Sam Brusco, Associate Editor03.23.22
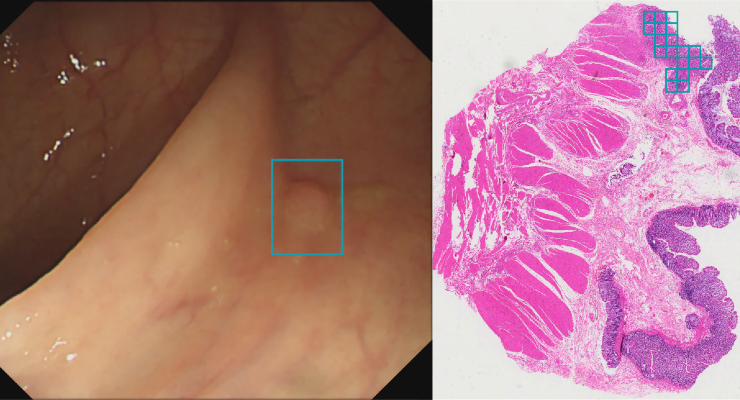
Artificial intelligence (AI) optical medical imaging diagnostics startup Wision A.I. earned U.S. Food and Drug Administration (FDA) 510(k) clearance for EndoScreener, its AI-assisted polyp detection software during colonoscopy. The company also has FDA presubmission in queue for histopathology AI software to help localize high-grade dysplasia (HGD) in whole-slide imaging (WSI)
A clinical study of EndoScreener showed 32.2% increased adenoma detection and 35.6% reduced adenoma miss rate.
The new histopathological AI product helps localize cancerous characteristics of HGD for colorectal specimens. Precise histological diagnosis is used to determent risk and surveillance interval, identifying high-risk signs leading to interval colorectal cancer. It can save the pathologist time to determine the correct diagnosis.
“Obtaining 510(k) Clearance from the FDA for EndoScreener and initiating a U.S. based investigational validation of the new tool for histopathological diagnosis demonstrates how our broadened, enriched AI portfolio can help to reduce colorectal cancer and improve patient outcomes,” JingJia Liu, co-founder and CEO of Wision A.I. told the press. “This announcement comes at a pivotal time during Colorectal Cancer Awareness Month, where we are working towards optimizing CRC detection through new, efficient, and precise methods. We believe that AI polyp detection in colonoscopy and AI HGD localization in histological diagnosis will greatly synergize and contribute to CRC screening and prevention.”
A clinical study of EndoScreener showed 32.2% increased adenoma detection and 35.6% reduced adenoma miss rate.
The new histopathological AI product helps localize cancerous characteristics of HGD for colorectal specimens. Precise histological diagnosis is used to determent risk and surveillance interval, identifying high-risk signs leading to interval colorectal cancer. It can save the pathologist time to determine the correct diagnosis.
“Obtaining 510(k) Clearance from the FDA for EndoScreener and initiating a U.S. based investigational validation of the new tool for histopathological diagnosis demonstrates how our broadened, enriched AI portfolio can help to reduce colorectal cancer and improve patient outcomes,” JingJia Liu, co-founder and CEO of Wision A.I. told the press. “This announcement comes at a pivotal time during Colorectal Cancer Awareness Month, where we are working towards optimizing CRC detection through new, efficient, and precise methods. We believe that AI polyp detection in colonoscopy and AI HGD localization in histological diagnosis will greatly synergize and contribute to CRC screening and prevention.”