Sam Brusco, Associate Editor10.04.22
QT Medical has gained U.S. Food and Drug Administration (FDA) clearance for its PCA 500 resting 12-lead electrocardiogram (ECG) for pediatric patients. It was first cleared for adults in 2018 and the clearance expands PCA 500’s indication to newborns, infants, children, and adolescents.
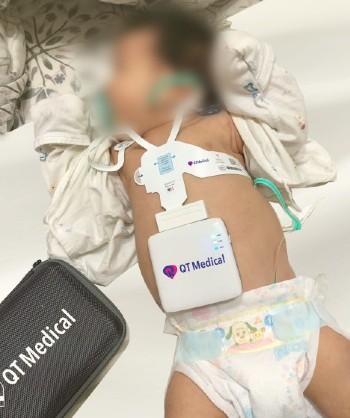
QT Medical announces FDA clearance of PCA 500, a resting 12-lead ECG, for use in pediatric patients.
PCA 500’s pre-positioned electrodes and compact recorder were initially funded by the NIH for an ECG designed for pediatric use. It provides digital, mobile, and cloud-based ECG management. The company plans to introduce PCA 500 to the pediatric market at this year’s American Academy of Pediatrics National Conference.
"The potential of PCA 500 in improving the heart health of children is extremely exciting. As a pediatric cardiologist, I know how difficult it is to get an ECG on a child. With PCA 500, we can make ECG technology easily accessible to all children. We believe this will make a difference in many lives, which is the exact reason why I founded QT Medical." Dr. Ruey-Kang Chang, CEO of QT Medical told the press.
The company will also announce Youth Xpress ECG, in partnership with Who We Play For, a non-profit organization dedicated to preventing sudden cardiac arrest of young people. It’s a mail delivery home 12-lead ECG testing service with interpretation by expert pediatric cardiologists. The company will also introduce Baby Xpress ECG, an at-home ECG screening service for newborns and infants with increased risks for long QT syndrome.

QT Medical announces FDA clearance of PCA 500, a resting 12-lead ECG, for use in pediatric patients.
"The potential of PCA 500 in improving the heart health of children is extremely exciting. As a pediatric cardiologist, I know how difficult it is to get an ECG on a child. With PCA 500, we can make ECG technology easily accessible to all children. We believe this will make a difference in many lives, which is the exact reason why I founded QT Medical." Dr. Ruey-Kang Chang, CEO of QT Medical told the press.
The company will also announce Youth Xpress ECG, in partnership with Who We Play For, a non-profit organization dedicated to preventing sudden cardiac arrest of young people. It’s a mail delivery home 12-lead ECG testing service with interpretation by expert pediatric cardiologists. The company will also introduce Baby Xpress ECG, an at-home ECG screening service for newborns and infants with increased risks for long QT syndrome.












