Sam Brusco, Associate Editor09.21.22
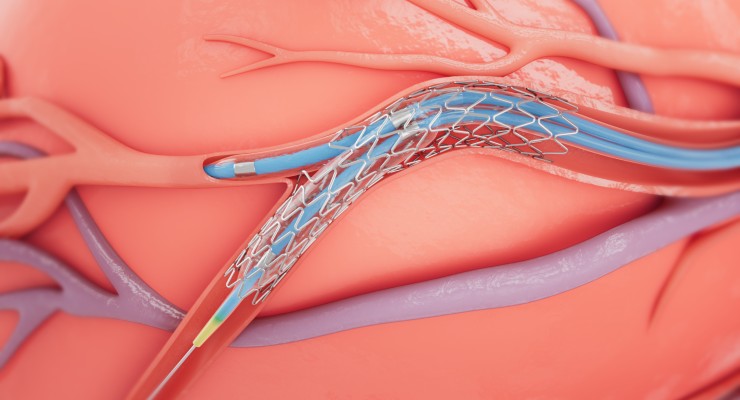
Medtronic received U.S. Food and Drug Administration (FDA) approval to treat non-left main bifurcation lesions using the provisional bifurcation stenting technique with the Onyx Frontier drug-eluting stent (DES) and Resolute Onyx DES.
Bifurcation lesions happen when plaque collects around the junction of two coronary arteries, and are considered tough to treat due to anatomical variations in vessels and difficulty with reaching the side branches. Stent attributes like those in Onyx Frontier and Resolute Onyx, according to Medtronic, can be important to optimize treating these lesions.
“The bifurcation expanded indication is yet another exciting milestone for our Coronary business this year,” said Jason Weidman, senior VP and president of Medtronic’s Coronary & Renal Denervation business told the press. “As the first and only medical device company to offer this indication to U.S. interventional cardiologists, Medtronic remains committed to investing in DES technology, clinical evidence, and physician education. We are looking forward to helping even more physicians access the tools they need to give their patients best-in-class care.”
Medtronic claims it is the only company to offer drug-eluting stents with a single-wire design, where a single strand of wire is formed into a sinusoidal wave to make a stent. According to the company, it optimizes treatment of complex bifurcation anatomy via vessel conformability, easier access to side branches, and the option to open the stent cell while keeping consistent stent scaffolding.
The Onyx Frontier and Resolute Onyx DESs are FDA and CE mark approved.
Bifurcation lesions happen when plaque collects around the junction of two coronary arteries, and are considered tough to treat due to anatomical variations in vessels and difficulty with reaching the side branches. Stent attributes like those in Onyx Frontier and Resolute Onyx, according to Medtronic, can be important to optimize treating these lesions.
“The bifurcation expanded indication is yet another exciting milestone for our Coronary business this year,” said Jason Weidman, senior VP and president of Medtronic’s Coronary & Renal Denervation business told the press. “As the first and only medical device company to offer this indication to U.S. interventional cardiologists, Medtronic remains committed to investing in DES technology, clinical evidence, and physician education. We are looking forward to helping even more physicians access the tools they need to give their patients best-in-class care.”
Medtronic claims it is the only company to offer drug-eluting stents with a single-wire design, where a single strand of wire is formed into a sinusoidal wave to make a stent. According to the company, it optimizes treatment of complex bifurcation anatomy via vessel conformability, easier access to side branches, and the option to open the stent cell while keeping consistent stent scaffolding.
The Onyx Frontier and Resolute Onyx DESs are FDA and CE mark approved.