series A financing round to scale VSI HoloMedicine as a modern foundation of surgical care.
"With mixed reality, we are no longer bound to physical objects in a physical world. We can leverage digital objects and services on top of the real world for equal or greater utility and usually at a fraction of the cost. Mixed Reality is a completely new way for people, and in our case surgeons, physicians and technologists, to continue to experience the real world around them but with an entire virtual layer placed on top" Sirko Pelzl, co-founder and CEO of apoQlar told the press.
The mixed-reality software, according to apoQlar, gives surgeons an almost “X-ray vision” perspective on surgical planning processes with its 3D holographic tech. Surgeries can be planned in 3D and medical data can be visualized in and out of the operating room.
VSI HoloMedicine leverages Microsoft’s HoloLens 2 mixed-reality head-mounted display to transform flat CT, angio CT, MRI, CBCT, PET, and SPECT sources into interactive 3D holograms.
"FDA clearance marks a major milestone for us. We are a young company, but this serves as a true testament of our collective team mindset and diligence" said Liliana Duarte, COO of apoQlar. This is the latest achievement for apoQlar in their global expansion plan and serves as a tailwind for further market expansion efforts into South-East Asia, India, and the Gulf Coast States for 2023.
Availability is expected in Q2 2023 and the company is raising a Series A financing round to scale VSI HoloMedicine as a modern foundation of surgical care.
"With mixed reality, we are no longer bound to physical objects in a physical world. We can leverage digital objects and services on top of the real world for equal or greater utility and usually at a fraction of the cost. Mixed Reality is a completely new way for people, and in our case surgeons, physicians and technologists, to continue to experience the real world around them but with an entire virtual layer placed on top" Sirko Pelzl, co-founder and CEO of apoQlar told the press.
The mixed-reality software, according to apoQlar, gives surgeons an almost “X-ray vision” perspective on surgical planning processes with its 3D holographic tech. Surgeries can be planned in 3D and medical data can be visualized in and out of the operating room.
VSI HoloMedicine leverages Microsoft’s HoloLens 2 mixed-reality head-mounted display to transform flat CT, angio CT, MRI, CBCT, PET, and SPECT sources into interactive 3D holograms.
"FDA clearance marks a major milestone for us. We are a young company, but this serves as a true testament of our collective team mindset and diligence" said Liliana Duarte, COO of apoQlar. This is the latest achievement for apoQlar in their global expansion plan and serves as a tailwind for further market expansion efforts into South-East Asia, India, and the Gulf Coast States for 2023.