Sam Brusco, Associate Editor06.08.22
Peripheral vascular intervention firm Okami Medical has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its LOBO-7 and LOBO-9 vascular occluders, the latest addition to the firm’s LOBO (LOw-profile Braided Occluder) vascular occlusion system.
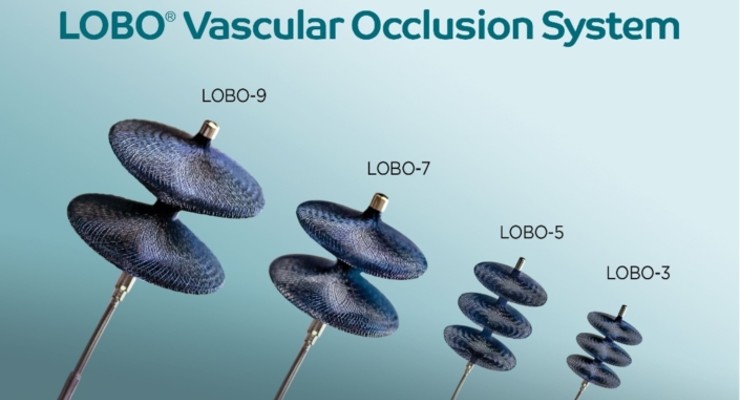
The system was purpose-built for fast, complete peripheral arterial target occlusion and now includes LOBO-3, LOBO-5, LOBO-7, and LOBO-9. The braided structure rapidly reduces flow and allows single-device blood vessel occlusion throughout the body. LOBO-7 and LOBO-9 are used for 5-7mm and 7-9mm diameter vessels.
The previously FDA-cleared LOBO-3 and LOBO-5 are meant for 1.5-3mm and 3-5mm vessels, respectively.
LOBO represents the next evolution in vascular embolization. As opposed to coils, which rely on the formation of an irregular mass with relatively large voids to try to occlude vessels, LOBO utilizes a high density, uniform small pore structure to occlude vessels nearly instantly,” Raj Pyne, MD, FSIR, interventional radiologist at Rochester Regional Health told the press. “The ability to occlude vessels quickly and consistently with a single-device not only improves procedure efficiency, but also is critical and often life-saving in situations such as trauma and unstable bleeding. The smaller occluders, LOBO-3 and LOBO-5, have demonstrated excellent performance and I am excited to see the larger sizes in use.”
“The FDA clearance of LOBO-7 and LOBO-9 is a testament to Okami Medical’s commitment to provide patients and physicians with access to advanced technologies that address the numerous challenges in peripheral vascular occlusion,” added Bob Rosenbluth, Ph.D., president and CEO of Okami Medical. “The LOBO system now includes a portfolio of devices that is specifically designed and built to quickly and completely occlude a wide range of vascular targets, thus eliminating the need for multiple embolic devices and enabling more efficient interventions.”
The system was purpose-built for fast, complete peripheral arterial target occlusion and now includes LOBO-3, LOBO-5, LOBO-7, and LOBO-9. The braided structure rapidly reduces flow and allows single-device blood vessel occlusion throughout the body. LOBO-7 and LOBO-9 are used for 5-7mm and 7-9mm diameter vessels.
The previously FDA-cleared LOBO-3 and LOBO-5 are meant for 1.5-3mm and 3-5mm vessels, respectively.
LOBO represents the next evolution in vascular embolization. As opposed to coils, which rely on the formation of an irregular mass with relatively large voids to try to occlude vessels, LOBO utilizes a high density, uniform small pore structure to occlude vessels nearly instantly,” Raj Pyne, MD, FSIR, interventional radiologist at Rochester Regional Health told the press. “The ability to occlude vessels quickly and consistently with a single-device not only improves procedure efficiency, but also is critical and often life-saving in situations such as trauma and unstable bleeding. The smaller occluders, LOBO-3 and LOBO-5, have demonstrated excellent performance and I am excited to see the larger sizes in use.”
“The FDA clearance of LOBO-7 and LOBO-9 is a testament to Okami Medical’s commitment to provide patients and physicians with access to advanced technologies that address the numerous challenges in peripheral vascular occlusion,” added Bob Rosenbluth, Ph.D., president and CEO of Okami Medical. “The LOBO system now includes a portfolio of devices that is specifically designed and built to quickly and completely occlude a wide range of vascular targets, thus eliminating the need for multiple embolic devices and enabling more efficient interventions.”